Introduction
Proper nutrition is essential for good health and the prevention or treatment of disease. Most people in Northern and North-eastern Thailand often consume glutinous rice as a source of carbohydrates. Glutinous rice has been a staple of the Thai diet for a long time. However, high consumption of glutinous rice also increases the risk of diabetes.1 In general, the glutinous rice has high amylopectin content2 and thus, it has a high Glycemic Index (GI).1 This means it can be digested quickly and can cause a short term rise in blood glucose level.3 Brown glutinous rice is a healthier alternative to white rice and glutinous rice because brown rice consists of rice bran, which contains fiber and phytochemical compounds such as tocopherol, tocotrienols, g-oryzanols, sterol, and carotenoid. It also composes of free fatty acids such as 43.7% Oleic acid and 30.9% Linoleic acid.4 In addition, most of the proteins in rice bran are essential amino acids such as leucine, phenylalanine, valine, threonine, isoleucine, and histidine.5 Although it is high in nutrient contents, brown rice is not widely accepted as white rice. The endosperm of brown rice is securely covered with bran, which is high in fiber. The water penetration into the kernels is limited for the hydration and swelling.6 Soaking the brown rice before cook is a necessary method for the proposes of uniformly water absorption and softer texture. However, the simple soaking takes a long time and unsuitable for commercial production.7
Another problem posed by brown glutinous rice used as food is its hard texture. Brown rice has a stronger and firmer texture than white rice. Enzymatic polishing was utilized to degrade the rice bran to produce a softer texture. Das et al.,8 stated that xylanase could also improve the texture of rice. During the enzymatic process, the degradation of rice bran occurred, allowing the rice bran to absorb water. Hence, the texture of the rice was softer. Parboiling is also used to reduce cooking time and improve the texture of the Jasmine 85 and Nerica 14 rice varieties.9 There are several studies in Thailand that have demonstrated the effects of parboiling on texture of rice product. Sareepuang et al.,10 also found that cooked rice became softer after parboiling. Kladsuk and Sirisomboon11 evaluated the hardness characteristic of various cooked rice (parboiled rice, new jasmine rice, old jasmine rice, and white rice). The white rice was the hardest rice but the parboiled rice was the softest rice. Thus, parboiling could improve the texture of cooked rice product.
Some researchers stated that parboiling could increase Resistant Starch (RS). During the retrogradation process, the long chain amylose in rice may also form an amylose-lipid complex or a Resistant Starch (RS). The RS is not broken down by the enzymes in the small intestine, and ferments in the large intestine.12 Bertoft et al.,13 have concluded that starch gel rigidity is increased by amylopectin retrogradation. This result is influenced by the molecular structure of amylopectin. The amylopectin retrogradation is linked to form double-helices through the external glucan chains and then is packed into crystallites. The crystallites have been considered to define a limit of enzyme accessibility. The higher crystalline regions cause a decrease in enzymatic hydrolysis.14
The GI is associated with carbohydrates in specific foods, and the level indicates the effects it will have on blood glucose levels.3 Dipnaik and Kokare15 stated that the amylose/amylopectin ratio affects the GI because starches with higher amylose content are difficult to gelatinize and do not efficiently hydrolyze to glucose. Thus, glutinous rice has a low amylose/amylopectin ratio, resulting in a higher GI rating when compared to non-glutinous rice. Normally, rice with higher levels of RS has a lower GI value.16 The effects of parboiling techniques on GI have been investigated by Kale et al.,17 The results showed that parboiling at temperatures higher than 60°C could decrease the GI from 58 to 54. This was due to amylose-lipid complexes being formed and digested more slowly in the small intestine.18 Ready-to-eat food is food that is commercially prepared for ready to consume without any preparation. Additionally, it may be a shelf-stable and easy to carry product. Some essential properties of retort pouches include their flatness when standing, their flexibility, and the ability to withstand high-temperature conditions (121 p°C). These properties are required for high temperature and high-pressure processes.19
However, the application of parboiling to decrease the GI of brown glutinous rice in a retort pouch, with or without enzymatic hydrolysis, has not yet been investigated. Therefore, this research is aimed exclusively at finding the effect of parboiling with different gelatinization degree on texture, RS, and GI of ready to eat glutinous rice.
Materials and Methods
Materials
The brown glutinous rice of Oryza sativa L. CV. San-pah-tawng was obtained from Chiang Mai rice research center. From preliminary results, the chemical compositions of the brown glutinous rice were 11.84% moisture content, 7.43% protein, 1.40% fat content, 1.24% fiber content, 0.84% ash content, 77.25% carbohydrates (6.03% amylose and 70.02% amylopectin). Xylanase (Pentopan mono BG) from Sigma-Aldrich, USA was also used.
Preparation of brown glutinous rice
The effects of parboiling and degree of gelatinization on the quality of brown glutinous rice were investigated using a completely randomized design. There were two parboiling methods involved in preparation; parboiling the rice and parboiling the rice with enzymatic hydrolysis. The parboiled rice from various preparations (9 treatments) were compared to raw brown glutinous rice (control). Parboiled rice was prepared by soaking at three specific temperatures (50, 70 and 90°C) until the moisture content reached a 40-50% dry basis. The pre-cooked brown glutinous rice was steamed at atmospheric pressure until the gelatinization levels were at 60, 80, and 100%. From preliminary trials, the optimal soaking time of brown glutinous rice was measured by using a Differential Scanning Calorimeter (DSC). The dried samples were packed in DSC aluminum pans; then, distilled water was added to achieve a sample: water ratio of 1:3. The pan was then sealed and stabilized overnight at 4°C. Afterward, the temperature of the pan was raised from 30 to 120°C with a 10°C/min scanning rate. At the soaking temperature of 50°C, it took 90, 120, and 140 min of soaking time respectively in order to reach 60, 80 and 100% gelatinization. In order to reach 60, 80, and 100% gelatinization at 70°C, it took 40, 60, and 90 min soaking time, respectively. At 90°C it took 30, 40, and 45 min soaking time respectively in order to reach 60, 80, and 100% gelatinization. Then, the pre-cooked brown glutinous rice was dried at lower than gelatinized temperature (40°C, adapted from Cheevitsapon and Noomhorm20 and Hashemi et al.,21) permitting molecular reorganization22 for 36-h to 11 – 12% moisture content and stored at -25°C until use.
Parboiled rice with enzymatic hydrolysis was prepared by immersing brown glutinous rice in water at room temperature for 24-h. The brown glutinous rice was immersed in a 0.5% calcium carbonate solution held continuously at a temperature of 10°C lower than the gelatinization temperature, which was formed in the preliminary experiment (to be 40°C for 1-h until the moisture content was 37 – 39%). The calcium carbonate solution was rinsed, and the brown glutinous rice was cleaned with water. The enzyme hydrolysis was processed using xylanase with enzyme activity at 256 IU/mL in an acetate buffer pH 5 (0.1 mol/L) at 50°C. The degree of rice polishing was determined using May Grunwald’s reagent to estimate the suitable reaction time. The brown glutinous rice was steamed at atmospheric pressure until fully gelatinization. Then, the pre-cooked brown glutinous rice was dried at 40°C for 36-h and stored at -25°C before use.
Thermal process for product preparation
The thermal process by retort was used to produce ready-to-eat brown glutinous rice. The parboiled brown glutinous rice was soaked in water for 3-h before being steamed at atmospheric pressure until the rice was completely cooked. The cooked brown glutinous rice was vacuum-packed in a retort pouch with a width of 12 cm, maximum height of 4 cm and length of 14 cm using a vacuum packing machine (VM – 203, Audion Electro, Holland) and sterilized at 116°C with F0 equal to 5 min in water spray rotary retort (AR – 091, FMC, France). The experiments were done in triplicate.
Quality measurement of ready-to-eat brown glutinous rice
Color Measurement
The color CIE (L*, a* and b* values) of brown glutinous rice (raw material, parboiled rice, and sterilized rice) were measured by a Minolta Chromameter (CR-410, Konica-Minolta, Tokyo, Japan) using a white plate for calibration. Color differences (DE*) from control, and total color differences between the L*, a*, and b* values of the sample and standard, were also calculated. The DE* formula used was DE* = √(∆L*2 + ∆a*2 + ∆b*2
Texture Profile Analysis
The texture of the samples was assessed using a table-top texture analyzer (TA-XT2, Stable Micro System, UK). Two cycles of compression tests at a 90% strain at a speed of 1 mm/s were conducted. The cylinder-type measurement probe was 100 mm in diameter. The force-time curve was generated using Exponent 32 software with maximum force. The parameters collected from the curve were hardness, adhesiveness, cohesiveness, springiness, gumminess, and chewiness.23 The texture profile analysis was repeated 10 times for each sample.
Morphological Analysis
The morphological properties of rice granules of raw material, sterilized parboiled rice, and sterilized parboiled rice with enzymatic hydrolysis were studied using a Scanning Electron Microscope (JSM5410-LV, JEOL, JAPAN). Each grain was cracked in the middle, and the bottom end was trimmed off. A cross section of samples was mounted and then, spattered with gold. Images were taken using magnifications of 60x, 1000x, and 5000x at an accelerating potential of 5 kV as described by Kumar et al.,24
In vitro starch digestion and predicted Glycemic Index (pGI)
The rapid in vitro starch digestion procedure (adapted from procedures of Sopade and Gidley25 and Mahasukhonthachat et al.,26) was performed. Milled 500 mg samples combined with 1 mL artificial saliva a-amylase (250 U/mL carbonate buffer, pH 7) were incubated in a shaking incubator at 37°C for 10–15 s at 85 rpm. A mixture of 2.5 mL of pepsin solution and 0.02 M HCl (pH 2) was added and then incubated in the shaking incubator at 37°C for 30 min. Afterward, the digested sample was neutralised by 0.02 M NaOH (5 ml). A 0.2 M Sodium acetate buffer (pH 6, 25 mL) was used to adjust the pH. A 5 mL mixture of amyloglucosidase and pancreatin (28 U/mL and 2 mg/mL in Sodium acetate buffer, respectively) was added. The digested sample in the shaking incubator was collected at 10, 20, 30, 45, 60, 90, 120, 150, and 180 min. The amount of glucose was determined in duplicate with a glucometer (Accu-Chek Performa, Roche Diagnostics GmbH, Mannheim, Germany). The pGI was obtained from the digested starch curve vs. time (hydrolysis index, HI) at 90 min. The equation; pGI = 0.803 HI90 + 39.21 was applied using a first-order kinetic model.27
Resistant Starch (RS) Analysis
A Megazyme Resistant Starch kit28 was used to determine RS. The samples were reduced to digestible starch in a shaking water bath at 37°C for 16-h in order to form glucose with amyloglucosidase and pancreatic α-amylase. A 4 mL measure of ethanol was used to stop the reaction. Then, the sample was centrifuged at 5000 g for 10 min, and the sediment of RS was recovered. The supernatant was separated and washed twice with 50% ethanol to get digested starch (DS). The sediment was dissolved in 2 M KOH (2 mL) in an ice bath. Then, the sediment was neutralized with 1.2 M sodium acetate (8 mL). The RS was hydrolyzed with 0.1 mL of 3300 U/mL amyloglucosidase at 50°C. The samples were incubated at 50°C for 20 min, and then absorbance was seen at 510 nm. RS and DS were calculated as glucose × 0.9.
Sensory Evaluation
A sensory evaluation was performed for the brown glutinous rice in a retort pouch. The samples, weighing 50 g each, were packed into plastic cups with lids. The temperature of the served samples was between 35 – 40°C. A total of 50 consumer panelists were randomly selected. The sensory attributes of brown glutinous rice in the retort pouches, including color, appearance, odor, texture, and overall likeability were measured using a hedonic scale of 1 to 9, with 1 representing extremely dislike and 9 representing extremely like, respectively.
Statistical Analysis
A one-way analysis of variance (ANOVA) was calculated using the SPSS version 12.0.1. The means were compared using Duncan’s multiple range tests. The level of significance was set at p < 0.05. All experiments were performed in triplicate and data were presented as a mean ± standard deviation.
Results and Discussion
All samples (parboiled rice, and parboiled rice with enzymatic hydrolysis), were sterilized in retort and then, the physicochemical properties were analyzed compared to control (raw brown glutinous rice) as follows;
Color
The color of the brown glutinous rice after retorting from each preparation condition is shown in Table 1. The average lightness (L*) values ranged from 54.62-78.64. The average red/green (a*) values were 1.20 – 1.77. The average yellow/blue (b*) values were 10.12 – 17.91. The average color differences (DE*) were 22.26-25.41. The brown glutinous rice which was parboiled at 50°C until it reached 60% and 80% gelatinization exhibited the lightest colors. Most samples were not significantly different in yellow color. The reddest colors were found in the brown glutinous rice which was parboiled by soaking at 70°C and 90°C and steamed until achieving 100% gelatinization, and in the brown glutinous rice which was subjected to enzymatic hydrolysis before parboiling. Moreover, these also showed the highest DE* value. The color changes during the soaking process demonstrated that red and yellow bran pigments diffused from the bran into the endosperm. The color changes may demonstrate that the variations in soaking temperatures and time affected the rate of water absorption and the leaching of bran pigments. The rate of water absorption increased at higher temperatures. When the temperature decreased, the red and yellow bran pigments firmly combined.29 Furthermore, non-enzymatic browning, or a Maillard reaction, probably occurred and melanoidins were compound generated, caused the brown glutinous rice to be darker and yellower at higher temperatures.30
Table 1: Color in L*, a*, b*, and DE* of parboiled brown glutinous rice after retorting with different conditions.
| Soaking Temperature and % Gelatinization | L* | a* | b* | DE* |
| Control | 78.64 a ±0.06 | 1.18 d ±0.03 | 10.12 c ±0.13 | – |
| Soaking at 50°C | ||||
| 60% Gelatinization | 57.22 b ±0.95 | 1.20 d ±0.20 | 16.84 b ±0.07 | 22.45 |
| 80% Gelatinization | 57.53 b ±0.06 | 1.28 c ±0.21 | 17.17 ab ±0.21 | 22.26 |
| 100% Gelatinization | 55.96 cd ±0.01 | 1.50 b ±0.06 | 17.42 a ±0.16 | 23.83 |
| Soaking at 70°C | ||||
| 60% Gelatinization | 56.62 c ±0.21 | 1.20 d ±0.04 | 17.16 ab ±0.09 | 23.12 |
| 80% Gelatinization | 56.42 c ±0.51 | 1.41 b ±0.11 | 17.07 ab ±0.19 | 23.28 |
| 100% Gelatinization | 55.27 d ±0.16 | 1.72 a ±0.05 | 17.52 a ±0.02 | 24.99 |
| Soaking at 90°C | ||||
| 60% Gelatinization | 56.83 c ±0.21 | 1.25 c ±0.01 | 16.12 b ±0.09 | 22.62 |
| 80% Gelatinization | 56.37 c ±0.49 | 1.30 c ±0.07 | 17.91 a ±0.08 | 23.59 |
| 100% Gelatinization | 54.62 d ±0.16 | 1.74 a ±0.02 | 17.46 a ±0.62 | 25.41 |
| Enzymatic hydrolysis before parboiling | 54.83 d ±0.67 | 1.77 a ±0.17 | 17.52 a ±0.79 | 24.75 |
The different letters in the same column mean significant difference (p<0.05). L* – lightness; a* – red/green value; b* – yellow/blue value; DE* – the color difference.
Amylose and Amylopectin Content
All samples showed a higher amylose content after parboiling (Table 2). This was especially true of the brown glutinous rice using enzymatic hydrolysis before parboiling, as it showed the highest amylose content (7.99 g/100 g). This reflected a trend similar to a previous study reported by Nakamura et al.,31 It is possible that the xylanase was able to hydrolyze the glycosidic linkage between the xylose of the cell walls in order to release arabinose. The changes in amylose and amylopectin content can also be explained by the fact that the leached amylopectin content was higher than the leached amylose content, and that insoluble amylose-lipid complexes were generated during the parboiling process. Thus, the amylose content was usually higher in parboiled rice.32 Moreover, Milasinovic et al.,33 stated that the amylopectin is debranched to form linear chain amylose during heating. The content of debranched amylopectin is increased, a higher amount of linear chain amylose is produced. In the amorphous regions, amylose molecules may be hydrolyzed initially by amylases, and then the molecules may associate with resisting enzymatic digestion. Thus, the RS content is directly proportioned to the amylose content.34 The rice granules absorb water during soaking and swell up, causing ruptures in the grains and then decrease the hardness.35
Table 2: Amylose and amylopectin contents of parboiled brown glutinous rice after retorting with different conditions.
| Soaking Temperature and %Gelatinization | Amylose content (g/100 g) | Amylopectin content (g/100 g) | ||||
| Control | 6.03 c | ± | 0.08 | 70.02 a | ± | 0.33 |
| Soaking at 50°C | ||||||
| 60% Gelatinization | 6.18 c | ± | 0.28 | 69.83 a | ± | 0.09 |
| 80% Gelatinization | 6.24 c | ± | 0.22 | 69.48 ab | ± | 0.22 |
| 100% Gelatinization | 6.53 c | ± | 0.17 | 69.43 ab | ± | 0.07 |
| Soaking at 70°C | ||||||
| 60% Gelatinization | 6.22 c | ± | 0.36 | 69.36 ab | ± | 0.13 |
| 80% Gelatinization | 7.03 b | ± | 0.07 | 69.23 ab | ± | 0.02 |
| 100% Gelatinization | 7.10 b | ± | 0.13 | 69.10 b | ± | 0.09 |
| Soaking at 90°C | ||||||
| 60% Gelatinization | 6.18 c | ± | 0.14 | 69.48 ab | ± | 0.06 |
| 80% Gelatinization | 7.06 b | ± | 0.31 | 69.07 b | ± | 0.31 |
| 100% Gelatinization | 7.31 ab | ± | 0.24 | 69.05 b | ± | 0.09 |
| Enzymatic hydrolysis before parboiling | 7.99 a | ± | 0.14 | 68.11 b | ± | 0.26 |
The different letters in the same column mean significant difference (p<0.05).
Textural Properties
Table 3 shows the textural characteristics of the brown glutinous rice obtained from different soaking times and different gelatinization percentages. The hardness values of the rice ranged from 73.02 – 88.23 N. The hardness of the brown glutinous rice, which used enzymatic hydrolysis before parboiling had the lowest value. The brown glutinous rice which was soaked at 50°C and steamed until 60% gelatinization showed the greatest hardness among the samples using parboiling without enzymatic hydrolysis. However, there were no differences in adhesiveness, cohesiveness, and chewiness values among the treated samples. The parboiled rice at the lower temperatures was harder than that at higher temperatures because the higher temperature may increase the hydration and diffusion rate.36 Nakamura et al.,31 have reported that the decomposition of the cell wall by cellulase and xylanase can cause a softening of cooked rice. Decreased hardness was also presumably due to the amounts of amylose-lipid complex and the amylose contents after parboiling. The complexes formed on the endosperm interface (rich of lipid and proteins) were more concentrated and contributed to kernel splitting when the endosperm swelled.37
Table 3: Textural properties of parboiled brown glutinous rice with different conditions.
| Soaking Temperature and % Gelatinization | Hardness (N) | Adhesiveness (N.s) | Cohesiveness | Chewiness (N) |
| Control | 88.23 a ± 0.25 | -0.56 a ± 0.06 | 0.33 a ± 0.07 | 18.23 a ± 0.11 |
| Soaking at 50°C | ||||
| 60% Gelatinization | 77.71 b ± 0.15 | -3.45 b ± 0.38 | 0.30 b ± 0.02 | 16.37 b ± 0.14 |
| 80% Gelatinization | 77.20 b ± 0.18 | -3.44 b ± 0.24 | 0.24 b ± 0.04 | 15.97 b ± 0.07 |
| 100% Gelatinization | 75.19 c ± 0.47 | -3.76 b ± 0.19 | 0.26 b ± 0.03 | 15.15 b ± 0.18 |
| Soaking at 70°C | ||||
| 60% Gelatinization | 76.43 b ± 0.08 | -3.53 b ± 0.36 | 0.25 b ± 0.02 | 16.24 b ± 0.08 |
| 80% Gelatinization | 75.81 c ± 0.11 | -3.37 b ± 0.27 | 0.28 b ± 0.01 | 15.47 b ± 0.12 |
| 100% Gelatinization | 75.38 c ± 0.50 | -3.11 b ± 0.13 | 0.22 b ± 0.01 | 14.83 b ± 0.09 |
| Soaking at 90°C | ||||
| 60% Gelatinization | 76.58 b ± 0.13 | -3.25 b ± 0.10 | 0.25 b ± 0.01 | 15.52 b ± 0.22 |
| 80% Gelatinization | 75.24 c ± 0.11 | -3.80 b ± 0.03 | 0.27 b ± 0.01 | 14.70 b ± 0.03 |
| 100% Gelatinization | 74.95 c ± 0.03 | -3.95 b ± 0.06 | 0.26 b ± 0.03 | 14.11 b ± 0.31 |
| Enzymatic hydrolysis before parboiling | 73.02 d ± 0.23 | -3.88 b ± 0.15 | 0.26 b ± 0.02 | 14.23 b ± 0.13 |
The different letters in the same column mean significant difference (p<0.05). NS indicated that no difference significantly (p≥0.05).
Morphological Properties
Scanned images of rice viewed under the Scanning Electron Micrographs (SEM) at different magnification ranges are shown in Figure 1. The SEM of rice obtained from various preparation methods showed variations in their sizes. Morphological properties showed that an uneven distribution of granule sizes in the starches of different preparation methods was due to hereditary characteristics, and was mainly responsible for any variations in their properties. The control group showed fully starch granules, while the parboiled brown glutinous rice showed a loose structure with higher surface smoothness. The irregular rough surface of the granules was due to starch damaged during pre-gelatinization. The brown glutinous rice which was subjected to enzymatic hydrolysis before parboiling exhibited a smooth surface, resulting from the utilization of xylanase to break down polysaccharides into xylose at b-1,4-xylan bond. Thus, an increase in water absorption was observed. Starch granules expand in volume, and then a homogeneous kernel structure was formed. The parboiled rice then became stale after cooling.38
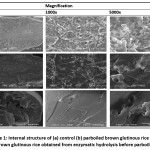 |
Figure 1: Internal structure of (a) control (b) parboiled brown glutinous rice and (c) brown glutinous rice obtained from enzymatic hydrolysis before parboiling. Click here to View figure |
RS and In vitro Starch digestibility
The highest RS contents were observed in parboiled rice which was soaked at 90°C and steamed until 100% gelatinization and in the brown glutinous rice which used enzymatic hydrolysis before parboiling (Table 4). RS content was higher when rice was soaked at a higher temperature. As a result, it can be stated that heating to temperatures above the gelatinization temperature allows recrystallization, leading to more amylopectin retrogradation.39 Bertoft et al.,13 studied the properties of retrogradation in glutinous rice starches about the internal structure of amylopectin. The formation of the helical structure in the internal segments of amylopectin was found. As the backbone model of amylopectin, some branches are dispersed into the crystalline region within granules. Debranching enzymes were easily penetrated to these branches for hydrolysis of starch to produce linear chain, and then intra-molecular double-helices are also formed, results in high RS.40 Moreover, the higher RS content was observed in parboiled rice, which was soaked until a higher degree of gelatinization. This is due to the rearrangement of the amylose and amylopectin chains. The amylopectin is debranched to form linear chain amylose during heating. According to increasing in double helices structure of amylose, the RS content tends to increase. Because of starch digestibility is decreased when the degree of retrogradation is increased.41 The enhancement of amylose content and RS content resulted in a lower GI value. GI values for the rice ranged from 61.32 – 66.32%. The brown glutinous rice which was soaked at 90°C and steamed until 100% gelatinization has the lowest GI value (61.32 or 7.72% reduction). This is a similar value when compared to the previous study of Kale et al.,17 which investigated the effect of parboiling on GI of Basmati rice, the result showed that there is 6-8% GI value reduction. During cooking, the heat, the amount of water, and the cooking time affected the GI of food. Water and heat expanded the starch granules to varying degrees during cooking. Formation of complexes among amylose, proteins, and lipids that resist enzymatic degradation can decrease the rate of starch digestion.42
Table 4: Resistant Starch (RS) and Glycemic index (GI) of parboiled brown glutinous rice with different conditions.
| Soaking Temperature and % Gelatinization | RS (%) | D0 | D¥ | K (min-1) | H90 | HI | pGI |
| Control | 0.20 e ± 0.54 | 10.59 | 30.92 | 0.00 | 33.76 | 40.22 | 66.32 a ± 0.45 |
| Soaking at 50°C | |||||||
| 60% Gelatinization | 2.03 d ± 0.14 | 12.45 | 33.51 | 0.01 | 30.09 | 35.94 | 63.37 b ± 0.21 |
| 80% Gelatinization | 2.03 d ± 0.13 | 11.53 | 33.89 | 0.02 | 30.54 | 35.63 | 63.73 b ± 0.02 |
| 100% Gelatinization | 2.05 d ± 0.23 | 13.77 | 35.46 | 0.01 | 28.57 | 34.99 | 63.35 b ± 0.03 |
| Soaking at 70°C | |||||||
| 60% Gelatinization | 2.25 c ± 0.07 | 14.98 | 35.24 | 0.01 | 29.98 | 39.11 | 63.28 bc ± 0.31 |
| 80% Gelatinization | 2.44 b ± 0.02 | 14.46 | 35.42 | 0.01 | 27.61 | 34.45 | 62.64 c ± 0.12 |
| 100% Gelatinization | 2.54 b ± 0.04 | 14.69 | 35.59 | 0.01 | 27.85 | 34.59 | 61.38 d ± 0.15 |
| Soaking at 90°C | |||||||
| 60% Gelatinization | 2.19 c ± 0.03 | 13.23 | 34.60 | 0.01 | 29.99 | 39.78 | 63.29 b ± 0.23 |
| 80% Gelatinization | 2.64 a ± 0.02 | 13.12 | 35.92 | 0.01 | 29.18 | 36.45 | 61.57 d ± 0.02 |
| 100% Gelatinization | 2.64 a ± 0.02 | 14.73 | 36.47 | 0.02 | 27.53 | 35.78 | 61.32 d ± 0.03 |
| Enzymatic hydrolysis before parboiling | 2.60 a ± 0.05 | 14.43 | 35.88 | 0.01 | 29.28 | 35.93 | 62.72 c ± 0.24 |
The different letters in the same column mean significant difference (p<0.05).
Predicted digestibility starch at 90 (H90) and 180 min (HI) using the modified first-order kinetic model of Mahasukhonthachat et al.,26 Predicted GI (pGI) using the equation of Goni et al., (1997) as pGI = 39.21+(0.803× H90).
There was interaction among RS, GI, and the amylopectin content of brown glutinous rice when parboiled without enzymatic hydrolysis, as shown in Figure 2. The regressive equation for a relationship correlating amylopectin content and GI was formulated as shown in equation (1) with a regression coefficient (R2) of 0.9425. The polynomial equation for RS as affected by amylopectin content was regressed in equation (2) with an R2 of 0.9704. The results indicated that starches with higher amylopectin content were less RS than starches with lower concentrations of amylopectin. This showed a trend similar to that found by Mir et al.,43 who confirmed relationships between GI and RS, starches with higher RS exhibiting a lower GI. This is due to the higher available carbohydrate, so the lower the glycemic response.
You et al.,14 claimed that In vitro digestibility and RS contents of waxy rice starches are influenced by the molecular structure of amylopectin. Long chains of amylopectin could make for higher and stable crystalline structures because of the increasing of H-bond between chains, which led to a higher resistance to enzymatic digestion. Amylopectin retrogradation is related to the ability of the external amylopectin chains to form double helices. The external chains of amylopectin and chain segments of amylose interact to form double helices during retrogradation.44 Hence, the greater rigidity persuades a decrease in enzyme susceptibility, then the RS increase and the GI decrease.
y = 1086.1x2 – 4625.2x + 6627 equation (1)
Where x = amylopectin content,
y = GI y = 10.071x2 – 85.599x + 188.01 equation (2)
Where x = amylopectin content, y = RS
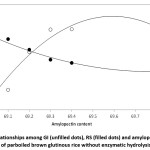 |
Figure 2: Relationships among GI (unfilled dots), RS (filled dots) and amylopectin content of parboiled brown glutinous rice without enzymatic hydrolysis Click here to View figure |
Sensory Evaluation
Mean scores of all sensory attributes for each sample, as evaluated by 50 participants, are shown in Table 5. The overall likeliness of the brown glutinous rice obtained from various soaking times and % gelatinization showed that brown glutinous rice which was soaked at 90C and steamed until 100% gelatinization was the most acceptable, with a mean score of 8.2 ± 0.6. There were significant differences among the treatments (p ≤ 0.05). Specifically, it received the highest ratings for color, appearance, odor, and texture scores of 7.4 ± 0.8, 7.3 ± 0.6, 7.0 ± 0.6, and 7.2 ± 0.6, respectively. Thus, the brown glutinous rice obtained from soaking at 90C and steaming until 100% gelatinization showed the highest sensory evaluation score and the lowest GI value. It is the optimal preparation condition for parboiled rice. As the acceptability and preference depend on culture, people who live in Northern of Thailand prefer glutinous rice with fluffy, tender, and not too loose. However, the brown glutinous rice with enzymatic hydrolysis has the burst grains and then loses its fluffiness. The rice becomes soggy and too lose texture.
Table 5: Sensory test of parboiled brown glutinous rice with different conditions.
| Soaking Temperature and % Gelatinization | Color | Appearance | Odor | Texture | Overall likeliness | ||||||||||
| Soaking at 50°C | |||||||||||||||
| 60% Gelatinization | 6.7 a | ± | 0.9 | 6.0 b | ± | 0.8 | 6.3 bc | ± | 0.7 | 6.3 bc | ± | 0.7 | 6.2 bc | ± | 0.8 |
| 80% Gelatinization | 6.5 ab | ± | 0.6 | 6.6 b | ± | 0.5 | 6.8 ab | ± | 0.8 | 6.3 bc | ± | 0.6 | 6.5 bc | ± | 0.8 |
| 100% Gelatinization | 6.7 ab | ± | 0.7 | 7.0 ab | ± | 0.9 | 6.9 ab | ± | 0.9 | 6.8 b | ± | 0.8 | 7.0 b | ± | 0.9 |
| Soaking at 70°C | |||||||||||||||
| 60% Gelatinization | 5.8 b | ± | 0.7 | 6.3 b | ± | 0.5 | 6.3 bc | ± | 0.8 | 6.2 bc | ± | 0.6 | 6.6 bc | ± | 0.9 |
| 80% Gelatinization | 6.5 ab | ± | 0.7 | 6.4 b | ± | 0.7 | 6.5 ab | ± | 0.5 | 6.3 bc | ± | 0.5 | 6.4 bc | ± | 0.6 |
| 100% Gelatinization | 7.0 ab | ± | 0.9 | 7.4 ab | ± | 1.1 | 6.7 ab | ± | 0.9 | 7.5 a | ± | 0.9 | 7.5 ab | ± | 0.6 |
| Soaking at 90°C | |||||||||||||||
| 60% Gelatinization | 6.5 ab | ± | 0.5 | 6.1 b | ± | 0.6 | 6.1 bc | ± | 0.7 | 6.5 bc | ± | 0.5 | 6.6 bc | ± | 0.6 |
| 80% Gelatinization | 6.7 ab | ± | 0.5 | 6.6 ab | ± | 0.5 | 6.9 ab | ± | 0.6 | 6.8 b | ± | 0.7 | 7.2 b | ± | 0.7 |
| 100% Gelatinization | 7.4 a | ± | 0.8 | 7.3 a | ± | 0.6 | 7.0 ab | ± | 0.6 | 7.2 ab | ± | 0.6 | 8.2 a | ± | 0.6 |
| Enzymatic hydrolysis before parboiling | 5.7 b | ± | 0.7 | 6.0 b | ± | 0.7 | 5.8 c | ± | 0.7 | 5.6 c | ± | 0.9 | 5.4 c | ± | 0.7 |
The different letters in the same column mean significantly difference (p<0.05).
Conclusion
The effects of two preparation methods on the qualities of parboiled glutinous rice showed that parboiling not only affected the nutritional composition, color, and texture of glutinous rice but also decreased the Glycemic index (GI) value and improved the Resistance starch (RS) content. Soaking temperature and degree of gelatinization are important factors of parboiled rice. Because of the thermal history of samples even for temperature above the gelatinization temperature influence starch retrogradation, RS content, and GI value. There are relationships between GI and RS with amylose content. The starches with higher amylose and RS exhibiting a lower GI. Moreover, the higher RS content than non-parboiled rice was observed in parboiled rice which was soaked until a higher degree of gelatinization. The enzymatic hydrolysis improved both the palatability of glutinous rice and the physicochemical properties. Glutinous rice soaked in hot water at 90°C with 100% gelatinization had the highest overall preference score with a slight decrease in the GI value of the product. According to the increase of starch retrogradation during storage, the change of the GI, RS, and texture of brown glutinous rice as well as the effect of anti-staling agents to improve texture during storage should be further study.
Acknowledgments
This research received financial support from National Research Council of Thailand.
We are grateful to Asst. Prof. Dr. Yuttana Pimolsiripol for assistance with writing this article.
Funding
The author(s) received financial support from the National Research Council of Thailand.
Declarations of Interest
The authors declare that they have no conflicts of interest.
References
- Nakayama T, Nagai Y., Uehara Y., Nakamura Y., Ishii S., Kato H., Tanaka Y. Eating glutinous brown rice twice a day for 8 weeks improves glycemic control in Japanese patients with diabetes mellitus. Nutrition and Diabetes. 2017; 7(5): 1-6.
CrossRef - Li H., Fitzgerald M. A., Prakash S., Nicholson T., Gilbert R. G. The molecular structural features controlling stickiness in cooked rice, a major palatability determinant. Scientific reports. 2017; 7: 43713.
CrossRef - Shozib H.B., Bhowmick S., Jahan S., Hoque F., Alam M. S., Das S. C., Alam S., Alam M. M., Siddiquee M. A In vivo screening for low glycemic index (GI) rice varieties in Bangladesh and evaluate the effect of differently processed rice and rice products on GI. Biojournal of Science and Technology. 2017; 5: m170001.
- Chen Y., Wang M., Ouwerkerk P.B.F. Molecular and environmental factors determining grain quality in rice. Food and Energy Security. 2011; 2(1): 1-22.
CrossRef - Faccine G. L., Miotto L. A., Vieira L. N., Barreto P. L. M., Amante E. R., Sensorial and rheological properties of a new organic rice bran beverage. Rice Science, 2009. 16(3): 226–234.
CrossRef - Zhang X, Wang L, Cheng M, Wang R, Luo X, Li Y, Chen Z. Influence of ultrasonic enzyme treatment on the cooking and eating quality of brown rice. Journal of Cereal Science. 2015; 63:140–146.
CrossRef - Yu Y., Ge L., Zhu S., Zhan Y., Zhang Q. Effect of presoaking high hydrostatic pressure on the cooking properties of brown rice. Journal of Food Science and Technology. 2017; 52:7904–7913.
CrossRef - Das M., Gupta S., Kapoor V., Banerjee R., Bal S. Enzymatic polishing of rice – A new processing technology. LWT – Food Science and Technology. 2008; 41: 2079-2084.
CrossRef - Ayamdoo A. J., Demuyakor B., Saalia F. K., Francis A. Effect of varying parboiling conditions on the cooking and eating/sensory characteristics of Jasmine 85 and Nerica 14 rice varieties. American Journal of Food Technology. 2014; 9(1): 1-14.
CrossRef - Sareepuang, K., Siriamornpun, S., Wiset, L., Meeso, N. Effect of soaking temperature on physical, chemical and cooking properties of parboiled fragrant rice. Journal of Agricultural Science. 2008; 4(4): 409-415.
- Kladsuk S., Sirisomboon P. The 6th TSAE International Conference 2013. 2013; 151-153.
- Raigond P., Ezekiel R., Raigond B. Resistant starch in food: a review. Journal of the Science of Food and Agriculture. 2014; 95: 1968–1978.
CrossRef - Bertoft E., Annor G. A., Shen X., Rumpagaporn P., Seetharaman K., Hamaker B. R. Small differences in amylopectinfine structure may explain large functional differences of starch. Carbohydrate Polymers. 2016; 140: 113–121.
CrossRef - You S. Y., Lim S. T., Lee J. H., Chung H. J. Impact of molecular and crystalline structures on in vitro digestibility of waxy rice starches. Carbohydrate Polymers. 2014; 112:720-735.
CrossRef - Dipnaik P., Kokare P. Ratio of amylose and amylopectin as indicators of Glycemic index and in vitro enzymatic hydrolysis of starches of long, medium and short grain rice. International Journal of Research in Medical Sciences. 2017; 5(10): 4502-4505.
CrossRef - Steffi S., Fiastuti W., Rahmawati R. Effect of cooling of cooked white rice on resistant starch content and glycemic response. Asia Pacific Journal of Clinical Nutrition. 2015; 24(4): 620-625.
- Kale S.J., Kale P.N., Jha S. K. Effects of parboiling steps on starch characteristics and glycemic index of basmati (pb1121) rice. International Journal of Agriculture Sciences. 2017; 9(49): 4826-4831.
- Joke A. P., Liesbeth J. D., Lieve L., Elin O. S., Inger M. B., Jan A. D. Functionality of short chain amylose-lipid complexes in starch-water systems and their impact on in vitro starch degradation. Journal of Agricultural and Food Chemistry. 2010; 58: 1939–1945.
CrossRef - Praharasti A. S., Nugroho J., Rahardjo B., Nurhikmat A. Evaluation for sterilization of rendang packaged by flexible retort pouch using finite difference method. International Symposium on Air Breathing Engines Proceedings. 2016; 9 – 11 August 2016, C03-1-7.
- Cheevitsopon E., Noomhorm A., Effects of parboiling and fluidized bed drying on the physicochemical properties of germinated brown rice, International Journal of Food Science and Technology, 2011; 46(12): 2498 – 2504.
CrossRef - Hashemi J., Borghei A., Shimizu N., Kimura T. Optimization of final moisture content of paddy in flat bed dryer with consideration of minimum losses and marketability. Journal of Agricultural Sciences and Natural Resources of Khazar. 2005; 3: 72-82.
- Leethanapanich K. Impacts of feedstock and parboiling conditions on quality characteristics of parboiled rice. University of Arkansas (Thesis). 2015.
CrossRef - Wassanai W., Roengchai T., Jidapah R., Tonapha P., Chanakan P. A comparison of the physicochemical properties of parboiled rice exported from Thailand and five consumer countries. Chiang Mai University Journal of Natural Sciences. 2017; 16(2): 99-112.
CrossRef - Kumar C. S., Malleshi N. G., Bhattacharya S. A comparison of selected quality attributes of flours: effects of dry and wet grinding methods. International Journal of Food Properties. 2008; 11(4): 845-857.
CrossRef - Sopade P.A., Gidley, M.J. A rapid in-vitro digestibility assay based on glucometry for investigating kinetics of starch digestion. Starch/Stärke. 2009; 61: 245–255.
CrossRef - Mahasukhonthachat K., Sopade, P. A., Gidley, M. J. Kinetics of starch digestion in sorghum as affected by particle size. Journal of Food Engineering. 2010; 96: 18–28.
CrossRef - Goni I., Garcia A. A., Saura C. F. A starch hydrolysis procedure to estimate glycemic index. Nutrition Research. 1997; 17: 427–437.
CrossRef - AOAC Method. 2002.02. Method for resistant starch in foods. Association of Official Analytical Chemists Method.
- Lamberts L., Rombouts, I., Brijs, K., Gebruers, K., Delcour, J. A. Impact of parboiling conditions on Maillard precursors and indicators in long-grain rice cultivars. Food Chemistry. 2006; 110(4): 916-922.
CrossRef - Phonchathri H., Insalud N., Poonnoy P. Effect of storage materials and conditions on grain color and anthocyanin content of colored parboiled rice. Khon Kaen Agriculture Journal. 2017; 45(1): 1066-1073.
- Nakamura S., Machida K., Ohtsubo K. Search for cell wall degrading enzyme of world wide rice grains by PCR and their effects on the palatability of rice. Bioscience. Biotechnology. Biochemistry. 2012; 76(9): 1645-1654.
CrossRef - Pathmanathapillai D., Prabhaharan M., Balakumar S. Effects of the various parboiling methods on the proximate composition, amylose and resistant starch of two rice varieties from Sri Lanka. Imperial Journal of Interdisciplinary Research (IJIR). 2016; 2(4): 295-300.
- Milasinovic M. S., Radosavljevic M. M., Dokic L. P. Effects of autoclaving and pullulanase debranching on the resistant starch yield of normal maize starch. Journal of the Serbian Chemical Society. 2010; 75(4): 449-458.
CrossRef - Zhu L. J., Liu Q. Q., Wilson J. D., Gu M. H., Shi Y.C. Digestibility and physicochemical properties of rice (Oryza sativa L.) flours and starches differing in amylose content Carbohydr Polym, 2011; 86(4),1751-1759.
CrossRef - Li H., Prakash S., Nicholson T. M., Fitzgerald M. A., Gilbert R. G., The importance of amylose and amylopectin fine structure for textural properties of cooked rice grains, Food Chemistry. 2016; 196: 702-711.
CrossRef - Nawaz M. A., Fukai S., Bhandari B. Effect of different cooking conditions on the pasting properties of flours of glutinous rice varieties from Lao people’s democratic republic. International Journal of Food Properties. 2016; 19: 2026–2040.
CrossRef - Ashley H., Jinn J. R., Mauromoustakos A., Wang Y. J. Effect of parboiling on milling, physicochemical, and textural properties of medium- and long-grain germinated brown rice. Cereal Chem. 2016; 93(1): 47-52.
CrossRef - ABVista Food Ingredient. Enzyme xylanase. http://www.abvista.com/ products/ enzymes/xylanase. Accessed May 29, 2015.
- Fisher D. K., Thompson D. B. Retrogradation of maize starch after thermal treatment within and above the gelatinization temperature range. Cereal chemistry. 2015; 74(3): 344-351.
CrossRef - Zhu F. Relationships between amylopectin internal molecular structure and physicochemical properties of starch. Trends in Food Science & Technology. 2018; 78: 234-242.
CrossRef - Wang S., Li C., Copeland L., Niu Q., Wang, S. Starch retrogradation: A comprehensive review. Comprehensive Reviews in Food Science and Food Safety. 2015; 14: 568–585.
CrossRef - Giuberti G., Gallo A. Reducing the Glycemic index and increasing the slowly digestible starch content in gluten-free cereal-based foods: a review. International Journal of Food Science and Technology. 2018; 53: 50–60.
CrossRef - Mir J. A., Srikaeo, K., García, J. Effects of amylose and resistant starch on starch digestibility of rice flours and starches. International Food Research Journal. 2013; 20(3): 1329-1335.
- Vamadevan V., Bertoft E. Impact of different structural types of amylopectin on retrogradation. Food Hydrocolloids. 2018; 80: 88-96.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






