Introduction
Melinjo tree (Gnetum gnemon L.) is a family of a Gnetaceae that is found in various regions in Indonesia, especially Sumatra and Java.1 The indigenous people of Indonesia have used melinjo as vegetables or processed delicacies. Melinjo seed has been shown to contain 15-19% protein.2 The seed extract contains some beneficial effects on human health. Several bioactive compounds are found in melinjo. The seed extracts contain various stilbenoids including trans-resveratrol (3,5,40-trihydroxy-trans-stilbene), gnetin C (GC; resveratrol dimer), gnetin L (GCderivative), gnemonoside A (GC-diglucoside), gnemonoside C (GC-monoglucoside), and gnemonoside D (GC-monoglucoside).3,4 These compounds have been reported to have potential utilization as drugs or antibody, antimicrobial, pigments or even anti-inflammatory agents.5,6,7 The seed extract may reduce uric acid and enhance lipid metabolism by escalating HDL cholesterol.8 The side effects of the melinjo seed extract for human consumption have not been found by recent researchers.9
According to Siswoyo et al.,10 the protein extract from melinjo seed possessed a very high antioxidant potential and was able to scavenge various type of free radicals. Protein hydrolysis of melinjo seeds can increase the ability of antioxidant and oxidative DNA damage protection activities.4,10 Therefore, it is necessary to further develop the use of melinjo both as a daily foods and as nutraceutical compound for human health. Recently, the use of natural substances in the development of nutraceutical compounds for human health is growing rapidly, primarily related to its role as an alternative to the use of chemical drugs that cause side effects on body health, such as Nerium oleander L,11 Nigella sativa L12 and Teucrium polium L,13 which contain high antioxidant potential.
According the International Diabetes Federation (IDF),14 there are 440 million cases of diabetes mellitus worldwide, and 87-91% of those cases are type-2 diabetes mellitus. Diabetes is caused by the increase of glucose to high levels , also known as hyperglycemia, which is directly involved in the process of free radical formation.15 Furthermore, the high accumulation of blood sugar level will also lead to complications by forming free radicals in human cells. High levels of glucose in diabetics are caused by the role of α-amylase; therefore, one method to overcome the accumulation of glucose in the blood is by inhibiting the performance of these enzymes. The inhibition activity of α-amylase and α-glucosidase is needed to manage the blood sugar level in people with diabetes.16 α-amylase and α-glucosidase are the key enzymes involved in starch breakdown into glucose that contribute to the increase of blood sugar level of diabetic patients. Inhibition of α-glucosidase is a therapeutic approach that aims to reduce postprandial blood glucose levels,17 with the inhibition of the performance of α-glucosidase so that the decomposition of oligosaccharides and disaccharides into monosaccharides will be delayed.18
Siswoyo et al.,10 also mentioned that the protein extract from melinjo seed indicated antihypertension activity through angiotensin I-converting enzyme (ACE) inhibition. The addition of the antioxidant compound for diabetic treatment will also help in reducing the possibility of complicated symptoms by scavenging the accumulation of free radicals in the human cell. This study is conducted to evaluate the double functions of melinjo seed as antioxidant and antidiabetic compounds based on the inhibitory activity of α-amylase and α-glucosidase. Melinjo seeds used in this study are derived from the different maturation levels (green, yellow, and red) of the seeds, which will be the most effective source of protein as antioxidants and antidiabetic compounds.
Materials and Methods
Material
Melinjo seeds at various stages of maturity from green (GM), yellow (YM) and red (RM) were obtained from the experimental farm at University of Jember, East Java, Indonesia in April 2016 (Figure 1). The reagents used in this study were as follows: 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulphonic acid) (ABTS•+), Trinitrobenzene sulfonic acid (TNBS), bovine serum albumin (BSA), 2-deoxy-D-ribose, pyrogallol, 3,5-Dinitrosalicylic acid (DNS), ferric chloride solution (FeCl3), hydrogen peroxide (H2O2), Tertiary Butyl Alcohol (TBA), trichloroacetic acid (TCA), ethylenediaminetetraacetic acid (EDTA), alcalase 2.4 L FG (EC 3.4.21.62),α-amylase (30 U/mg), and α-glucosidase (10 U/mg) procured from Sigma-Aldrich, Singapore.
 |
Figure 1: Melinjo seed in a different stage of maturation GM (green), YM (yellow), and RM (red) Protein Extraction and Hydrolysis Click here to View figure |
Protein extraction of melinjo seed samples was performed by grounding fresh seeds (1 g) Na2HPO4 (0.1 M, pH 7.0, 3 mL), followed by centrifugation (10.000 rpm, 4oC, 15 min). Bradford method19 was then used to determined soluble protein from the pooled supernatant. A portion of supernatant (5 μL) was added to a mixture containing aquadest (45 μL) and Bradford solution (950 μL). Absorbance was quantified by spectroscopy (Hitachi U-2900UV/VIS) at wavelength of 595 nm, and the results were compared with BSA standard to determine the dissolved protein content.
Protein extract (200 μL) was added to the alcalase 2.4L and phosphate buffer (pH 8, 300 μL) to perform the protein hydrolysis. The mixture was then incubated at 50oC for various durations (0-180 min), and the resulting mixtures were centrifuged (10.000 rpm, 25 °C,10 min). The degree of hydrolysis (DH) is determined using the TNBS method, as mentioned by Adler-Nissen.20 A portion of the sample (5 μL) was mixed with phosphate buffer (0.2 M, pH 8, 400 μL), TNBS (0.1%, 200 μL) and was then incubated (50°C, 30 min). The reaction was stopped by adding Na2SO3 (0.1 N, 400 μL) followed by cooling to room temperature prior to absorbance measurement at wavelength of 420 nm. The standard curve of L-leucine was used to determine amino acid concentrations. The percentage of DH is calculated by the following equation: DH (%) = h/htot x 100, where h = number of hydrolyzed peptide bonds and htot = the total number of equivalent protein peptide bonds.
Protein Profiling by SDS-PAGE
The profiling of protein hydrolysate from each seed (GM, YM, and RM) was determined using sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) method Laemmli.21 Protein extract (10 μg) from each sample was loaded into 15% polyacrylamide gel to perform the protein profile.
ABTS•+ (2,2’-azinobis 3-ethyl benzothiazole-6-sulfonic acid) Radical Scavenging
The antioxidant activity was performed based on the ABTS•+ radical scavenging activity. The assay of ABTS•+ radical scavenging activity was conducted through the method as described by Re et al.,22 The ABTS•+ was generated by the reacting ABTS•+ aqueous solution (7 mM) with potassium persulfate (2.45 mM) and incubated in the dark for 12–16 h. Before the assay, the ABTS solution was diluted with sodium phosphate buffered saline (0.2 M, pH 7.4) to absorb 0.70–0.75 AU at wavelength of 734 nm. The assay solution contained ABTS•+ solution (950 μL) with PBS (0.2 M, 30 μL) and protein extracts (20 μL). The absorbance was measured at wavelength of 734 nm. The antioxidant activity was calculated by the following formula: scavenging ABTS•+ (%) = [(Ac – As) / Ac] x 100%, Ac = absorbance control and As = absorbance sample. Glutathione (G-SH) was used as a positive control.
Hydroxyl Radical Scavenging
Hydroxyl radical scavenging activity was analyzed through the analysis described by Halliwell et al.,23 Protein extract (200 μL) was introduced with 2-deoxy-D-ribose (28 mM, 50 μL) in KH2PO4 (20 mM, pH 7.4), EDTA (1 mM 100 μL), FeCl3 (10 mM, 10 μL) H2O2 (1 mM, 10 μL) ascorbic acid (1 mM, 100 μL) and KH2PO4 (20 mM, pH 7.4, 530 μL) and was incubated at 37oC for 1 h. After the incubation, TBA (1%, 500 μL) and TCA (2.8%, 500 μL) were added to the solution followed by incubation for 30 minutes at 80oC. The absorbance was measured at wavelength of 532 nm using a spectrophotometer (Hitachi U-2900UV/VIS). Glutathione (G-SH) was used as a positive control.
Superoxide Radical Scavenging
Superoxide anion radical scavenging was analyzed by Tang et al.,24 The sample (200 μL) was added into Tris-HCl (50 mM, pH 8.2, 1.7 mL), incubated for 10 minutes, supplemented with pyrogallol (10 mM, 100 μL) in HCl (10 mM), and measured by a slope for 1 to 4 minutes at wavelength of 320 nm. Glutathione (G-SH) was used as a positive control.
Antidiabetic Activity Assay
The antidiabetic activity of melinjo seeds was performed based on the inhibition of α-amylase and α-glucosidase assay. The α-amylase inhibition was assayed using DNS procedure according to Hashim et al.,25 with a slight modification. To the sample (100 μL), α-amylase (0.1 U/mL) in Na2HPO4 (20 mM, pH 6.9, 150 μL) was added. The solution was pre-incubated for 15 min at 37oC, followed by the addition of soluble starch (1%, w/v, 250 μL). The solution was re-incubated for 15 min at 37oC. Hydrolysis was quenched by boiling the solution for 1 minute. DNS reagent (50 μL) was then added to determine the reduction in total sugars and absorbance was measured with a microplate reader at a wavelength of 540 nm.
The analysis of α-glucosidase inhibition refers to the method by Miyazawa et al.,26 Sample (200 μL) was added with maltose (0.25 M, 100 μL) KH2PO4 (0.1 M, pH 7.0, 190 μL) and α-glucosidase (1U/μL, 10 μL) and incubated at 37oC for 1 h. The solution was then brought to boil for 3 minutes. A portion of the mixture (235 μL) was procured and introduced with phenol buffer (0.1 N KH2PO4, Triton-X (3 mg/mL), and phenol (3 mg/mL), peroxidase (0.5 U/μL, pH 7.0, 5 μL), glucose oxidase (0.8 U/μL, 5 μL) and aminoantipyrine (1 mg/mL, 5 μL) and then, incubated at 37oC for 10 minutes. The absorbance was recorded at wavelength of 500 nm. Acarbose was used as a positive control.
Statistical Analysis
All the experimental results were analyzed statistically using standard deviation (±SD) and analysis of variance (ANOVA). The significant differences were determined by Duncan’s test at p < 0.05.
Result
Degree of Hydrolysis
The degree of hydrolysis (DH) of protein hydrolysate (RMH, YMH, and GMH) increased rapidly with the increasing of time of hydrolysis. Furthermore, the hydrolytic activity of alcalase weakened gradually with time when the reaction was longer than 180 min. The percentage of DH from each sample at 180 minutes showed a different value, with the highest percentage in the RMH (84.33%), followed by YMH (68.59%) and GMH (52.35%), which had the lowest hydrolysis degree value. This result might be related to the enzyme ability in breaking peptide bond down in each different proteins.
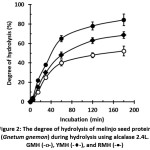 |
Figure 2: The degree of hydrolysis of melinjo seed protein (Gnetum gnemon) during hydrolysis using alcalase 2.4L. GMH (-o-), YMH (-¨-), and RMH (-·-) Click here to View figure |
Protein Profiling
The results showed (Figure 3) two dominant proteins in the melinjo seed protein with molecular weights of 30 and 12 kDa. There are differences in these two dominant protein patterns before and after hydrolysis (180 minutes). The most noticeable difference is the pattern of proteins with a molecular weight of 12 kDa. The 30 kDa protein is degraded into a protein with a lower molecular weight; thus, it is not seen in the electrophoresis gel at GM, YM, and RM. This proves that the melinjo seed protein has been hydrolyzed to a simpler protein. Hence, an increase in the ability of antioxidant activity and the inhibition activity of α-amylase and α-glucosidase is expected.
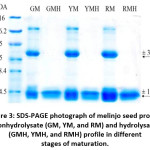 |
Figure 3: SDS-PAGE photograph of melinjo seed protein nonhydrolysate (GM, YM, and RM) and hydrolysate (GMH, YMH, and RMH) profile in different stages of maturation. Click here to View figure |
Free Radicals Scavenging Activities
The results of ABTS•+ radical reduction activities, hydroxyl radicals, and superoxide radicals in GM nonhydrolysate indicated EC50 values of 0.33, 7.83 and 2.36 µg/mL and in GMH showed EC50 values of 0.028, 0.05 and 0.11 µg/mL, respectively (Table 1).
Table 1: Free radical scavenging (EC50) of melinjo (Gnetum gnemon) seed protein nonhydrolysate and hydrolysate*
| Non-hydrolysate | Hydrolysate | G-SH | |||||
| GM | YM | RM | GMH | YMH | RMH | ||
| ABTS•+ | 0.33±0.01b | 0.45±0.01a | 0.47±0.02a | 0.03±0.01f | 0.05±0.00e | 0.11±0.01d | 0.22±0.08c |
| OH• | 7.83±0.02d | 10.96±0.02b | 12.46±0.01a | 4.36±0.01f | 5.71±0.02e | 6.80±0.02e | 8.24±0.12c |
| O2•- | 2.36±0.02d | 3.55±0.01c | 5.20±0.02a | 1.69±0.02f | 1.85±0.01e | 2.21±0.01d | 4.99±0.15b |
*EC50 value (µg/mL) is the effective concentration of antioxidant to inhibit 50% activity of radical ABTS•+, hydroxyl radical (OH•), and superoxide radical (O2•-). Glutathione (G-SH) was used as positive control. The values are the means (n=6) followed by standard deviation, and Duncan’s test at p<0.05, where the same letters in a row are not significantly different.
The YM nonhydrolysate inhibited ABTS•+, hydroxyl radicals and superoxide radicals with EC50 values of 0.45, 10.96 and 3.55 µg/mL, respectively, and YMH indicated EC50 values of 0.05, 5.71 and 1.85 µg/mL, respectively. RM nonhydrolysate has EC50 values of 0.47, 12.46 and 5.20 µg/mL for ABTS•+ radical inhibition, hydroxyl radical and superoxide radical, respectively, and RMH has EC50 values of 0.11, 6.80 and 2.21 µg/mL, respectively. These results indicated that the antioxidant activity of alcalase hydrolyzed melinjo protein varies depending on the extent of hydrolysis.
Inhibitory Activity of α-amylase and α-glucosidase
The results of the inhibitory activities of α-amylase and α-glucosidase in GM have IC50 values of 18.68 and 10.86 µg/mL, respectively (Table 2). GMH has IC50 values of 6.98 and 5.72 µg/mL, respectively. YM protein inhibits α-amylase and α-glucosidase activity with IC50 values of 35.46 and 32.88 µg/mL, respectively and YMH has IC50 values of 9.19 and 7.17 µg/mL, respectively. RM protein inhibits α-amylase and α-glucosidase activity with IC50 values of 36.10 and 35.78 µg/mL, respectively, while RMH has IC50 values of 12.55 and 8.52 µg/mL, respectively. The results of hydrolysis of the melinjo seed protein have increased inhibitory activity of α-amylase and α-glucosidase.
Table 2: The Inhibition of α-amylase and α-glucosidase (IC50) of protein melinjo seed (Gnetum gnemon) nonhydrolysate and hydrolysate
| non-hydrolysate | Hydrolysate | |||||||||||
| GM | YM | RM | GMH | YMH | RMH | |||||||
| α-amylase | 18.68±0.25d | 35.46±0.21b | 36.10±0.05a | 6.98±0.23g | 9.19±0.18f | 12.55±0.16e | 31.20±1.01c | |||||
| α-glucosidase | 0.86±0.10g | 32.88±0.15b | 35.78±0.26a | 5.72±0.05f | 7.17±0.15e | 8.52±0.25d | 19.54±1.22c | |||||
*IC50 value (µg/mL) is the concentration of protein that inhibits 50% activity of α-amylase and α-glucosidase. Acarbose was used as positive controls. The values are the means (n=6) followed by standard deviation, and Duncan’s test at p < 0.05, where the same letters in a row are not significantly different.
Discussion
Various proteins are being developed in the nutritional field, one of which is a protein that acts as an antioxidant.27 Antioxidants play a role in preventing cellular damage caused by free radical exposure.28 Melinjo seeds are known to have a fairly high protein content of 15-18%.2 Analysis of SDS-PAGE profile was performed to reveal protein distribution, which show antioxidant and antidiabetic activities. The results showed that the SDS-PAGE profile in melinjo seeds had molecular weights of 30 and 12 kDa. Both of these proteins were more dominant than other proteins. In hydrolyzed proteins, proteins with a molecular weight of 30 kDa were cut off by alcalase into simpler proteins so that they were not detected in an electrophoresis gel. Protein hydrolysis resulted in simpler protein yet increased protein solubility along with the increase of antioxidant activity ability.29
The inhibition of free radicals, hydroxyl, and superoxide tests were carried out to confirm antioxidant potential in proteins derived from each stage of melinjo seed maturation. All stages of maturation showed antioxidant activity with an EC50 value lower than 12.5 µg/mL protein (Table 1). Valco et al.,30 reported that natural resources containing IC50 values lower than 500 µg/mL have high potential as nutraceutical resources and antidiabetic agents. The hydrolyzed protein of melinjo seeds has antioxidant activity that is higher than protein before hydrolysis, reported by the research conducted by Siswoyo et al.,31 suggesting that proteins hydrolyzed using alcalase can increase the ability of proteins to ward off free radicals.
The increasing ability of hydrolyzed proteins in counteracting free radicals is related to the value of the DH, which is shown by the antioxidant activity.32,33 The inhibition activity of ABTS2+, hydroxyl, and superoxide radicals from hydrolyzed and nonhydrolyzed protein may indicate their antioxidant activities in inhibiting free radicals. The result (Table 1) shows that the hydrolyzed protein has EC50 values lower than nonhydrolyzed proteins. Hydrolysis proteins donate more electrons and hydrogen atoms from each active peptide that converts free radicals to more stable radical.34 It can be stated that hydrolysis using alcalase will cause the formation of shorter peptides (tri- and dipeptide) and free amino acids. Thus, peptides become more hydrophilic and more easily inhibit hydroxyl ABTS and superoxide radicals.35
Antioxidant compounds can contribute their hydrogen atoms into lipid radicals and convert them into stable forms as indicated by Valco et al.,30 Antioxidants can inhibit or prevent the automatic oxidation of lipids and oils, thus blocking oxidation reactions at the stage of initiation or propagation and balance of antioxidants and free radicals.15 The inhibition of free radical activity is performed by amino acid donors which act as antioxidants.36 Amino acid composition in proteins is limited by its tertiary structure because many of them have antioxidant potentials buried in the protein nucleus. Enzymatic hydrolysis proteins have been reported to increase antioxidant peptide activity and 20 amino acids found in proteins interacting with free radicals.37 The ability of hydrolysis proteins to inhibit free radicals suggest its potency to be developed as an antioxidant resource to overcome diabetes. Diabetes can be associated with hyperglycemia, which causes glucose autooxidation, protein glycation, and the metabolic activation pathway of polyol, which can accelerate the formation of free radicals.15 The formation of free radicals leads to diabetic complications, hence antioxidants are necessary.
Many approaches that have been developed to resolve diabetes are carried out, including enzymatic inhibition of α-amylase and α-glucosidase by certain proteins to reduce the level of glucose in the blood.38,39,40 Protein peptides can donate their electron to bind to α-amylase and α-glucosidase, which can then inhibit the action of α-amylase and α-glucosidase on metabolic pathways.41,42 Acarbose is an α-glucosidase inhibitor that is commonly used to control postprandial blood glucose. It plays a role as a competitor and reversible inhibitor of small intestinal brush border glucosidase. It blocks the degradation of starch and sucrose, then delays the absorption of glucose and fructose in the alimentary tract.43
This study shows that the protein from melinjo seeds has the potential to inhibit α-amylase and metabolic α-glucosidase with a similar mechanism as the actin of acarbose. Hydrolysis proteins have a very low IC50 value compared to nonhydrolysate proteins. This hydrolysis changed protein into a simpler form in which its molecular size was more flexible and was able to interact with the active site of the enzyme showed by inhibition ability. According to Motyan et al.,44 inhibition of α-amylase and α-glucosidase was related to amino acids containing cationic and branching chain residues such as Tyr, Phe, Trp, and Lys, which increased due to hydrolysis using alcalase.
Conclusion
This study demonstrated that antioxidant and antidiabetic activities could be generated from melinjo seed protein using enzymatic hydrolysis. The green melinjo seed is a good source of protein, antioxidant and antidiabetic peptide. The melinjo seed protein hydrolysate prepared with alcalase has potential potency as nutraceutical source for human health.
Conflict of Interest
The authors declare no conflict of interest
Acknowledgments
This research was supported by the Ministry of Research, Technology and Higher Education, Indonesia. SKIM IPTEK/Stranas and Hibah kompetensi.
References
- Mannner I., Elevitch C. R. Gnetum gnemon (Gnetum). In Craig R. Elevitch.Species Profiles for Pacific Island Agroforestry – Ecological, Economic, and Cultural Renewal. Hawai: Permanent Agriculture Resources (PAR); 2006: 285-392. http://traditionaltree.org (accessed February 06, 2019).
- Bhat R., Yahya N. Evaluating Belinjau (Gnetum gnemon) Seed Flour Quality as a Base for Development of Novel Food Products and Food Formulations. Food Chem. 2014; 156: 42-49.
CrossRef - Kato E., Tokunaga Y., and Sakan F. Stilbenoids isolated from the seeds of melinjo (Gnetum gnemon) and their biological activity. Journal of Agricultural and Food Chemistry. 2009; 57; 2544–2549.
CrossRef - Siswoyo T.A., Ardyati T., and Hosokawa K. Fermentation-induced changes in antioxidant activities and oxidative DNA damage protection of melinjo (Gnetum gnemon) flour. J. Food. Biochem. 2017; 1–8.
CrossRef - Khan M. R., Omoloso A. D., Kihara M. Antibacterial activity of Artocarpus heterophyllus. Fitoterapia. 2003; 74(5): 501–
CrossRef - Rauha J. P., Remes S, Heinonen M., Hopia A., Kähkönen M., Kujala T., Pihlaja K., Vuorela H., Vuorela P. Antimicrobial effects of finish plant extracts containing flavonoids and other phenolic compounds. Int. J. Food Microbiol.2000; 56(1): 3–
CrossRef - Uboh F. E, Okon I. E, Ekong M. Effect of aqueous extract of Psidium guajavaleaves on liver, enzymes, histological integrity and hematological indices in rats. Gastroenterol Res. 2010; 3(1): 32–38.
CrossRef - Kato H., Samizo M., Kawabata R., Takano F., Ohta T. Stilbenoids from the melinjo ( L.) fruit modulate cytokine production in murine peyer’s patch cells ex vivo. Planta Med. 2011; 77(10):1027–
CrossRef - Tatefuji,Miyako Y., Shinobu F., Ken H. Safety assessment of melinjo (Gnetum gnemon L.) seed extract: Acute and subchronic toxicity studies. Food and Chemical Toxicology. 2014; 67: 230–235.
CrossRef - Siswoyo T. A., Eka M., Lee K. O., Hosokawa K. Isolation and Characterization of Antioxidant Protein Fractions from Melinjo (Gnetum gnemon) Seed. Agric. Food Chem. 2011; 59(10): 5648–5656.
CrossRef - Mohadjreni M. Antioxidant Activity and Total Phenolic Content of Nerium oleander Grown in North of Iran. Iranian Journal of Pharmaceutical Research. 2012; 11(4): 1121–1126.
- Burits M., and Bucar F. Antioxidant Activity of Nigella sativa Essential Oil. Res. 2000; 14: 323–328.
CrossRef - Sharififara, Gholamreza D. N., and Mansour M. K. Major flavonoids with antioxidant activity from Teucrium polium L. Food Chemistry; 2009; 112: 885–888.
CrossRef - Piemonte L.Type 2 diabetes. International Diabetes federation. https://idf.org/52-about-diabetes.html. (Accessed March 25, 2019).
- Droge W. Free Radicals In the Physiological Control of Cell Function. Rev.2002; 82(1): 47–95.
CrossRef - MaritimC., Sanders J. R.A., Watkins B. Diabetes, Oxidative Stress, and Antioxidants: A Review. J. Biochem. Mol. Toxicol. 2003; 17(1): 24–38.
CrossRef - Manaharan, Ling L.T., Appleton, D., Cheng, H.M., Masilamani T. and Palanisamy, U.D. Antioxidant and Antiglycemic Potential of Peltophorum pterocarpum plant parts. Food Chem. 2011; 129(4):1355–1361.
CrossRef - Shinde J., Taldone T., Barletta M., Kunaparuju N., Bo H., and Kumar S. Alpha glucosidase Inhibitory Activity of syggiumcumini (Linn) Skeels Seed Kernel In Vitro Goto-kakizaki (KG) R Carbohydrate research. 2008; 243: 1278–1281
CrossRef - Bradford M. M. A Rapid and Sensitive Method for the Quantitation of Microgram Quantities of Protein Utilizing the Principle of Protein-Dye Binding. Biochem. 1976; 72(1-2): 248–54.
CrossRef - Adler-Nissen J. Determination of the Degree of Hydrolysis of Food Protein Hydrolysates by Trinitrobenzenesulfonic Acid. J. Agric. Food Chem. 1979; 27(6): 1256-1262.
CrossRef - Laemmli U. K. Cleavage of Structural Proteins During the Assembly of the Head of Bacteriophage Nature. 1970; 277(5259) :680–685.
CrossRef - Re N. P., Proteggente A., Pannala A., Yang M., Rice-Evans. Antioxidant Activity Applying an Improved ABTS Radical CationDecolorization Assay. Free Rad. Bio.Med., 1999; 26(9-10): 1231–1237.
CrossRef - Halliwell B., Gutteridge J. M. C., Arouma I. The Deoxyribose Method: A Simple Test Tube Assay for the Determination of Rate Constants for Reactions of Hydroxyl Radicals. Anal. Biochem. 1987; 165(1): 215–219.
CrossRef - Tang X., He Z., Day Y., Xiong Y. L., Xie M., Chen J. Peptide Fractionation and Free Radical Scavenging Activity of Zein Hydrolysate. Agri. Food Chem. 2010; 58(1): 587–593.
CrossRef - Hashim A., Khan M. S., Khan M. S., Baig M. H., Ahmad S. Antioxidant and a-Amylase Inhibitory Property of Phyllanthus Virgatus: An In Vitro and Molecular Interaction Study. Biomed Res. Inter., 2013; 1-12.
CrossRef - Miyazawa M., Yagi N., Taguchi K. Inhibitory Compounds of α-glucosidase Activity from Arctium lappa J. Oleo Sci. 2005; 54(11): 589–594.
CrossRef - Lobo V., Patil A., Phatak A., Chandra N. Free Radicals, Antioxidants,and Functional Foods: Impact on Human Health. Rev. 2010; 4(8): 118–126.
CrossRef - Fatmah A.M., Siti B.B., Zariyantey A.H., Nasar A., Jamaludin The Role of Oxidative Stress and Antioxidants In Diabetic Complications. SQU Med J. 2012; 12(1): 5-18.
- Smith D. M., Nielsen S. S. Protein Separation and Characterization Procedures. In Heldman D. R. (ed). Food Analysis. Springer US. 2010: 261-281.
CrossRef - Valco M., Leibfritz D., Moncol J., Cronin M. T. D., Mazur M., Telser J. Free Radicals and Antioxidants in Normal Physiological Functions and Human Disease. J. Biochem.2007; 39(1): 44–84.
CrossRef - Siswoyo T.A, Matra F.N, Safiera A.A, Supriyadi A. Synthesis of Antioxidant Peptides from Melinjo (Gnetum Gnemon) Seed Protein Isolated Using Sol-Gel Immobilized Inter. J. Adv. Sci. Eng. Inf. Tech. 2017; 7(4): 2088–5334
CrossRef - Jamdar N., Rajalakshmi, V., Pednekar M. D., Juan F., Yardi V., and Sharma A. Influence of Degree of Hydrolysis on Functional Properties, Antioxidant Activity and ACE Inhibitory Activity of Peanut Protein Hydrolysate. Food Chemistry. 2010; 121(1): 178–184.
CrossRef - Karamać M., Kulczyk A., and Sulewska K. Antioxidant Activity of Hydrolysates Prepared from Flaxseed Cake Proteins Using Pancreatin. Polish Journal of Food and Nutrition Sciences. 2014; 64(4): 227–
CrossRef - Pazinatto C., Malta L. G., Pastore G. M., and Maria F. Antioxidant Capacity of Amaranth Products: Effects of Thermal and Enzymatic Treatments. Food Science and Technology. 2013; 33(3): 485–
CrossRef - Chen H. M., Muramoto K., Yamauchi F., Fujimoto K., and Nokihara K. Antioxidative Properties of Histidine-Containing Peptides Designed from Peptide Fragments Found In The Digest of A Soybean Protein. Journal of Agricultural and Food Chemistry. 1998; 46(1): 49–
CrossRef - Osman A., Makris D.P., Kefalas P. Investigation on Biocatalytic Properties of a Peroxidase-Active Homogenate from Onion Solid Wastes: An Insight into Quercetin Oxidation Mechanism. Process Biochem. 2008; 43(8): 861- 867.
CrossRef - Elias R. J., Kellerby S. S., and Decker E. Antioxidant Activity of Proteins and Peptides. Critical Reviews in Food Science and Nutrition.2008; 48: 430–441.
CrossRef - Sarah C. F., Yeyi G., Joshua D. L. Inhibition of Starch Digestion by the Green Tea Polyphenol, (−)-epigallocatechin-3-gallate. Nutr. Food Res. 2012; 56(11): 1647–1654.
CrossRef - Rahbani-Nobar M.E., Rahimi-Pour A., Rahbani-Nobar M.Total Antioxidant Capacity, Superoxide Dismutase,and Glutathione Peroxidase In Diabetic Patients. J. Islamic World Acad. Science. 1999; 12(4): 109-114.
- Kwon Y. I., Apostolidis E., Kim Y. C. Shetty K. Health Benefits of Traditional Corn, Beans, and Pumpkin: In Vitro Studies for Hyperglycemia and Hypertension Management. Med. Food. 2007; 10(2) :266–275.
CrossRef - Afifah B. S. N. H., Chee–Yuen, G. Antioxidative and Amylase Inhibitor Peptides from Basil Seeds. J. Peptide Res. Ther. 2015; 22(1): 3–10.
CrossRef - Alam, Gourinath S., Dey, S., Srinivasan A., Singh, T.P. Substrate-Inhibitor Interactions in the Kinetics of alpha-Amylase Inhibition by Ragi alpha-amylase/Trypsin Inhibitor (RATI) and Its Various N-Terminal Fragments. Biochem. 2001; 40(14): 4229–4233.
CrossRef - He K., Jun-C. S., Xiao M. M. Safety and Efficacy of Acarbose In the Treatment of Diabetes In Chinese Patients. Therapeutics and Clinical Risk Management. 2014; 10: 505–
CrossRef - Motyan, Toth F., and Tozser J. Research applications of proteolytic enzymes in molecular biology. Biomolecules. 2013; 3: 923–942.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






