Introduction
The oxidative rancidity of fats and oils in food is one of the serious problems of the modern society. Oxidative rancidity is responsible for off-flavor and odor, decreasing storage life, altering texture, nutritional quality also decreased due to the formation of secondary compounds which are potentially toxic.1 The need of addition of antioxidants is necessary to preserve flavor, color, shelf-life, and texture as well as vitamins.
Butylated hydroxytoluene (BHT) and Butylated hydroxyanisole (BHA) are synthetic antioxidants and commonly add in fats and oils to repress the development of oxidative rancidity. It has been reported that these synthetic antioxidants shows carcinogenic and toxic effects for human health.2, 3 Therefore, it is necessary to replace these antioxidants by effective antioxidants obtained from natural sources to prevent oxidative deterioration of fats and oils.
In India fruits and vegetable processing industries generates considerable quantities of waste. Fruits wastes and by-products are an ample source of antioxidant.4 Citrus peels are obtained from citrus fruit after the extraction of juice. It has been reported that flavonoids found in citrus fruits peel show health-related properties like antiviral, anti-inflammatory and anticancer activities.5, 6 Ref already added
Pomelo (Citrus maxima) is one of the large fruit of citrus family with a common name of pomelo or shaddock that belongs to the family of Rutaceae.7, 8 Pomelo peels are rich source of antioxidant as compared to its pulp.9
Use of waste as a source of antioxidants and polyphenols may have considerable economic benefit to food processors. The efficiency of many species like bergamot and pomelo nevertheless, has not been investigated yet. Therefore an efficient, environmentally and cheap utilization of these wastes is needed.
Usually organic solvents are used for the extraction of valuable natural materials. However, some of them are toxic for this reason; a food grade ethanol is widely used instead of methanol for the extraction of phenolic compounds from various citrus peels.10
In the present study, an attempt has been made to explore the utilization of pomelo peels extracts as a source of natural antioxidants and also studied for analyzing its effect in peanut oil during storage period of 21 days at room temperature.
Materials and Methods
Raw Material
Ripened and freshly harvested pomelo and freshly prepared peanut oil was procured from the local fruit market of Dehradun, India.
Extraction of Antioxidant from Pomelo Peels
Cold percolation method11 with little modification using ethanol as solvent was used for extraction of antioxidants from dried pomelo peels powders. In brief 10 gram of the dried pomelo peel powder was soaked in 100 ml of ethanol in a conical flask with cotton plugge and then kept in incubator shaker at 120 rpm at room temperature for about 24 hours. The extract was then centrifuged at 3000 rpm for 20 minutes and then supernatant was taken and concentrated using rotary vacuum evaporator at 40oC for about 4-5 days. The dry extract was store at 4oC for further analysis.
Extraction Yield
The dry extract obtained after centrifugation was weighed and extraction yield will be measure by the following formula
![]()
2, 2-diphenyl-1-picrylhydrazyl (DPPH) radical scavenging activity
Singh and Immanuel (2014) 12 method with some modification was applied for determination of scavenging activity. Briefly, 1 ml sample of variable concentrations (1–12 mg/ml) in ethanol was added to 4.0 ml of a DPPH solution (0.004 %) and at room temperature for 30 min kept in dark after that absorbance was measured at 517 nm. The scavenging activity was calculated by using the following correlation:
![]()
Where, AC = absorbance with control
AS = absorbance with sample
Free fatty acids
Free fatty acids in term of % oleic acid in peanut oil, were determined by AOAC (1990) 13 methods. Briefly, 1 gram of peanut oil sample was dissolved in 25 ml solvent mixture of petroleum ether and ethanol (2:1). After mixing and cooling the solution was titrated against N/10 KOH using phenolphthalein indicator. Free fatty acid (mg/g) was calculated by using the following equation
![]()
Where,
V = KOH (ml) exhausted by samples
C = KOH (M) concentration
p-Anisidine value
IUPAC method 2.504 14 was followed for the determination of p-anisidine value. Briefly, in 25 ml isooctane, 2 gram sample was dissolved and absorbance was measured at 350 nm. 1 ml p-anisidine (0.25%) in acetic acid (w/v) was mixed with 5ml of this mixture and incubate for 10 min at room temperature, after 10 min absorbance was measured at 350 by using a Spectrophotometer (UV-7804C). p-anisidine value was calculated according to following equation
![]()
Iodine Value
Wij’s method 13 was used for the determination of Iodine value in peanut oil samples.
Incorporation of Extract Antioxidants in Peanut Oil
Peanut oil, without any additives, was used for oxidative rancidity studies. In a 250 ml dark amber color glass bottle 100 ml peanut oil containing 1000, 1500 and 2000 ppm ethanolic pomelo peel extract were prepared separately. Peanut oil without antioxidant was consider as control and placed under same conditions. All samples were prepared in triplicate and stored at room temperature for 21 days. The samples were taken periodically after the intervals of 7 days to analyze the antioxidant activity of pomelo peels extract.
Data Analysis
The experiments were executed in triplets and readings were expressed as mean ± standard deviation. The significant differences among the means was calculated by One-way ANOVA followed by Tukey’s HSD test (p<0.05) using SPSS.
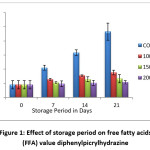 |
Figure 1: Effect of storage period on free fatty acids (FFA) value diphenylpicrylhydrazine Click here to View figure |
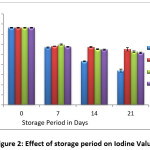 |
Figure 2: Effect of storage period on Iodine Value Click here to View figure |
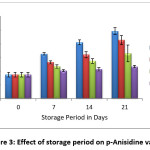 |
Figure 3: Effect of storage period on p-Anisidine value Click here to View figure |
Result and Discussion
% Extraction Yield of Pomelo peels
Table 1 represents % extraction yield of pomelo peel extract obtained from dried pomelo peel powder refluxing with ethanol, methanol, hexane and combination of chloroform and methanol (2:1) solvents.
Table 1 shows that minimum yield (11.1%) was obtained with chloroform +methanol (2:1), maximum (21.6%) with methanol whereas (15.9%) with ethanol and (12.7%) with hexane. From the comparison, the maximum yield of the present study was slightly higher with methanol and ethanol extract obtained by 15 in citrus peel.
Table 1: Extraction Yield (%) of Pomelo peel extract with different organic solvents
| Organic solvent | Pomelo peel extract yield (%) |
| Ethanol | 15.9% |
| Methanol | 21.6% |
| Hexane | 12.7% |
| Chloroform +methanol | 11.1% |
Total Antioxidant Activity
Due to presence of antioxidant compound in the peels of pomelo the stable free DPPH radical reduced to diphenylpicrylhydrazine and color of solution changed from deep violet to light yellow. The reduction was measured by the decrease in absorbance at 517 nm. The maximum scavenging activity of 88.8% was observed at 14mg/ml concentration. Scavenging activity of peels is due to the presence of flavonoids and polyphenols. The results were similar to12 for orange peel and lemon peel extract.
Effect of storage period on free fatty acids value of peanut oil with and without antioxidants
Table 2 and table 3 shows the free fatty acid value of peanut oil without (control) antioxidant and with antioxidant extracted from pomelo peel. The free fatty acid in control sample was 1.80±0.60% and from table 3 it was observed that, as the concentration of pomelo peel extract increased from 1000 ppm to 2000 ppm, the inhibitory effects on free fatty acids also increased considerably. After 21 days of storage period at room temperature , free fatty acids values of peanut oil treated with 1000, 1500 and 2000 ppm of pomelo peel extract were 3.52±0.32%, 2.66±0.59%
and 2.15±0.22%. Significant differences in free fatty acids were observed between the control and the peanut oil treated with different concentrations of pomelo peel extract. Hydrolysis of triglycerides and reaction of oil with moisture may be the reason of increased free fatty acids.16
The free fatty acids values were reduced from 9.32±1.2 % (control) to 2.66±0.59% and 2.15±0.22 %, The decrease in free fatty acids specify that autoxidation of peanut oil was highly diffident in the presence of pomelo peel extract at the concentrations of 1500 and 2000 ppm. Our findings favor the findings of.15
Table 2: Effect of storage period of free fatty acids, iodine value and p-anisidine value of peanut oil without Antioxidant
| Storage period (Days) | Free Fatty Acid | Iodine Value | p-Anisidine Value |
| 0 | 1.80 a ±0.60 | 76.33 a ±0.50a | 3.49 a ±0.33 |
| 7 | 4.2 b ±0.36 | 56.73 b ±0.50 | 6.54 b ±0.18 |
| 14 | 6.34 c ±0.38 | 43.2 c ±0.40 | 8.22 c ±0.35 |
| 21 | 9.32 d ±1.2 | 33.9 d ±1.54 | 9.85 d ±0.50 |
All the values with different superscripts are significantly different at P < 0.05.
Table 3: Effect of storage period of free fatty acids, iodine value and p -anisidine value of peanut oil with Antioxidant
| Storage period (Days) | Free fatty acids (%) | Iodine value | p- anisidine value |
| 1000ppm | |||
| 0 | 1.80 a ±0.60 | 76.33a ±0.50a | 3.49a ±0.33 |
| 7 | 2.56b±0.13 | 58.12b±0.25 | 5.31±0.22 |
| 14 | 3.11b±0.09 | 57.30b±0.50 | 6.42±0.45 |
| 21 | 3.52b±0.32 | 55.10b±1.56 | 8.55±0.66 |
| 1500ppm | |||
| 0 | 1.80 a ±0.60 | 76.33a ±0.50a | 3.49a ±0.33 |
| 7 | 2.10±0.12 | 59.7b±0.50 | 4.71±0.26 |
| 14 | 2.57±0.35 | 55.0c±0.45 | 5.49±0.33 |
| 21 | 2.66±0.59 | 52.8d±0.73 | 6.6±1.22 |
| 2000ppm | |||
| 0 | 1.80a ±0.60 | 76.33a ±0.50a | 3.49a ±0.33 |
| 7 | 1.95±0.41 | 57.56±0.45 | 4.10±0.15 |
| 14 | 2.05±0.36 | 54.66±0.55 | 4.30±0.24 |
| 21 | 2.15±0.22 | 51.46±0.66 | 4.68±0.18 |
Effect of storage period on Iodine Value of peanut oil with and without antioxidants
Table 2 and table 3 shows the Iodine value of peanut oil without (control) antioxidant and with antioxidant extracted from pomelo peel. The Iodine value in control sample was 76.33±0.50 and from table 3 it was observed that, on increasing the concentration of pomelo peel extract from 1000 ppm to 2000 ppm, inhibitory effects on Iodine value also increased. After 21 days of storage period at room temperature , Iodine value of peanut oil treated with 1000, 1500 and 2000 ppm of pomelo peel extract were 55.10±1.56, 52.8±0.73 and 51.46±0.66.
During storage decreasing trend in iodine value of fats and oils indicate the formation of secondary oxidation products which may cause the development of rancidity. Breaking of double bonds present in unsaturated fatty acids during storage may be the reason for decreasing iodine value.17 During study it observed that addition of pomelo peel extract retard the decreasing trends of iodine value. Our findings favor the findings of.15
Effect of storage conditions on p-anisidine value of peanut oil with and without antioxidants
p-anisidine value of oil and fat plays a vital role in the oxidation process. p-anisidine value plays an important role for evaluation of secondary lipid oxidation .
Table 2 and table 3 shows the p-Anisidine value of peanut oil without (control) antioxidant and with antioxidant extracted from pomelo peel. The p-Anisidine value in control sample was 3.49±0.33 and from table 3 it was observed that, as the pomelo peel extract concentration increased from 1000 ppm to 2000 ppm, inhibitory effects on p-Anisidine value also increased considerably. After 21 days of storage period at room temperature, p-Anisidine value of peanut oil treated with 1000, 1500 and 2000 ppm of pomelo peel extract were 8.55±0.66, 6.6±1.22 and 4.68±0.18. Significant differences in p-Anisidine value were observed between the control and the peanut oil treated with pomelo peel extract at different concentration.
The p-Anisidine value was reduced from 9.85±0.50 (control) to 6.6±1.22 and 4.68±0.18. The decrease in p-Anisidine value revealed that oxidative rancidity of peanut oil was effectively inhibited by peel extract of pomelo at concentrations of 1500 and 2000 ppm. Our findings favor the findings of.18
Conclusions
In current study, antioxidants were extracted from pomelo peels. Maximum extraction yield was found with methanol and then ethanol. Pomelo peel was utilized in food as natural antioxidant. The antioxidant activities of pomelo peels extracts revealed the free radical scavenging activity due to the flavonoids, phenolic acids, and their derivatives. It can be concluded that due to high antioxidant activity of pomelo peel extract, it can be used as a substitute of synthetic antioxidants for extending the shelf life of peanut oil.
Acknowledgements
The authors are grateful to Department of Food Technology, Uttaranchal University, Dehradun, Uttarakhand, India, for permitting to carry out the study at the Institute.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
References
- Alamed J, Chaiyasit W, McClements D J and Decker EA. Relationship between free radical scavenging and antioxidant activity in foods. Journal of Agricultural and Food Chemistry 2009; 57(7): 2969–2976. DOI: 10.1021/jf803436c
- Ito N, Hiroze M, Fukushima G, Tauda H, Shira T, and Tatematsu M. Studies on antioxidant: their carcinogenic and modifying effects on chemical carcinogensis. Food and Chemical Toxicology 1986; 24:1071–1081.
- Martin A D and Gilbert D. Enzyme changes accompanying liver enlargement in rats treated with 3-tert butyl-4-hydroxyanisole. Biochemistry Journal 1968; 106:22–27.
- Balasundram N, Sondra K and Samman S. Phenolic compounds in plants Andagri-industrial by-products: Antioxidant activity, occurrence, and potential uses. Food Chemistry 2006; 99: 191-203. https://doi.org/10.1016/j.foodchem.2005.07.042
- Huet R. Constituants des agrumes a` effet pharmacodynamique: les citroflavonoı¨des (Constituents of citrus fruits with pharmacodynamic effect: citroflavonoids). Fruits 1982; 37:267-271.
- Benavente-Garcia O, Castillo J , Marin F R , Ortuno A and Del Rio J A. Uses and properties of citrus flavonoids. J Agric Food Chem 1997; 45(12): 4505–4515. DOI: 10.1021/jf970373s
- Morton J F. Fruits of warm climates. 1987. http://www.hort.purdue.edu/newcrop/morton/pummelo.html.
- Scora R W. On the history and origin of Citrus. Bulletin of Torrey Botanical Club 1975; 102: 369–375.
- Isabelle M, Lee B L, Lim M T, Koh W P, Huang D. and Ong CH. Antioxidant activity and profiles of common fruits in Singapore. Food Chemistry 2010; 123(1): 77–84. https://doi.org/10.1016/j.foodchem.2010.04.002
- Diankov S , Karsheva M , Hinkov I. Extraction of natural antioxidants from lemon peels. Kinetics and antioxidant capacity. Journal of the University of Chemical Technology and Metallurgy 2011; 46(3):315-319.
- Parekh J and Chanda S. In vitro antibacterial activity of the crude methanol extract of Woodfordia fructicosa Kurz. Flower (Lythraceae) Braz. J Microbiol 2007; 38: 204-207. http://dx.doi.org/10.1590/S1517-83822007000200004
- Singh S and Immanuel G. Extraction of Antioxidants from Fruit Peels and its Utilization in Paneer. J Food Process Technol 2014; 5 (7): 1-5. http://dx.doi.org/10.4172/2157-7110.1000349
- Official method of analysis (15th ed.). Washington, DC: Association of Official Analytical Chemists (1990).
- Determination of the p-Anisidine Value, Method 2.504 (1987). IUPAC standard methods for the analysis of oils, fats and derivatives (7th ed., pp. 210–211). Oxford: Alden Press.
- Zia-ur-Rehman. Citrus peel extract – A natural source of antioxidant. Food Chemistry 2006; 99(3): 450–454. https://doi.org/10.1016/j.foodchem.2005.07.054
- Frega N, Mozzon M and Lercker G. Effects of free fatty acids on oxidative stability of vegetable oil. Journal of the American Oil Chemists Society 1999; 76(3): 325–329. https://doi.org/10.1007/s11746-999-0239-4
- Noor N, and Augustin M A. Effectiveness of antioxidant on the stability of banana chips. Journal of the Science of Food and Agriculture 1984; 35: 805–8112. https://doi.org/10.1002/jsfa.2740350719
- Ying Zhang, Lei Yang, Yuangang Zu, Xiaoqiang Chen, Fuji Wang and Fang Liu. Oxidative stability of sunflower oil supplemented with carnosic acid compared with synthetic antioxidants during accelerated storage. Food Chemistry 2010; 118(3): 656–662. https://doi.org/10.1016/j.foodchem.2009.05.038

This work is licensed under a Creative Commons Attribution 4.0 International License.







