Introduction
Under heavy anthropogenic loads, aggravated stress situations and other unfavorable impacts on human body, dietary (preventive and therapeutic) as well as functional foods are extremely desired. 1 Both functional foods and dietary supplements relate to nutraceuticals or food-related products that have purported health or medical benefits.1-3 The objectives of the approved “Strategy for Improving the Quality of the Food Products in the Russian Federation until 2030” are: promoting the healthy nutrition principles, production of functional specialized as well as production and use of functional food ingredients in products.4
Manufacturing of fermented milk products has always been one of the key priorities of the dairy industry.4 At the same time, it is possible to develop functional properties of the product by introducing probiotic microorganisms. Another direction in functional food manufacture development is extensive use of nutrients, especially those of domestic origin. Undoubtedly, the examples of such nutrients include Whey protein hydrolysate (WPH) with high degree of hydrolysis (DH of at least 60%), which is a mixture of amino acids, peptides, vitamins, minerals and lactose (detailed WPH composition see elsewhere6-10). Skimmed milk and buttermilk are the products obtained in a course of milk material processing having a complex of biologically active substances and low energy value.11-13
The use of WPH in food technology allows manufacturing products with high nutritional density. As far as the manufacture of fermented milk products is concerned, biologically active compounds, the concentrations of which contained in WPH may serve as an additional source for feeding starter culture microorganisms. This is the basis for using of various proteins hydrolysate as a part of culture media for the cultivation and isolation of most microorganisms in their pure form.6-8 For example, animal hydrolysated protein are known as growth promoters for the enhanced growth of Bifidobacterium.8-10,14-15 The present research positions WPH, first of all, as a source of animal protein, essential amino acids and means for increasing the biological value of the product, and as such, a growth stimulator for starter culture microorganisms.
Previously, experiments focused on determining the maximum amount of introduced WPH by the method of organoleptic evaluation. The results of tasting have shown that it is possible to add not more than 3% of WPH to obtain optimal organoleptic characteristics. The amino acid composition of the product was also analyzed and it has been concluded that the product may be considered as a functional food with respect to vaine, isoleucine, leucine, threonine and tryptophan subject to WPH content of 3 wt% since in this case the composition provides 15% of adequate daily intake of the amino acids.16 At the same time, there is no information regarding the use of such amount of WPH with high degree of hydrolysis in dairy products as a functional nutrient. Therefore, in order to obtain a product with acceptable consumer characteristics, the influence of WPH on the development of lactic acid microorganisms in the fermented milk products was studied. Since the use of WPH may increase the product cost, it is important to find ways for optimizing production parameters, for example, by reducing the introduced starter proportion or by shortening the technological process. As such, the influence of WPH on technological and biochemical particulars of the most important representatives of lactic acid microorganisms in various model systems is of both scientific and practical interest defining the overall relevance of this study.
The free amino acids, amines, and mineral salts contained in WPH may possibly make an impact on cultivated microorganisms. The introduction of WPH increases the total protein of the milk material to 5 wt% as well as the ratio between proteins and carbohydrates, which may affect the starter activity and consumer properties of the product, such as dry matter content, titrated and active acidity, flavour, odour, colour, and texture.
Since the microbiotica composition of starter cultures determines the consumer properties of fermented milk products, the processes of lactic acid fermentation of skimmed milk and buttermilk both containing from 1-3 wt% of WPH with monostrain and monospecies multistrain starter microorganisms were studied aiming at developing a probiotic product with typical consumer characteristics. The study of fermentation process was based on the analysis of the behavioural signs of microorganisms in the presence of WPH in accordance with regulatory documents. The present study did not focus on nucleotide species attribution and revealing the basis of metabolic processes that take place in starter culture cells under the influence of WPH.
The objective of this study was a comparative analysis of the fermentation processes in buttermilk and skimmed milk caused by starter microorganisms in the presence of WPH without identifying microorganisms and counting the number of cells.
Materials and Methods
The skimmed milk17, buttermilk18, and WPH (manufactured by All-Russian Research Institute of Butter and Cheese Making, RAS) were used in this study. The starter cultures available on Russian market, such as Streptococcus salivarius thermophilus, Lactococcus lactis subspecies, Lactobacillus, Lactobacillus plantarum, bifidobacterium concentrate (Bifidobacterium adolescentis, Bifidobacterium bifidum, Bifidobacterium breve, Bifidobacterium infantis, Bifidobacterium longum), and the kefir starter culture meeting Russian National Standards were used. Test samples contained from 1-3 wt% of WPH, whereas samples without WPH were used for control.
Organoleptic characteristics of both control and test samples were determined according to International Standards ISO 22935-2:2009, ISO 22935-2:2009, and ISO 5492:2008.19-21 Physical and chemical characteristics were determined according to the Standards ISO/TS 22113:201222 and GOST 32892-201423. The organoleptic evaluation of flavour, odour, colour and consistency of the fermented milk samples were carried out by experts using a discrete five-point scale. The experiments were carried out in series with repetitions.
The increase in the amount of synthesized lactate was calculated assuming that 1°T corresponds to 0.009 g of lactic acid. The degree of acid formation was calculated accounting for titrated acidity values before and after fermentation that indirectly characterizes the activity of lactic acid processes. This indicator shows the amount of lactic acid in 100 cm3 of the product, and it was calculated by the formula (1):
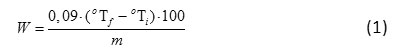
where W is the percentage of lactic acid in wt.% 0.009 is the lactate content within 100 cm3 of product corresponding to 1 °T; 0Тi is the initial titrated acidity in °T; 0Тf is the final titrated acidity in °Т; m is the weight of100 cm3 of the product.
The average rate of acid formation is the ratio of the formed concentration of lactic acid to the duration of the fermentation process, which was determined according to the formula (2):
![]()
Where ν is the acid formation rate in g/h; W is the percentage of lactic acid in wt%; τ is the fermentation period in h.
Results and Discussion
The introduction of WPH has been shown to modify several indicators of the culture medium, which is a standard for lactic acid microorganisms (Table 1). The absolute error range (± Δ) never exceeded the values of relevant standards.22-24
Table 1: Changes in Physical and Chemical Parameters of Milk Material Depending on the Weight Content of WPH.
| Raw Milk | Content of WPH, wt% | Acidity | Buffer capacity, cm3 NaOH* | Osmolality, mmol/kg Н2О | |
| Titrated, °Т | Active, рН | ||||
| Skimmed milk | 0 | 16.0±1.0 | 6.68±0.04 | 1.00±0.04 | 312.4±2.4 |
| 1 | 31.0±1.0 | 6.67±0.04 | 1.39±0.04 | 367.6±5.5 | |
| 2 | 46.0±1.0 | 6.61±0.04 | 1.64±0.04 | 421.5±6.0 | |
| 3 | 61.0±1.0 | 6.58±0.04 | 1.81±0.04 | 475.1±5.8 | |
| Buttermilk | 0 | 15.0±1.0 | 6.69±0.03 | 1.00±0.04 | 277.5±3.7 |
| 1 | 30.0±1.0 | 6.67±0.04 | 1.38±0.02 | 332.2±5.4 | |
| 2 | 45.0±1.0 | 6.65±0.04 | 1.63±0.04 | 386.2±5.7 | |
| 3 | 60.0±1.0 | 6.62±0.04 | 1.88±0.04 | 440.8±5.6 | |
| * equivalent molar concentration 0.1 mol/dm3 | |||||
Despite the increasing titrated acidity, the mixtures of milk material with WPH never demonstrated a pronounced sour flavour. The samples differed only in albumin flavour intensity. The titrated acidity of the milk material containing 1 wt% of WPH increased by 15±1.0°T, and the active acidity was not substantially changed. The media with different buffer systems are known to behave this way. This suggests that WPH containing polar amino acids, peptides, and protein molecules in its composition, promotes the rise of buffer properties in buttermilk and skimmed milk. According to the obtained results, it can be concluded that the addition of WPH in skimmed milk and buttermilk leded to an increase in buffer capacity and prevented microorganisms against the exposure to lactic acid. An increase in the osmotic concentration of milk material with the addition of WPH ensured the saturation of bacterial enzymes with accessible substrates. Thus, the studied milk materials intended for the cultivation of microorganisms were significantly different subject to the WPH content.
At present, standard specifications of bacterial starter cultures should be met for the production of milk and fermented milk products. 24 The document includes the requirements for starter cultures used in producing well-known fermented milk products, such as yogurt, kefir, fermented baked milk, yogurt, fermented milk product enriched with Bifidobacteria, acidophilus, etc.
The use of Lactococcus lactis monospecies starter culture may be reasonable in several aspects. First, lactococci strains have different degrees of acid formation. Lactococcus lactis subspecies lactis biovar diacetylactis have the weakest lactic acid synthesis activity. The same applies to Lactococcus lactis subspecies cremoris. However, Lactococcus lactis subspecies lactis are considered strong acidifiers. Secondly, when fermenting, the strain of Lactococcus lactis subspecies lactis biovar diacetylactis, forms aromatic compounds (diacetyl, acetoin), which may neutralize actual flavour and odour of WPH. Thirdly, free amino acids of WPH may be used by lactococci for growth. In addition, these cultures form a sufficiently thick clot with the typical flavour of fermented milk.
Lactobacilli were chosen due to the fact that they are auxotrophs; hence they are sensitive to culture medium composition. They need free amino acids for their growth. Moreover, they support mechanism for substrate phosphorylation of not only carbohydrates, but also amino acids.25 Thermophilic streptococcus microorganisms develop well in the milk material. They are distinguished by not very strong but active acid formation, and form thick smooth-textured clots in the milk material. Activity of Streptococcus salivarius thermophilus may be increased by adding free amino acids of valine, leucine, isoleucine, lysine, arginine, methionine, histidine, and proline. Under production conditions, Streptococcus salivarius thermophilesis combined with various lactic acid microorganisms, since thermophilic streptococcus, as the most active producer of β-galactosidase, activates the growth of a joint culture.
Lactobacillus plantarum and Bifidobacterium ssp. were used in the study. Lactobacillus plantarum is characterized by proteolytic activity and can grow in the medium with a high content of free amino acids. The probiotic properties of Lactobacillus plantarum arise from the ability to form a natural antibiotic lactoline and other metabolites.25
Bifidobacteria are probiotic cultures that dominate among beneficial microorganisms in the intestine and play an important role for human health for the whole life period. Bifidobacteria have antagonistic activity against a wide range of pathogenic and opportunistic microorganisms due to the ability of producing natural antibiotic-like substances, decreasing the pH level in the intestine, and forming organic acids and other biologically active compounds.26 By adhering to the intestinal mucosa, Bifidobacteria protect a living organism and reduce the effect of toxic substances. Bifidobacteria play the main role in maintaining and improving intestinal microbiocenosis.25-27 In the case of developing Bifidobacterium strains in a milk medium, it must be borne in mind that they do not have hydrolytic activity in relation to casein, and milk is not their common medium. That is why Bifidobacteria need additional sources for their growth. It is known that with an increase in protein content in relation to lactose and the presence of milk protein hydrolysates, Bifidobacteria may consume fixed nitrogen from N-acetyl derivatives of milk carbohydrates.25
In this study, cultivation of microorganisms was carried out in accordance with the characteristics specified in the manufacturer’s documents. At the initial stage, dry bacterial concentrates and starter cultures were activated in the sterile skimmed milk in accordance with the fermentation parameters recommended by the manufacturer. The bulk starter was introduced into buttermilk and skimmed milk in an amount of 2% of the sample’s volume. The Bifidobacterium concentrate was dissolved in sterilized distilled water and then, being inactive, introduced into the milk material. Dry WPH was introduced into the milk material then the mixture was pasteurized at the temperature of (92±2) °С and cooled to the fermentation temperature. Titrated acidity was determined in the ready-made clots. This indicator is one of the most important in the production of fermented milk products, since it affects the production process, as well as the organoleptic characteristics of the product. Titrated acidity in the control samples was lower than in the test ones. Thus, the introduction of from 1-3 wt% of WPH did not slowdown the development of starter microorganisms. On the contrary, a clear active lactic acid process was observed in all test samples in comparison to the control samples. As a result of the fermentation, the titrated acidity index of the test samples was higher, and the increase in the buffer capacity promoted assimilating food sources by microorganisms from WPH.
It is possible to increase the ability of starter microorganisms for using the amino acids of WPH since the total charge of the carboxyl groups of amino acids in acidic medium is shifted towards more positive values, and dissociation is inhibited, as such this stimulates interaction with the negative surface of bacteria. In his research, James M. Jay has noted that bacterial cells have a residual negative charge.27 At a neutral or alkaline pH level, organic acids do not penetrate the membrane, while at the acidic pH level these components are not ionized and can enter a negatively charged cell.27 Figures 1-4 show the curves of titrated acidity in the control and test samples of skimmed milk and buttermilk with added WPH.
In majority of samples, fermentation goes through typical phases of the fermentation process. At the stage of adaptation, the titrated acidity slowly increases then, the acidity level rises sharply, which denotes the active development of microorganisms and further generation inhibition, being in this case the limiting value of acidity. The analysis shows that the lag phase of Streptococcus salivarius thermophilus (Fig. 1) lasted for about 1 hour, the active growth of microorganisms was observed in the next four hours, and from the 5th to the 6th hour the titrated acidity was not substantially changed. As a result, the optimal titrated acidity of the test samples was achieved after 4 hours of fermentation. According to the manufacturer’s information, the optimal titrated acidity is to be achieved after 10 hours of fermentation at the culturing temperature of 40°C. Therefore, the use of WPH accelerated the process of fermentation by 5 hours on the average that may shorten the technological process, reduce costs, and ensure the amount of beneficial microorganisms in the finished product to comply with the regulatory documents. The control samples demonstrated a sufficiently high titrated acidity index (skimmed milk – 90°T, buttermilk – 82°T) after the 4th hour of fermentation, but the texture was not sufficiently thick. On the contrary, at this fermentation time the test samples contained solid clots.
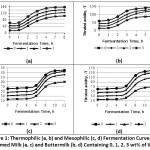 |
Figure 1: Thermophilic (a, b) and Mesophilic (c, d) Fermentation Curves for Skimmed Milk (a, c) and Buttermilk (b, d) Containing 0, 1, 2, 3 wt% of WPH. |
According to the manufacturer’s accompanying documentation, the period of lactococci fermentation is to last for 16 hours at 30°C. During cultivation, the lag phase was observed for 6 hours (Fig. 1), in both control and test samples. Further, active growth of microorganisms was observed during 4 hours. The titrated acidity of the test samples was slightly changed from the 10th to the 12th hour, which indicates the termination of fermentation. In the test samples, the titrated acidity of 90 °T was reached after 8 hours. Thus, it can also be concluded that the time of fermentation process was shortened in the presence of WPH.
 |
Figure 2: Acidophilic Fermentation Curves of Acid-Forming Strain (a, b) and Weak Acid-Forming Strain (c, d) of Skimmed Milk (a, c) and Buttermilk (b, d) Containing 0, 1, 2, 3% of WPH. |
As far as Lactobacillus acidophilus strains are concerned, the lag phase lasted for about 1 hour (Fig. 2). Their active growth was observed from the 2nd to the 4th hour of fermentation. When using an acid-forming strain and WPH in an amount of 2 wt% and 3 wt%, the titrated acidity attained values of more than 140°T after 4 hours. Thus, it is unacceptable to use this strain with the introduction of more than 1 wt% of WPH. Fermentation of a weak acid-forming strain proceeded approximately in the same way as in case of using thermophilic Streptococcus starter, though the clots demonstrated non-standard organoleptic parameters. No uniform clot formation was observed in the samples, but there were protein flakes.
The control samples obtained in a course of weak acid-forming strain fermentation showed low titrated acidity indices after 6 hours. In accordance with the manufacturer’s recommendations, this strain is to be fermented for 16 hours. The aim of the present research was to shorten the fermentation process by introducing WPH, and to develop acceptable consumer properties.
In the test samples obtained by using Bifidobacterium ferment, the titrated acidity increased only after addition of WPH (Fig. 3). Although, the samples with a 3 wt% of WPH reached an acidity of about 70°T, but there were no clots.
The fermentation process of the milk base with Lactobacillus plantarum starter was less active (Fig. 3). Adaptation of microorganisms to the milk medium was during 4 hours. The acidity level started to increase only after 5 hours of fermentation. The maximum titrated acidity in the control samples reached (40±2) °T. When introducing WPH, the test samples demonstrated following titrated acidity indices in buttermilk: -59°T, 70°T, 91°T with 1, 2 and 3 wt% of WPH respectively, and in skimmed milk: -78°T, 94°T, 101°T respectively. No clots were formed in the samples during six-hour fermentation. This strain of Lactobacillus plantarum is probiotic strain, and is more often used industrially as a culture protective against pathogenic and opportunistic microbiota. In this regard, it is advisable to use this starter culture as a probiotic component of multicomponent starter.
Kefir starter, prepared on kefir grains, was kindly provided for the research from the collection of AO Educational and Experimental Dairy Plant of the Vologda State Dairy Farming Academy named after N.V. Vereshchagin. The fermentation process was carried out for 18 hours, and in this process, the lag phase was observed for 6 hours on average. Theoretically, it would be possible to assume that the lag phase may take longer time period, but this never happened, despite the fact of increasing buffer capacity. Active development of microorganisms was observed from the 8th to the 14th hour. After the next 4 hours, the titrated acidity increased slightly. As a result of introducing WPH, the titrated acidity of the test samples demonstrated faster increase, and by the 12th hour of fermentation was more than 90°T. Since the total fermentation period with the kefir starter is quite long, and the species composition of the kefir starter microbiota is very diverse, it is impossible to conclude undoubtedly which microorganisms in this study were activated and influenced the fermentation acceleration, the kefir starter was not used.
 |
Figure 3: Bifidobacteria (a, b) and Plantarum (c, d) Fermentation Curves of Skimmed Milk (a, c) and Buttermilk (b, d) Containing 0, 1, 2, 3% of WPH. |
The process of Lactobacillus Bulgaricus fermentation of buttermilk and skimmed milk proceeded according to typical scheme (Fig. 4). The lag phase duration was 2 hours on average. The active growth of microorganisms was seen from the 2nd to the 6th hour. The titrated acidity of the test samples with the 3 wt% of WPH reached 100°T after the 4th hour of fermentation, and after the 5th and 6th hours with introduced 2 wt% and 1 wt. % of WPH respectively. Similarly to all previous cases, the titrated acidity in the test samples increased faster than in the control samples. Therefore, we can again conclude that the introduction of WPH shortens the process of skimmed milk and buttermilk fermentation when using addition of studied starter cultures.
 |
Figure 4: Kefir (a, b) and Lactobacillus Bulgaricus (c, d) Fermentation Curves of Skimmed Milk (a, c) and Buttermilk (b, d) Containing 0, 1, 2, 3% of WPH. |
Differences in the fermentation processes utilizing studied microorganisms derive from different amounts of lactic acid and final products produced (Fig. 5). Typically, lactic acid bacteria are divided into the groups of homolactates and heterolactates. Under the influence of various factors, for example, changes in carbohydrate concentration, pH of the medium, temperature, decrease in nutrients concentration, the overall picture of fermentation process may change. Therefore, based on the study of enzymatic properties, Stiles M. E. and W. H. Holzapfel provide for not only definite homolactates and definite heterolactates, but also optional heterolactates.28 The group of definite homolactates includes Lactobacillus acidophilus, Lactobacillus delbrueckii subspecies bulgaricus, etc. Optional heterolactates include Lactobacillusplantarum, etc.25, 28The conducted research has revealed differences in the fermentation processes subject to the types of milk materials, in both control and test samples. The index of titrated acidity in the skimmed milk samples is higher than in buttermilk samples. Apparently, the particular composition of these milk materials may give an explanation to the differences. Buttermilk is characterized by concentrating most of the fat globule membrane proteins, which have an increasing affinity for various low-molecular compounds, such as vitamins, minerals, and free fatty acids. These protein-bound compounds are no longer available for the use by starter microbiota.29 As a result, microorganisms lack from biologically active substances for growth and development, which affects their activity.
The study of the activity and dynamics of acid formation in both control and test samples of skimmed milk and buttermilk with WPH addition shows that in many samples, lactic acid rods and lactococci produce 0.6 to 0.8%and0.5% of lactic acid respectively (Fig. 6), which corresponds well to the literature data.27 The lowest lactic acid content was observed in the samples without WPH addition obtained by using Lactobacillus plantarum and Lactobacillus acidophilus (a weak acid-forming strain). The experimental samples showed an increase in lactate content after introducing WPH (1 wt% to 3 wt%).
Bifidobacterium starter culture was acting in different way. Bifidobacterium starter fermentation of the milk material was carried out for 6 hours and for 16 hours. After six-hour period, the lactate amount was less than 0.1 wt%. At the end of the 16-hour period of fermentation, the lactate amount was 0.6 wt% on average in both control and test samples. In both cases, no activation of lactic acid synthesis with the introduction of WPH was observed.
Analysis of the research results confirms that WPH contains nutrients that stimulate the development of lactic acid bacteria microorganisms of different taxonomic groups. This effect shows itself during the development of lactic acid bacteria in both skimmed milk and buttermilk with the addition of 1-3 wt% of WPH, which is proved by an increase in the amount of the formed lactic acid and an increase in the titrated acidity in studied milk materials (Fig. 5). The revealed regularity does not concern studied Bifidobacterium starter. This is probably due to the fact that milk materials are not its habitat and they practically do not produce β-galactosidase, and the introduction of WPH is not sufficient to activate its development. Obtained results correlate with the data presented by other authors.25, 28
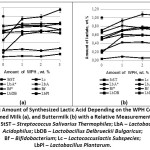 |
Figure 5: Amount of Synthesized Lactic Acid Depending on the WPH Content in Skimmed Milk (a), and Buttermilk (b) with a Relative Measurement Error of 5%. |
The fermentation period of milk materials is largely determined not only by the types of used lactic acid bacteria, but also depends on the individual characteristics of the starter culture strains. Since the present work involved commercially available microorganisms used in the dairy industry, and milk materials with WPH, the index evaluation, i.e. the average rate of acid formation is of practical interest. This index shows the amount of lactate produced per hour. Figure 6 presents the results on the acid formation rate. The analysis of the obtained data indicates intensifying acid formation by Lactobacillus acidophilus starter (LbA*, acid-forming strain) which develop in the skimmed milk with added WPH.
Buttermilk samples demonstrated higher rate of acid formation after introducing 2 wt% of WPH (LbA* acid-forming strain) and 1 wt% of WPH (LbA weak acid-forming strain) compared to control samples (Fig. 6). Thus, WPH may have an effect on the protein metabolism of bacteria, the finish products of which comprise not only lactic acid, but also other metabolites.
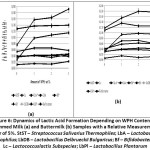 |
Figure 6: Dynamics of Lactic Acid Formation Depending on WPH Content in Skimmed Milk (a) and Buttermilk (b) Samples with a Relative Measurement Error of 5%. |
Organoleptic parameters (flavour, odour, texture, and colour) of the samples were evaluated according to the 5-point scale for each parameter. The results of the sample evaluation were summed up in points, and the quality was determined according to the total number of points. According to the organoleptic evaluation results, the number of points in the control samples was slightly higher than in the test samples (Table 2).
Table 2: Organoleptic Evaluation of the Samples (Total Score)
| Type of Starter Culture | Skimmed Milk | Buttermilk | ||||||
| Content of WPH, wt% | Content of WPH, wt% | |||||||
| 0 | 1 | 2 | 3 | 0 | 1 | 2 | 3 | |
| Streptococcus thermophilus | 20.0±0.0 | 19.0±0.5 | 18.7±0.7 | 18.1±0.6 | 20.0±0.0 | 19.1±0.5 | 18.8±0.5 | 18.2±0.7 |
| Lactococcuslactis | 19.7±0.3 | 17.3±0.9 | 17.1±0.6 | 16.4±0.7 | 19.7±0.1 | 17.3±0.8 | 16.9±0.8 | 16.4±0.7 |
| Lactobacillus acidophilus (acid-forming strain) | 18.3±1.2 | 16.8±0.6 | 15.2±0.6 | 13.6±0.2 | 18.2±0.7 | 16.9±0.7 | 15.3±0.8 | 13.9±0.6 |
| Lactobacillus acidophilus | 18.5±1.0 | 17.2±1.0 | 15.5±0.9 | 13.8±0.3 | 18.2±1.0 | 17.1±0.6 | 15.3±0.6 | 13.2±0.1 |
| Bifidobacterium (six-hour fermentation) | 17.0±0.9 | 16.0±0.6 | 14.7±0.6 | 12.4±0.4 | 16.7±0.8 | 15.7±0.4 | 14.2±0.6 | 12.4±0.2 |
| Bifidobacterium (sixteen-hour fermentation) | 18.1±0.7 | 16.9±1.1 | 16.1±0.6 | 14.0±0.2 | 17.9±0.7 | 16.9±0.6 | 15.9±0.8 | 14.0±0.7 |
| Lactobacillus plantarum | 18.1±0.9 | 16.4±0,6 | 14.5±0.5 | 13.9±0.5 | 18.2±0.7 | 16.5±1.2 | 14.3±0.4 | 13.6±0.4 |
| Kefir starter culture | 18.1±1.0 | 16.5±0.9 | 14.8±0,6 | 13.4±0.5 | 18.2±0.8 | 16.5±0,8 | 14.9±0.8 | 13.6±0.5 |
It should be noted, that the larger dose of added WPH had the lower value the evaluation point. When using Lactobacillus acidophilus, Bifidobacterium, Lactobacillus plantarum, the kefir starter culture and 2 wt% and 3 wt% of WPH in studied milk materials, experts identified salty and albuminous off-flavours with different saturation. The samples obtained by fermenting skimmed milk and buttermilk with the kefir starter culture and Lactobacillus acidophilus (acid-forming strain) with the introduction of 3 wt% of WPH demonstrated bitter taste. Buttermilk and skimmed milk samples fermented with Bifidobacterium starter for 6 hours also showed low points.
The samples obtained after 16-hour fermentation contained weak clots, which is considered unsatisfactory for consumer. When evaluating the odour, the experts gave fewer points to the test samples than to the control samples, which are explained by the WPH flavour.
Almost all test samples never exhibited a sufficiently thick clot texture.13 Buttermilk and skimmed milk samples fermented with Bifidobacterium, Lactobacillus plantarum, Lactobacillus acidophilus and the kefir starter culture did not exhibit intensive gelation after introducing 3 wt.% of WPH. When fermenting with Bifidobacterium starter for 16 hours with a 2 wt% of WPH, a weak clot was formed. The same texture was found in the samples obtained by fermenting with Lactobacillus acidophilus starter. When using Bifidobacterium starter for 6-hour fermentation, no clots were formed in studied milk materials. A loose clot was formed when fermenting milk materials with the kefir starter culture and Lactobacillus plantarum with 1 wt% of WPH. Mesophilic cultures produced weak gelation in all samples of buttermilk and skimmed milk. The test samples with Lactobacillus Bulgaricus demonstrated satisfactory consumer characteristics.
According to the expert-tasters, maximum points were given to buttermilk and skimmed milk samples fermented with Streptococcus salivarius thermophilus. These samples were characterized by a clean, pleasant, fermented-milk flavour and odour, a homogeneous dense clot with added 1-3 wt% of WPH. That is why thermophilic microorganisms are to be included in the starter for fermenting buttermilk and skimmed milk with WPH.
Conclusions
The research results denote the possibility of using WPH in producing fermented milk products. The use of certain amount WPH in studied milk materials contributes to the development of a number of fermenting microorganisms and accelerates the fermentation process, which would ultimately reduce production costs, and the overall cost of the product. Using WPH in the studied types of milk materials helps to increase the protein content, raise the biological value, and ensure a normalized amount of starter microbiota including probiotic bacteria. It has been found that the use of skimmed milk or buttermilk with added WPH as a basis material in the starter, probiotic cultures should be used in combination with thermophilic lactic streptococci to develop a fermented milk product for dietary prophylactic purposes.
Acknowledgements
Declared none.
Funding Source
This work is a self-funded thus nothing to declare.
Conflict of Interest
The author(s) confirm that this article content has no conflict of interest.
References
- Halsted, C. H. Dietary supplements and functional foods: 2 sides of a coin? J. Clin. Nutr. 2003; 77 (4): 1001S–1007S.
CrossRef - Phillips, M.M., Rimmer, C.A. Functional foods and dietary supplements. Anal. Bioanal. Chem. 2013; 405: 4323–4324.
CrossRef - Amitava, D., Chandan, K. S. Chapter 2. Nutritional Supplements and Functional Foods: Functional Significance and Global Regulations. In: Bagchi, D. Nutraceutical and Functional Food Regulations in the United States and Around the World. Second Edition. Amsterdam, Netherlands: Academic Press; 2014: 13-39.
CrossRef - Karanina, E. V., Selezneva, E. Y. Quality Assurance of Food Raw Materials and Food Products as the Main Factor of Safety of theConsumer Market, In: Nyila, M. Alfred. Listeria Monocytogenes, London, UK: IntechOpen; 2018: 405-416.
CrossRef - Tutelyan, V. A., Sukhanov, B. P., Kochetkova, A. A., Sheveleva, S. A., Smirnova, E. A. Chapter 18. Russian Regulations on Nutraceuticals and Functional Foods. In: Bagchi, D. Nutraceutical and Functional Food Regulations in the United States and Around the World. Second Edition. Amsterdam, Netherlands: Academic Press; 2014: 309-326.
CrossRef - Nakayama, K., Tagawa, R., Saito, Y., Sanbongi, C. Effects of whey protein hydrolysate ingestion on post-exercise muscle protein synthesis compared with intact whey protein in rats. Nutr. Metab. 2019; 16 (90): 1-7, DOI: 1186/s12986-019-0417-9.
CrossRef - Lockwood, C. M., Roberts, M. D., Dalbo, V. J., Smith-Ryan, A. E., Kendall, K. L., Moon, J. R., Stout, J. R. Effects of Hydrolyzed Whey versus Other Whey Protein Supplements on the Physiological Response to 8 Weeks of Resistance Exercise in College-Aged Males. J. Am. Coll. 2017; 36 (1): 16-27.
CrossRef - Gomes, A.M., Malcata, F.X., Klaver, F.A. Growth enhancement of Bifidobacterium lactis Bo and Lactobacillus acidophilus Ki by milk hydrolyzates. J. Dairy Sci. 1998; 81 (11): 2817‐2825.
CrossRef - Lazzi, C., Meli, F., Lambertini, F., Bottesini, C., Nikolaev, I., Gatti, M., Sforza, S., Koroleva, O., Popov, V., Neviani, E., Dossena, A. Growth promotion of Bifidobacterium and Lactobacillus species by proteinaceous hydrolysates derived from poultry processing leftovers. Int. Food Sci. Tech. 2013; 48 :341-349.
CrossRef - Nikolaev, I.V., Sforza. S., Lambertini, F., Ismailovac, D.Yu., Khotchenkova V.P., Volikc, V.G., Dossenab, A., Popova, V.O., Koroleva, O.V. Biocatalytic conversion of poultry processing leftovers: Optimization of hydrolytic conditions and peptide hydrolysate characterization. Food Chem. 2016; 197( A) :611-621.
CrossRef - Bezie, A., Regasa, H. The Role of Starter Culture and Enzymes/Rennet for Fermented Dairy Products Manufacture – A Review. Nutr. Food Sci. Int. 2019; 9 (2): 555756. DOI: 10.19080/NFSIJ.2019.09.555756.
- Paulino, B.C., de Souza Aquino, J., de Souza, E. L., de Sousa Gomes, J.A., Lins, P.P., Sena de Lima, S.A., dos Santos Alves, E.M., do Nascimento, E. Goat Milk Whey Improves Nutritional Status, Fecal Microbial Composition and Intestinal Morphology in Female Rats Fed a Westernized Diet and Their Offspring, J. Food Nutr. Res. 2019; 7 (4): 291-302.
CrossRef - Hickey, C.D., O’Sullivan, M.G,. Davis, J., Scholz, D., Kilcawley, K.N., Wilkinson, M.G., Sheehan, J.J. The effect of buttermilk or buttermilk powder addition on functionality, textural, sensory and volatile characteristics of Cheddar-style cheese. Food Res. Int., 2018; 103: 468-477.
CrossRef - Zhang, C., Zhang, Y., Li, H., Liu, X. The potential of proteins, hydrolysates and peptides as growth factors for Lactobacillus and Bifidobacterium: current research and future perspectives. Food Funct. 2020; 11 (3): 1946‐195.
CrossRef - Li, W., Zhang, Y., Li, H., Zhang, C., Zhang, Y., Uddin, J., Liu, X. Effect of soybean oligopeptide on the growth and metabolism of Lactobacillus acidophilus JCM 1132. RSC Adv. 2020; 10 (28): 16737-16748.
CrossRef
- Novokshanova, A.L., Topnikova, E.V., Ababkova, A.A. Analysis of amino acid composition of skim milk and buttermilk for the production of dairy drink when introducing whey protein hydrolysate (in Russian). Vopr. Pitan. 2019; 88 (3): 90-96.
- Russian National standard GOST 31658-2012, Skim milk – raw material. Specifications (in Russian). Moscow, Russia: Federal Agency on Technical Regulating and Metrology; 2012.
- Russian National standard GOST 34354-2017, Buttermilk and drinks on its base. Specifications. (in Russian). Moscow, Russia: Federal Agency on Technical Regulating and Metrology; 2017.
- ISO 22935-2:2009 [IDF 99-2:2009]. Milk and milk products — Sensory analysis — Part 2: Recommended methods for sensory evaluation. Geneva, Switzerland: International Organization for Standardization; 2009.
- ISO 22935-3:2009 [IDF 99-3:2009]. Milk and milk products — Sensory analysis — Part 3: Guidance on a method for evaluation of compliance with product specifications for sensory properties by scoring. Geneva, Switzerland: International Organization for Standardization; 2009.
- ISO 5492:2008 Sensory analysis – Vocabulary. Geneva, Switzerland: International Organization for Standardization; 2008.
- ISO/TS 22113:2012. Milk and milk products — Determination of the titratable acidity of milk fat, Geneva, Switzerland: International Organization for Standardization; 2012.
- Russian National standard GOST 32892-2014. Milk and milk products. Method for measuring active acidity (amended).(in Russian). Moscow, Russia: Federal Agency on Technical Regulating and Metrology; 2015.
- ISO 27205:2010/IDF 149:2010. Fermented milk products — Bacterial starter cultures — Standard of identity. Geneva, Switzerland: International Organization for Standardization; 2010.
- Quigley, E.M.M. Prebiotics and probiotics in digestive health. Clin. Gastroenterol. H. 17 (2), 2019, pp. 333- DOI: 10.1016/j.cgh.2018.09.028.
CrossRef - Golovin, N.A., Ganina, V.I. A new strain of bifidobacteria as a factor of improving the food product biosafety. (In Russian). Food Proc.: Tech. Technol. 2012; 4: 139-144.
- Jay, J.M., Loessner, M.J., Golden, D.A. Modern food microbiology. New York, USA: Springer, 2005. ISBN 978-0-387-23413-7.
- Stiles, M.E. Holzapfel, W.H. Lactic acid bacteria of foods and their current taxonomy. Int. J. Food Microbiol. 1997; 36 (1): 1-29.
CrossRef - Damodaran, S., Parkin, K.L., Fennema O.R. Fennema’s food chemistry. (Fifth edition). Boca Raton, FL, USA: CRC Press; 2017. ISBN 9781482208122.

This work is licensed under a Creative Commons Attribution 4.0 International License.






