Introduction
Artemisia absinthium L., commonly known as wormwood, is a yellow-flowering perennial plant that growing in various parts of Europe and Siberia, and it is used for its antiparasitic and digestive properties for the treatments of anorexia and indigestion. The aerial parts of the plant are used in many herbal preparations, dietary supplements, and alcoholic beverages (e.g. absinthe)1. Wormwood infusions are being used as a medicinal remedy, due to the wide range of its therapeutic activities2.
Salvia sp. (sage) is an aromatic small perennial shrub native to the Mediterranean region. Sage is cultivated for culinary and medicinal purposes and it is mainly used as infusion. Also, it has been used as a preservative or flavoring agent in food products and as an aromatic agent in perfumes and cosmetics3. The significant amount of terpenoids contained in sage teas, are contributing to their antioxidant, hepatotoxic, antimutagenic and antidiabetic effects4.
Thujone is a natural monoterpene ketone present in variable amounts in a large number of plants5, such as sage (Salvia officinalis), clary (Salvia sclarea), tansy (Tanacetum vulgare), wormwood (Artemisia artemisia) and white cedar (Thuja occidentalis L.). As so, it is included as a significant component in many dietary botanical supplements and herbal medicinal products6. Thujone occurs naturally in two diastereomeric forms: (-)-α-thujone and (+)-β-thujone (Figure 1). Concerning health effects parent compound is potentially toxic therefore, the presence of α– or β-thujone in food and beverages has been regulated by law in several countries7. Additionally, clinical tests to experimental animals has proved that α-thujone was more toxic than β-thujone or a mixture of isomers8.
The compound thujone is the psychotropic component of absinthe, wormwood oil and some other herbal medicines9, as sage4. α-Thujone reduces activity of 5-ΗΤ3 receptor which explains the psychotropic action9. Thujone (α– and β-) has neurotoxic action5, causing tonic-clonic convulsions due to inhibitory action on the GABAA receptor10. Finally, both isomers have shown hepatotoxicity11 and porphyrogenic activity12 .
The restrictions applied to thujone levels in food products are limited compared to the same levels referred in medicines. A. absinthium L. and especially S. officinalis L. are used for their therapeutic properties as infusions, decoctions or extracts (e.g., sage tea)13. According to European Medicines Agency (EMA), during the assessments of Artemisia absinthium L.14 and Salvia officinalis L.15 it became apparent that the risk assessment of thujone, which is a major component in both herbal preparations, poses considerable uncertainties and difficulties. A case-by-case assessment led to a difference in the maximum limit of daily intakes of thujone, which in the case of wormwood was set at 3 mg/day/person and in the case of sage leaf preparations at 5 mg/day/person, both for a maximum duration of 2 weeks. Furthermore, EMA/Committee on Herbal Medicinal Products (HMPC)16 has concluded that the benefits of use sage essential oil do not outweigh its risks. Considering that thujone is a natural constituent of the essentials oils of a number of plants widely used, the HMPC (2010) decided to prepare a public statement on the use of herbal preparations containing thujone17.
Microtox® test provides a rapid method of determining the acute toxicity of aqueous compounds by measuring decreases in light output from the luminescent bacterium Vibrio fischeri. Light emission is directly proportional to the metabolic activity of the bacterial population and any inhibition of enzymatic activity causes a corresponding decrease in luminescence18. Recently, Microtox® analyzer has been used as evaluation tool19 for several extracts of dietary plants20.
Due to the rising trend on consuming herbal infusions on a daily basis for the treatment and/or prevention of minor ailments it is very important tο determine their possible toxicity levels for humans.
Therefore, present work aimed at the determination of the quantities of α– and β-thujone in wormwood and sage infusions and the investigation of a possible correlation between thujone content and toxicity levels in both infusions.
Materials and Methods
Plant material
Dried plant material of wormwood and sage were obtained from several areas around Greece by local farmers as seen on Table 1. In detail, wormwood the plant material was purchased from the Island of Crete (Eastern Mediterranean, W1), Kozani (Western Macedonia, W3) and Agrinio (Western Greece, W4) along to a sample bought from the flea market (W2). Sage was supplied from Agrinio (Western Greece, SP), Aetolia-Acarnania (Western Greece, SF1), Kozani (Western Macedonia, SF2), Thessaloniki (Central Macedonia, SF3) and Ileia (Western Greece, ST). The different species of sage are native species of each region. In case of sage, they were used only the plant leaves while from wormwood were used both leaves and flowers. In all cases the plant material was grinded to powder before treatment.
Reagents and materials
The reagents used were: diethyl ether (DEE) stabilized with butylated hydroxytoluene (BHT) (purity 99.84%, Lach-Ner, s.r.o, HU), anhydrous magnesium sulfate (purity 97%, Acros Organics, NJ, USA), (-)-α-thujone (purity ≥ 96.0%; Sigma–Aldrich Co., St. Louis, MO, USA), cyclohexanone (purity ≥ 99.0% , Merck KGaA, Germany).
Toxicity tests were performed using Microtox® Analyzer Model 50021 equipped with 30 temperature controlled wells regulated at 15 o C in addition with a reactivation well regulated at 5 o C used to store the bacteria suspension before dilution. Acute toxicity reagents such as Diluent (sterile 2% sodium chloride), OAS (osmotic adjusting solution 22% sodium chloride), reconstitution solution and test organisms Vibrio fischeri, formerly known as Photobacterium phosphoreum, (MODERN WATER/Microtox®), NRRL, No B-11177 were supplied by Strategic Diagnostics INC.
Preparation of herbal infusions
Fifty (50) g of each plant material were powdered in an electrical blender. Infusions were prepared by stepping 2 g of plant material in 200 mL of boiled water for 10 minutes, followed by filtration. This procedure simulates the conventional procedure of tea preparation, as 2 g is the average mass of a commercial tea bag, while the volume of 200 mL is corresponding to a cup of tea. 50 mL from each infusion were stored in labeled sterile screw capped bottles at 5oC until test by the Microtox® Analyzer. The rest (150 mL) of infusions was then further extracted by diethyl ether (3 x 50 mL) in order to isolate the no polar compounds. The organic layers were merged to one, dried over anhydrous magnesium sulfate and filtered. Furthermore, the organic solvent removed in vacuo by a rotary flash evaporator. The distillation was stopped when the volume of solvents was reduced to ~5 mL and the samples were then stored in labeled sterile screw capped bottles at -22oC until use. The all procedure was done in triplicate for every plant sample. Before analysis, by gas chromatography-mass spectrometry (GC-MS), each stored sample was further concentrated until the volume of 1 mL by flushing through nitrogen. Finally, a volume (50 μL) of a cyclohexanone solution (10 μL/mL diethyl ether) added, as an internal standard, in each sample.
Gas Chromatography-Mass Spectrometry (GC-MS)
The volatile compounds΄s fraction from the herbal infusions was determined using gas chromatography (Thermo Scientific, TRACE GC ULTRA) coupled with mass spectrometry (DSQ II), operating in the electron impact mode (70 eV). Compounds were separated with column Thermo-5MS (30 m length, 0.25 mm ID, 0.25 μm film thickness). The carrier gas was helium with a constant flow of 1 mL/min. Τhe inlet temperature of the sample was 220 o C and the transfer line to MSD was 250 o C. The temperature program was 60 o C to 250 o C at 3 o C /min with duration 63.33 min for each sample analysis. The volume of the samples manually injected was 1 μL (in splitless mode).
Chromatographic peaks were identified based on retention times, mass spectra of authentic compounds when available, Adams (2007), Χ-Calibur library of mass spectra and published data.
Microtox ® acute toxicity assay
The Microtox® Toxicity Test evaluates the extent to which luminescence is inhibited in the marine bacterium Vibrio fischeri. Toxicity assessment of the plant extracts was performed according to a protocol known as Basic Test 81.9% 21 applied for aqueous samples, but with appropriate primary dilution of samples in order that the EC50 (Effective Concentration where bioluminescence inhibition is 50%) could be obtained without extrapolation of data. A dilution factor 2.0 was used. Dilutions and assays were performed in incubator wells at 15±0.5 ◦C.
Toxicity estimations were obtained after 15 min exposure and expressed as EC50 value and EC20 value (sample concentration that gives 50% and 20% luminescence inhibition respectively) were also calculated, as described below.
The data were processed using the Microtox® Omni Software, according to the Basic Test Protocol (81.9%). Omni software provided the linear correlation of Gamma (G) values with sample concentration. The relationship between the concentration of a toxic material and the response of a susceptible organism, when the response is measured in terms of Gamma (G) values, can be described by a linear equation for the prediction of concentration from Gamma values:
log C = b log G + log a
This equation describes a line with a slope of b and an intercept of log a, in which C represents concentration and G the corresponding Gamma value. EC20 concentrations were calculated by placing G = 0.250 (effect 20%) in correlation equations. As, according to the ISO 11348 (1998) guidelines, a toxic sample shows an effect higher than 20%, the above values are the maximum concentrations for which a sample can be considered as non-toxic 19.
Table 1: Plant material from different origin of Greece
|
Sample code |
Plant material |
Origin |
|
W1 |
Artemisia absinthium |
Creta |
|
W2 |
Wormwood |
Flea Market |
|
W3 |
Artemisia absinthium |
Kozani |
|
W4 |
Artemisia absinthium |
Agrinio |
|
SP |
Salvia pomifera |
Agrinio |
|
SF1 |
Salvia officinalis |
Aetolia-Acarnania |
|
SF2 |
Salvia officinalis |
Kozani |
|
SF3 |
Salvia officinalis |
Thessaloniki |
|
ST |
Salvia triloba |
Ileia |
Quantitative determination of α– and β-thujone in herbal infusions
Τhujone (α– and β-) in herbal infusions of wormwood and sage were quantitatively (mg/mL) determined by the method of internal standard and calibration curve (the correlation coefficient was r2 = 0.9996). Cyclohexanone which was used as internal standard was not co-eluted with the compounds α– and β-thujone (Figure 1 and 2) during GC-MS analysis.
Results and Discussion
Volatile components found in herbal infusions
The volatiles components of wormwood and sage infusions that were determined by GC-MS analysis of their organic extracts were expressed as relative percentage concentration.
In Table 2, are presented only the major (over 10%) volatile compounds identified in infusions of the two herbs, along with their Retention Time (RT). Interestingly, of the data analysis of wormwood samples did not came out qualitative similarities concerning volatiles of their infusions. Trans-sabinyl acetate was the only component that found in common and only in samples W3 and W4. In sample W1, β-thujone was the only compound identified at a percentage of 53.28% while rest of the components were less than 10%. Similarly, in sample W2 o-xylene (95.08%) was found as the predominant ingredient. Wormwood W3 was the only sample in which three main compounds were identified as follow: Z-epoxy-ocimene (41.65%), nootkatin (40.26%) and trans-sabinyl acetate (17.81%). In sample W4, m-xylene (47.42%) and trans-sabinyl acetate (26.11%) were observed as the major ingredients.
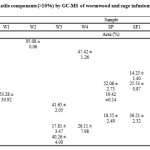 |
Table 2: Quantification of major volatile components (>10%) by GC-MS of wormwood and sage infusions of different geographical origin Click here to View table |
To the best of our knowledge there are no literature references concerning volatile composition of wormwood infusion. Literature references on wormwood extract΄s compounds are focused mostly on organic extracts and the essential oils composition. β-Thujone, as main constituent (>10%) was indentified also by Abad et al.1 and Ben Jemma et al.22 while Orav et al.23 estimated at a percentage of 8.73% and Judþentienë et al.24 did not detected it even in traces. Orav et al.23 were the only who also found in essential oils Z-epoxy-ocimene similar to our results (sample W3). trans-Sabinyl acetate is a compound found by all the previous studies1, 22, 23, 24 regarding wormwood.
As shown in Table 2, camphor was the only in common component in all sage infusion’s volatiles reaching the highest percentage in sample SF3 (48.67%). α-Thujone was found in samples SP, SF1, SF2 and SF4 at percentages that ranged from 11.27% to 52.06%. 1,8-Cineole identified in samples SF1,SF2,SF3 and ST ranged from 10.20% (in SF2) up to 45.05% (in ST). Finally, β-thujone (19.42%) was found only in samples SP while p-xylene (14.37%) only in SF2.
The identified compounds in samples SF1,SF2 and SF3 are alike to those reported by Arceusz et al.4, where 1,8-cineole, α-, β-thujone and camphor were the major compounds in sage infusions. In the case of sample ST, the same compounds have been found in ethanol extracts but at different percentages25. Compared to the essential oil profile by Karousou et al.26 were also identified α-and β-thujone as the major components similar to our results for sample SP.
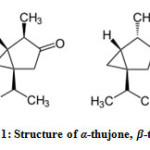 |
Figure 1: Structure of α–thujone, β-thujone |
Content of α– and β-thujone in herbal infusions
In addition to the estimation of the relative percentage concentration of the volatile constituents in the infusions, it was concerned to further quantify α– and β-thujone. Parent investigation is of critical importance due to existed limits in the consumption of food and drinks that contain sage or wormwood in terms of EMA14, 15 .
The results of quantitative determination of α– and β-thujone of wormwood and sage infusions from different parts of Greece are shown in the Figure 3. Concentrations were calculated using the calibration curve and the areas of οf α– and β-thujone from GC-MS plots and expressed as mg/mL of herbal infusion. Satisfactory precision was achieved by the use of cyclohexanone (Figure 1) as an internal standard. A typical GC-MS chromatogram of the above compounds is shown in Figure 2.
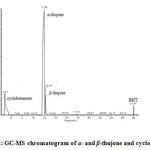 |
Figure 2: GC-MS chromatogram of α– and β-thujone and cyclohexanone |
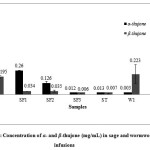 |
Figure 3: Concentration of α– and β-thujone (mg/mL) in sage and wormwood infusions
|
Wormwood infusions showed much lower levels of α– and β-thujone compared to sage. In the case of wormwood, in samples W1 and W2, β-thujone was predominant among two diastereomeric forms at concentration of 0.223 mg/mL and 0.017 mg/mL respectively. In samples W3 and W4 neither of compounds α– and β-thujone were detected.
In contrast to wormwood, α-thujone was the predominant isomer in sage infusions. According to the results presented in Figure 3, quantities of the mixture of thujones varied between the Salvia species. Regarding α-thujone, sample SP presented the highest concentration (0.614 mg/mL) followed by SF1 (0.26 mg/mL), SF2 (0.126 mg/mL), ST (0.012 mg/mL) and SF3 (0.013 mg/mL). Concentration levels of β-thujone estimated from 0.006 mg/mL to 0.195 mg/mL.
Table 3 presents the thujone quantities, as sum of α-and β– isomers in the tested samples expressed as mg/200 mL, where 200 mL is an average cup volume.
According to the maximum daily intake of α– and β-thujone in wormwood (3 mg/day/person)14, sample W2 does not exceed the established limit in contrast to W1 which is presented in high level (45.24 mg/200 mL). The high content of thujone in sample W1 is probably due to the origin of plant (soil and climatic conditions).
In the case of sage where the maximum daily intake is 5 mg/day/person15, the calculated thujone in samples ST and SF3 was found within the limits in contrast, to the quantities of thujone in samples SF2, SF1 and SP. Particularly, in the case of sample SP our data revealed 30 fold higher quantity of thujone than acceptable value, probably due to the differentiation of the species.
Consequently, in light of our results and before any generalization, there is a need to further investigate a greater number of sage and wormwood samples regarding the quantification of thujone in their infusions.
Table 3 : Content (mg/200 mL) of thujone (sum α– and β-) in wormwood and sage infusions
|
Sample |
Content (mg/200 mL) of thujone (sum α– and β–) in wormwood and sage infusions |
|
W1 |
45.24 |
|
W2 |
3.50 (only β-form) |
|
SP |
161.94 |
|
SF1 |
58.81 |
|
SF2 |
32.18 |
|
SF3 |
3.66 |
|
ST |
4.03 |
Table 4: Toxicity and concentration that causes 20% luminescence inhibition
|
Sample |
mass (mg) of dry plant material /mL of infusion |
Toxicity as EC20, after 15 min (%) |
Concentration that causes 20% luminescence inhibition after 15 min (mg of dry plant material/mL of infusion) |
|
W1 |
0.007 |
9.92 |
0.79 |
|
W2 |
0.007 |
5.34 |
0.43 |
|
W3 |
0.007 |
9.77 |
0.68 |
|
W4 |
0.007 |
12.32 |
0.86 |
|
SP |
0.005 |
5.19 |
0.26 |
|
SF1 |
0.005 |
1.63 |
0.08 |
|
SF2 |
0.005 |
5.76 |
0.29 |
|
SF3 |
0.005 |
7.06 |
0.35 |
|
ST |
0.005 |
4.79 |
0.24 |
Toxicity
In addition to the quantification of thujone (α– and β-) levels in infusions, current research was proceed further to the estimation of their relative toxicity on the marine bacterium Vibrio fischeri using Microtox® analyzer by using specific protocols that have been previously done for other plant infusions and extracts by other researches 19, 20. Toxicity evaluation of the infusions was performed by applying the Basic test protocol. This protocol furnishes a curve dose/effect that guarantees the repeatability and the reliability of the test. These features were demonstrated by statistic analysis of the obtained data. The operating software calculates the toxicity in terms of EC50 or as percentage of effect at each concentration value. The final values are calculated for all the assayed samples as an average of the values from triplicate assays.
According to the guidelines ISO (ISO 11348, 1998)27, a toxic sample shows an effect percentage greater than 20%. For this reason toxicity was calculated and expressed as EC20 values.
The results on toxicity that are presented in Table 4, indicate that between wormwood samples, the most toxic is W2, and the less toxic is W4, as EC values decreased by the increase of toxicity. Comparing the α– and β-thujone levels and resulted EC values of samples tested, no correlation was found (r2: 0.18 and 0.14 respectively) indicating that toxicity is maybe attributed to other constituents of the infusions included the water soluble substances as also referred by Skotti et al.19.
The toxicity values of sage infusions, as presented in Τable 4, varied also, but with no great differences between the samples tested, except SF1 which gave the greatest EC20 value (1.63%). In the case of sage, as also in wormwood, no correlation was observed between thujone (α– and β-) concentrations and toxicity values (r2: 0.14 and 0.03 respectively), thus toxicity of infusions is probably attributed to the other infusion constituents which are mainly water soluble.
In conclusion, in all sage infusions examined thujone was determinated, as main ingredient in form of α- in contrast to wormwood infusions where it was found only in some samples in the form of β-thujone. Most of wormwood and sage infusions exceeded thujone limits set by EMA. Regarding toxicity against the bacterium Vibrio fischeri, sage infusions showed higher levels compared to wormwood. Furthermore, no correlation was found between thujone content and toxicity levels of both infusions, indicating that toxicity might be attributed to other constituents of the infusions. For the reasons described above and taking into account the relationship between benefits and risks, wormwood and sage infusions needs further analysis and investigation.
References
- Abad MJ, Bedoya LM, Apaza L, Bermejo P. The Artemisia L. Genus: A Review of Bioactive Essential Oils. 2012:2542-2566. doi:10.3390/molecules17032542.
CrossRef - Gonzalez-Coloma A, Bailen M, Diaz CE, et al. Major components of Spanish cultivated Artemisia absinthium populations: Antifeedant, antiparasitic, and antioxidant effects. Ind Crops Prod. 2012;37(1):401-407. doi:10.1016/j.indcrop.2011.12.025.
CrossRef - Ben Farhat M, Jordán MJ, Chaouch-Hamada R, Landoulsi A, Sotomayor JA. Phenophase effects on sage (Salvia officinalis L.) yield and composition of essential oil. J Appl Res Med Aromat Plants. 2016;3(3):1-7. doi:10.1016/j.jarmap.2016.02.001.
CrossRef - Arceusz A, Occhipinti A, Capuzzo A, Maffei ME. Comparison of different extraction methods for the determination of ␣ – and  -thujone in sage ( Salvia officinalis L .) herbal tea. 2013:3130-3134. doi:10.1002/jssc.201300206.
CrossRef - Pelkonen O, Abass K, Wiesner J. Thujone and thujone-containing herbal medicinal and botanical products: toxicological assessment. Regul Toxicol Pharmacol. 2013;65(1):100-107. doi:10.1016/j.yrtph.2012.11.002.
CrossRef - Committee S. Opinion of the Scientific Committee on Food on Thujone. 2003;(December 2002).Dolan LC, Matulka R a, Burdock G a. Naturally occurring food toxins. Toxins (Basel). 2010;2(9):2289-2332. doi:10.3390/toxins2092289.
CrossRef - Waidyanatha S, Johnson JD, Hong SP, et al. Toxicokinetics of α-thujone following intravenous and gavage administration of α-thujone or α- and β-thujone mixture in male and female F344/N rats and B6C3F1 mice. Toxicol Appl Pharmacol. 2013;271(2):216-228. doi:10.1016/j.taap.2013.05.001.
CrossRef - Deiml T, Haseneder R, Zieglgänsberger W, et al. α-Thujone reduces 5-HT3 receptor activity by an effect on the agonist-induced desensitization. Neuropharmacology. 2004;46(2):192-201. doi:10.1016/j.neuropharm.2003.09.022.
CrossRef - Rivera EM, Cid MP, Zunino P, Baiardi G, Salvatierra N a. Central α- and β-thujone: similar anxiogenic-like effects and differential modulation on GABAA receptors in neonatal chicks. Brain Res. 2014;1555:28-35. doi:10.1016/j.brainres.2014.01.039.
CrossRef - Abass K, Reponen P, Mattila S, Pelkonen O. Metabolism of α-thujone in human hepatic preparations in vitro. Xenobiotica. 2011;41(2):101-111. doi:10.3109/00498254.2010.528066.
CrossRef - Bonkovsky HL, Cable EE, Cable JW, et al. Porphyrogenic properties of the terpenes camphor, pinene, and thujone. (with a note on historic implications for absinthe and the illness of vincent van gogh). Biochem Pharmacol. 1992;43(11):2359-2368. doi:10.1016/0006-2952(92)90314-9.
CrossRef - Lachenmeier DW, Uebelacker M. Risk assessment of thujone in foods and medicines containing sage and wormwood–evidence for a need of regulatory changes? Regul Toxicol Pharmacol. 2010;58(3):437-443. doi:10.1016/j.yrtph.2010.08.012.
CrossRef - Nl A. COMMUNITY HERBAL MONOGRAPH ON ARTEMISIA ABSINTHIUM L., HERBA. 2009;(July).
- Wharf C. COMMUNITY HERBAL MONOGRAPH ON SALVIA OFFICINALIS L., FOLIUM. 2010;(November 2009).
- Wharf C, Kingdom U. Public statement on the use of herbal medicinal products containing thujone Public statement on the use of herbal medicinal products containing thujone Table of contents. 2011;44(January).
- Discussion F, Party W, Products HM, et al. Public statement on the use of herbal medicinal products containing thujone. 2012;44(May):1-9.
- Montalbán MG, Hidalgo JM, Collado-González M, Díaz Baños FG, Víllora G. Assessing chemical toxicity of ionic liquids on Vibrio fischeri: Correlation with structure and composition. Chemosphere. 2016;155:405-414. doi:10.1016/j.chemosphere.2016.04.042.
CrossRef - Skotti E, Anastasaki E, Kanellou G, Polissiou M, Tarantilis PA. Total phenolic content, antioxidant activity and toxicity of aqueous extracts from selected Greek medicinal and aromatic plants. Ind Crops Prod. 2014;53:46-54. doi:10.1016/j.indcrop.2013.12.013.
CrossRef - Conforti F, Ioele G, Statti GA, Marrelli M, Ragno G, Menichini F. Antiproliferative activity against human tumor cell lines and toxicity test on Mediterranean dietary plants. Food Chem Toxicol. 2008;46(10):3325-3332. doi:10.1016/j.fct.2008.08.004.
CrossRef - Microtoxomni AE. User Manual.
- Ben Jemaa JM. Essential Oil as a Source of Bioactive Constituents for the Control of Insect Pests of Economic Importance in Tunisia. Med Aromat Plants. 2014;3(2). doi:10.4172/2167-0412.1000158.
CrossRef - Orav A, Raal A, Arak E, Müürisepp M, Kailas T. Composition of the essential oil of Artemisia absinthium L . of different geographical origin. 2006:155-165.
- Judþentienë A. Chemical composition of essential oils of Artemisia absinthium L . ( wormwood ) growing wild in Vilnius. 2004;(4):64-68.
- Al-Kalaldeh JZ, Abu-Dahab R, Afifi FU. Volatile oil composition and antiproliferative activity of Laurus nobilis, Origanum syriacum, Origanum vulgare, and Salvia triloba against human breast adenocarcinoma cells. Nutr Res. 2010;30(4):271-278. doi:10.1016/j.nutres.2010.04.001.
CrossRef - Karousou R, Vokou D, Kokkini S. Distribution and essential oils of Salvia pomifera subsp. pomifera (Labiatae) on the island of Crete (S Greece). Biochem Syst Ecol. 1998;26(8):889-897. doi:10.1016/S0305-1978(98)00033-7.
CrossRef - En iso 11348.03 2. 2011.

This work is licensed under a Creative Commons Attribution 4.0 International License.





