Introduction
Fermentation is among the oldest methods of food preservation.1 Traditional fermented foods have their functionalities enhanced through development of flavours, aromas and textures, and are preserved through acidic, alcoholic or alkaline fermentations and enriched with protein and many nutrients.2,3 African fermented maize based products include Uji (porridge) andBusaa in Kenya, kenkey, banku, ogi, and koko from Ghana and Nigeria.4,5 Microorganisms are found in fermented foods as a result of indigenous microbiota of substrates, utensils and containers or added as starter culture.6 The major microorganisms implicated in fermentation of maize products include lactic acid bacteria (LAB) such as Lactobacillus fermentum, Lactobacillus reuteri, Lactobacillus rhamnosu, Lactobacillus casei, Lactobacillus plantalum, Lactobacillusbulgaricus,Lactobacillus acidophilus, Lactobacillus alimemtarius, Lactococcus lactis, Entorococcus faecium, Leuconostoc mesentoroids and Pediococcus spp as well as yeasts.7–9 Traditional fermentation depends on inoculation of earlier fermented product; however, commercial starter cultures are currently available to ensure constancy and reliability of processes and products.10 Traditional fermented products are mostly processed in a non-sterile environment which creates a selection of microorganisms that produce the desired end product however there is an increased risk of spoilage and unsafe products11,12 as a result of uncontrolled fermentation. Thepresent studycontributes towards determining the microbial status of fermented maize products from Western Kenya and isolating inherent lactobacilli which can be used in the development of starter culture for a controlled fermentation of Mkarango and other cereal products. This may result in enhancedfermentation process formaize products leading to optimization of their desired qualities as well as identifying the lactobacilli with probiotic properties. In Western part of Kenya, maize is a staple food of great socio- economic importance and a source of income and employment for millions of farming families in the region.13,14 In the present study, the microbial populations of maize flour, overnight soaked dough, fermented cooked porridge, Mkarango and Busaa were enumerated and the naturally present lactobacilli isolated. The microbial and biochemical profiles of the 6 days spontaneous Mkarango fermentation process were also determined.
Materials and Methods
Sampling of Fermented maize based products
Fermented maize based products were sampled from Kakamega and Homa Bay Counties in western Kenya where the population predominantlyconsumes maize moreover, maize is a staple food of great socio- economic importance and a source of income and employment.13,14 Five fermented maize based products: flour, overnight soaked dough, spontaneously fermented cooked porridge, Mkarango (roasted fermented maize flour) and Busaa (anaerobically fermented alcoholic beverage from maize) were sampled from five randomly selected producers, each providing all the five products. A total of 25 samples (15 from Kakamega and 10 from Homa Bay) were analysed. Samples were aseptically packed and taken to University of Nairobi for Laboratory analysis.
Enumeration of total viable count of fermented maize based products
Total viable count (TVC) was determined on Plate count agar (PCA, HMEDIA M091-500G, India). The plates were incubated at 37oC for 48 hours as per AOAC Method 966.23.15 The plates with 30-300 colonies were considered.
Enumeration of Lactobacillus
The Lactobacillus were enumerated on the deMan, Rogosa and Sharpe agar (MRS,1.10661.0500, Merck KGaA, Germany) which was incubated anaerobically in gas pack jar at 300C for 72 hours as per the method by De Man et al.,16 The colonies with different shape, size and morphology were sub-cultured on MRS until pure clear colonies observed. Identification was done by gram staining and biochemical tests.
Enumeration of yeast and moulds of fermented maize based products
Yeast and moulds were enumerated on Potato Dextrose Agar (PDA, HMEDIA M096-500G, India) as per FDA.17 The medium was acidified with 10% tartaric acid to pH 3.5 and incubated at 300C for 5 days. The petri dishes containing less than 150 colonies were counted. For identification of yeast and moulds, areas of fungal growth were selected and observed under a microscope
Enumeration ofEnterobacteriaceae of fermented maize based products
By the use of pour plate technique, Violet red bile agar (VRBA, HMEDIA M049-500G, India) on which glucose (10%) was added was used to enumerate Enterobacteriaceae. as per method by ISO 21528-2.18 Petri dishes were incubated at or 370C for 24 hours. The plates containing less than 150 typical colonies were counted using a colony counter. Pink to red colonies were isolated and used for biochemical confirmation, oxidase and glucose fermentation tests as per ISO 21528-2.18 For oxidase test, isolated colonies were streaked on filter paper containing oxidase reagent. A positive detection was observed by colour change within 10 seconds. For glucose fermentation test, using an inoculation needle, selected colonies were stabbed into tubes of glucose agar and incubated at 37oC for 24 hours. The positive reaction was indicated by yellow colour development.
Characterization of Lactobacilli in fermented maize based products
In order to characterize Lactobacilli, the confirmation and biochemical tests were conducted as described in the literature.19–21 Colonies of Lactobacilli were examined for cell morphologies, cell arrangement, Gram staining, spore formation and catalase test. Rod shaped, Gram positive, catalase negative and non- spore forming isolates were purified and characterised by biochemical tests (sugar fermentation, growth in 6.5% Sodium chloride and motility test) and the isolates were identified in reference to Bergey’s Manual of Determinative of Bacteriology as per Holt et al.,22
Gram staining test and cell morphology
Colonies of lactobacilli formed on MRS were examined for Gram staining using Gram staining kit (SKU-Pack Size, SIGMA ALDRICH, USA) as described by Pyar and Peh.23 The colour, morphology and arrangement were observed under light microscopy (LABOMED, USA) using oil immersion objective.
Spore formation test
The method by Goyal et al.,24 was used for endospore formation test. A smear of lactobacilli isolate was aseptically made on a slide and heat fixed. The slides were placed ona boiling water bath and a primary stain (malachite green) was applied and for 5 minutes then rinsed with water until clear water is observed. The counter stain (safranin) was applied on slide for 20 sand rinsedwith water again and the slides wereblot dried then observed under the light microscope(LABOMED, USA). Red coloured (non-spore forming) isolates were kept and subjected to further tests.
Catalase test
Catalase test was performed as per the method described by Kale.25 An isolated presumptive lactobacillus colony was streaked on a slide and a drop of 3 % hydrogen peroxide was added. The absence ofoxygen effervescence indicated the negativeresponse.
Purification of Lactobacilli isolates from fermented products from western Kenya
Colonies of lactobacilli with different features were streaked on MRS (1.10661.0500, Merck KGaA, Germany) and incubated at 30 ˚C. The pure cultures were then streaked on Tryptone Soy Agar (TSA) from which they were used for other tests. The isolates were stored in brain heart infusion (BHI) supplemented with yeast extract to avoid their damage at lower temperature.
Sugar fermentation test for Lactobacilli
An overnight culture of each isolate on Tryptone Soy Agar (TSA) was used to test the fermentation of different sugars. The carbohydrate fermentation test was done on the following sugars: D-Glucose, Lactose, Galactose, Mannitol, Mannose, L-arabinose, D-xylose, Cellobiose, Rhamnose and Fructose. Nutrient broth was supplemented by each sugar (1%). As per the method by Reiner26 sterile tubes of the solution were inoculated with loopful of isolated Lactobacilli and incubated at 370 C for 24 hours after which each tube was inoculated with 2 drops of Phenol red and incubated for 24 hours. Tubes in which red colour changed to yellow indicated fermentation of sugars and acid production. An uninoculated medium was used as control.
Growth of Lactobacilli in 6.5% Sodium chloride
As per Bhardwaj et al.,27 the isolates were inoculated in MRS broth having NaCl concentration of 6.5%. The tubes were observed for the presence or absence of growth.
Motility test for Lactobacilli
The method byRamírez-chavarín et al.,28 was used. Sulfide inodole motility (SIM) was used as motility medium. The isolates were inoculated into the centre of a tube containing SIM medium by stabbing method. The motility of the bacteria was checked by observing the spreading growth in the incubated tubes.
Microbial changes during fermentation of maize dough used to make Mkarango
Mkarango is a roasted fermented maize flour from wester Kenya.It can be served as such or used further for Busaa making. Mkarango was produced as per Aka et al.,4 The white maize flour was mixed with water (45%) to form the stiff dough. The mixture was well covered and left at ambient temperature to ferment for six days
The total viable count, Lactobacilli, Enterobacteriaceae, moulds and yeasts counts were monitored daily as described above.
Determination of pH and titratable acidity during maize dough fermentation
The pH of fermenting dough was measured using a pH meter (pH 315i, WTW82362 Weilheim, Germany). The pH meter was calibrated using buffers of pH 4 and 7. Titratable acidity of fermenting dough was determined as per method by Kunyanga et al.,16 A sample of 10 ml was titrated with 0.1 N NaOH using phenolphthalein as an indicator. The titratable acidity was expressed as percent lactic acid.
Statistical analysis
Samples collected from Western Kenya producers were analysed in a completely randomized block design where a producer was considered as a block in order to analyse samples which have received the same treatment. Five producers (5 blocks) provided five products making 25 total number of sample. All samples were tested in duplicate and the mean values wererecorded.
The Analysis of Variance (ANOVA) was performed and the level of significance was evaluated at p≤0.005; separation of means was done using Duncan test. In order to determine the microbial and biochemical changes during fermentation, three replications of fermentation were made and the mean values with standard deviations were reported.
Results
Microbial composition of maize based fermented products from Western Kenya
Microbial content of maize based products analysed is shown in Table 1. Total plate counts significantly (p≤0.05) varied among the samples.The highest values were observed in overnight soaked dough (9.47 log cfu/g) and the lowest were found in fermented cooked porridge (6.93 log cfu/g). On the other hand, Lactobacilli count differed significantly (p≤0.05) among the products ranging from 5.12 log cfu/g in fermented cooked porridge to 7.51 log cfu/g in roasted maize flour (Mkarango).
Table 1: Microbial content (log cfu/g) of selected maize based fermented products from Western Kenya
|
Parameters |
Flour | Overnight Soaked dough | Fermented Cooked porridge | Roasted (Mkarango) |
Busaa |
|
TVC |
8.82 ±0.77a |
9.47 ±0.16a | 6.93 ±1.76b | 7.70 ±1.54b |
8.58±0.36a |
|
Lactobacilli |
7.43 ±0.76a |
7.51 ±0.38a | 5.14±2.32b | 5.97±2.21d |
7.38 ±0.59a |
|
Mould and Yeast |
5.16±0.37b |
7.08±0.82a | 4.64±1.41c | 5.94±1.36b |
7.25±0.16a |
|
Enterobacteriaceae |
4.87±0.12b |
6.16 ±0.64a | 4.48±1.31c | 4.84±2.21b |
4.99±0.94b |
TVC: Total Viable Count, Values are mean ± standard deviation, Values with different superscript in the same row are significantly different (p≤0.05).
Mould and yeast counts significantly (p≤0.05) varied withinthe products,with the highest counts (7.25 log cfu/g) being observed in Busaa and lowest counts (4.64 log cfu/g) in fermented cooked porridge. Enterobacteriaceae counts were not significantly different (p>0.05) in flour, porridge, Mkarango and Busaa.
Characterization of lactobacilli from fermented maize based products
Table 2 shows the presumptive lactobacilli isolates from maize based products from Western Kenya and their response to biochemical tests. Presumptively, fiftyfour (54) isolates of Lactobacilli were obtained from 25 samples collected from Western Kenya as well as from controlled laboratory fermentation. After morphological and biochemical test, 16 isolates were identified to be probably Lactobacillus fermentum, 10 isolates to beLactobacillus Plantarum, 6 isolates to be Lactobacillus cellibiosis, 5 isolates to beLactobacillus heliviticus and 4 isolates to beLactobacillus casei. Other isolates were presumptively Lactobacillus lactis, Lactobacillus mesontoroides and Lactobacillus acidophilus with 3 isolates each. Lactobacillus fermentum were the most predominantin all products with 29.62% of the isolates followed by Lactobacillus plantarum (18.51%). Lactobacillus copophilus and Lactobacillus brevis were fewest (3.70%). Further molecular tests are needed to confirm these isolates.
Table 2: Biochemical test results of Lactobacillus isolates
| Isolate Code | Number | XY | GL | LC | FR | GAL | RH | CL | MN | MA | AR | 6.5%S | MOT | GR | CAT | Probable Species |
| A3, A6, A7,A8, A9, B1, B2, B4, B6, B9, C1, C4, C5, D1, D5, E1, | 16 | + | + | + | + | + | – | + | + | + | + | + | – | + | – | L. fermentum |
| A2, A4, A5, C2, C8, C9, C10, D2, E4, E9 | 10 | – | + | + | + | – | – | + | + | + | + | – | – | + | – | L.plantarum |
| B8, B10, D9, E6, E10, | 6 | + | + | + | + | – | – | + | – | + | – | + | – | + | – | L. cellibiosis |
| B3, B5, B11, C6, D11 | 5 | – | + | + | + | – | – | + | – | – | – | – | – | + | – | L. heliviticus |
| A1, A11, C3, D3 | 4 | + | + | + | – | + | + | + | – | – | + | + | – | + | – | L. casei |
| B7, E5, E7 | 3 | + | + | – | + | + | + | + | + | + | + | – | – | + | – | L. lactis |
| D10, E3, E8 | 3 | + | + | + | + | – | – | + | – | – | + | – | – | + | – | L. mesontoroides |
| C7, D4, D6 | 3 | – | + | + | + | + | + | – | + | + | – | + | – | + | – | L. acidophilus |
| D7, D8 | 2 | + | + | + | + | + | – | – | – | – | + | + | – | + | – | L. copophilus |
| A10, E2 | 2 | + | + | + | + | + | – | – | – | – | + | + | – | + | – | L. brevis |
+= Positive results, -= Negative Results, L= Lactobacillus, XY= D-xylose, GL=D-Glucose, LC= Lactose, FR=Fructose, GAL=Galactose, RH= Rhamnose, CL= Cellobiose, MN= Mannitol, MA= Mannose, AR= L-arabinose. S= Sodium Chloride, MOT= Motility, GR= Gram staining, CAT= Catalase, L.= Lactobacillus, A=Isolates from Busaa, B= Isolates from overnight soaked, C=Isolates from roasted dough (Mkarango), D= Isolates from fermented cooked porridge and E= isolates from maize flour.
Microbial changes during fermentation process of Mkarangodough
Figure 1 illustrate the change in microbial content of fermenting Mkarango dough. Lactobacilli were observed to significantly (p≤0.05) increase from the beginning (6.62 log cfu/g) up to the third day of fermentation (12.46 log cfu/g) followed by a slow decrease from fourth to the sixth day
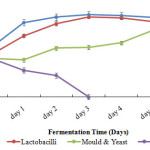 |
Figure 1: Lactobacilli, Mould and Yeast and Enterobacteriaceae counts during fermentation Click here to View figure |
A significant (p≤0.05) decrease in Lactobacilli was observed on the sixth day (11.32 log cfu/g). Moulds and yeasts significantly (p≤0.05) increased from the fourth up to sixth day of fermentation rising from 8.42 log cfu/g to 10.53 log cfu/g. The Enterobacteriaceae were found to gradually decrease significantly (p≤0.05) during the fermentation period dropping from 5.99 log cfu/g to 1.0 log cfu/g.
Titratable acidity and pH of fermenting Mkarango dough
Titratable acidity and pH change during fermentation of maize dough is shown in Figure 2. Titratable acidity and pH were monitored during fermentation cycle of Mkarangodough at the interval of 24 hours (1 day) for 6 days.
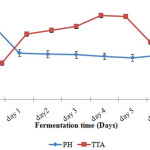 |
Figure 2: Titratable acidity and pH changes during fermentation of maize dough Click here to View figure |
The pH significantly (p≤0.05) decreased right from day 1 to day 5. It decreased from pH 6.64 on day1and steadily decreased throughout the fermentation process to 3.64 on day 5. The pH of the dough slightly increased on the sixth day. Titratable acidity significantly (p≤0.05 increased from 0.32% up to 0.73% on the fifth day and it significantly (p≤0.05) decreased on the sixth day.
Discussion
Microbial composition of fermented products from Western Kenya
The highest total viable count was observed in the overnight soaked dough and lowest count in fermented cooked porridge. This reduction of microbial load may be attributed to the effect of heat treatment during cooking which is known to have a lethal effect on microorganisms where many non-sporulating bacteria are inactivated at temperatures above 55 oC.29 The traditional fermentation in the village has little or no control of the microbial growth; this explains the elevated number of microorganisms that was found in all the products. The low pH of the fermented products and the heat treatment by roasting and/or cooking would make these products safe for consumption. However, poor hygienic conditions in which they are processed and handled may lead to the introduction of other microorganisms including pathogenic ones. The levels of Enterobacteriaceae which are hygiene indicators were high in all analysed products. Health Protection Agency30set 104 cfu/g (4.0 log cfu/g) as the upper limit for Enterobacteriaceae content in all ready-to-eat foods placed on the market. The presence of Enterobacteriaceae shows that a failure occurred during processing and their absence indicates that proper hygienic conditions were maintained during the food manufacturing process.31 Enterobacteriaceae include many pathogens especially Salmonellae, Shigella dysenteriae, Yersinia enterocolitica and Escherichia coli that may cause foodborne illness in people who regularly consume these foods.31
Yeast and mould count was most numerous in Busaa. It has been reported byKirui et al.,32 that Busaa production involves conditions (ingredients, moisture and ambient temperature) which favour the growth of yeast and mould. Other products analysed contained different counts of yeast and moulds. Most moulds and yeasts have low resistance to heat and do not survive thermal processes in low-acid foods and when found in processed foods, it is an indicator of poor processing or contamination.33 Moulds and yeasts prevalence in fermented Mkarango (Roasted) from Western Kenya could be attributed to the fact that during and after roasting, the processors continuously touch the dough with their hands in order to make uniform size roasted product. This may introduce other microorganisms including moulds and yeasts. It could also be attributed to poor storage conditions after production.
Lactobacilli count was high in all analysed fermented products and the highest values were observed in overnight soaked dough. This may be attributed to their natural presence in maize flour and on the availability of all growth requirements including moisture and sugars.The values in the present study are similar to those of Ogbonnaya and Chidinma34 in their studies on Akamu, a traditional fermented maize food (7.50log cfu/g after 24 hours of fermentation) and Omemu3 (7.91 log cfu/g) in Ogi. It has been reported that ingestion of live cells of some strains of lactobacilli in suitable amounts confer a number of positive physiological effects on the host including maintenance of a healthy and equilibrated intestinal flora and enhanced resistance to intestinal infections.35
Characterization of lactobacilli
Predominance ofLactobacillus fermentum and Lactobacillus plantarum isolates was also observed by Ijabadeniyi,36 Adegbehingbe37 and Atter et al.,38 who reported Lactobacillus plantarum and Lactobacillus fermentum to be the most abundant microorganisms in ogi (fermented gruel from maize), Masa (fermented food product produced from maize), mawe (fermented maize meal from Benin) and burukutu (traditional beer in Ghana). Lactobacilli fermentum is considered as an essential microorganism for maize fermentation and avails more starch for other organisms.39 None of the isolate was motile. This is an indicator that the isolates were lactobacilli since lack of motility is a characteristic of lactic acid bacteria especially the Lactobacilli acidophilus.23,25 However, it has been reported that certain species of Lactobacillii spp like Lactobacilli curvatus NRIC 0822 are motile and it was discovered that they contain smaller fragellum with peritrichous organisations.40
There was different behaviour toward growth of lactobacilli in 6.5% salt concentration. Most of the isolates were able to grow in such high concentration.Cai et al.,41 reported that lactic acid bacteria grow better in environment of less than 5% Sodium chloride however, some species such as Lactobacillus casie and Lactobacillus fermentum have shown the ability to grow in a medium of 7% Sodium chloride. Salt is an important additive that finds application in food processing and preservation,40 therefore the ability to growth in saline medium providelactobacilli a wide range of application especially in fermented food.The present findings are also in accordance with the report that many species of lactobacilli have the ability to withstand high salt concentrations and this makes them useful in food processing compared to other species.42 All isolates were able to ferment at least five different sugars. The breakdown of sugars into organic acid is important in preservation of fermented food and an indicator that they can feature into starter culture strain formulation.43 This is in agreement with the findings ofMithun et al.,19 Akinleye et al.,21 and Bhardwaj et al.,27 who reported the fermentation of simple sugars into acid by lactic acid bacteria.
Microbial and acidity changes during fermentation of Mkarango
The dough fermentation process was predominated by Lactobacilli. This is in line with other studies done on similar fermented products which reported that Lactobacillus bacteria were the predominant microorganisms involved in the fermentation of products like Ogi,20,21 Masa37 and Gari.44
Lactic acid bacteria grow in many foods and quickly decrease the pH to 3.5 or less and competing microorganisms can no longer grow.16 Moreover, Lactobacilli are able to produce hydrogen peroxide and antibiotics that have a significant effect on other organisms that would, otherwise, cause food spoilage.45 This may be related to the observed gradual decrease of the Enterobacteriaceae during fermentation dropping from 5.99 log cfu/g to less than 1 log cfu/g at the third day of fermentation.
Yeasts and moulds have also been observed to grow during fermentation of maize flour. Yeasts are widely spread in the environment and are mostly found in liquid foods that have sugars and they survive in a wide range of pH, pH 2 to above pH 9.17 The starch content of maize flour may have served as a substrate and the acidity produced by breakdown of sugars by Lactobacilli might have restricted their growth since they survive in medium pH values less than 3.38, which was the lowest observed during fermentation.
The pH of the fermented dough decreased and the titratable acidity increased correspondingly. This acidic condition of the product could be due to the lactic acid produced by Lactobacilli which were found predominant during maize dough fermentation.16 On the other hand, the observed increase in pH and a decrease in titratable acidity towards the end could be explained by the facts that moulds, which were shown to be growing rapidly towards the end of fermentation (6th day), have the ability to consume acids, which increase the pH and lower the titratable acidity of food products.46
Conclusion
The microbial status of finished fermented maize based products is predominated by Lactobacilli and a significant number of Enterobactriaceae whose growth is restricted by acidic environment. The presumptive isolates from fermented maize products from western Kenya are predominantly Lactobacilli especially Lactobacillus fermentum and Lactobacillus plantarum. However, further molecular tests are recommended to confirm these isolates so that they can be used in selection of starter culture for cereal products.
Acknowledgments
We acknowledge the contribution of the National Commission for Science Innovation and Technology (NACOSTI) of Kenya and the Department of Food Science, Nutrition and Technology at the University of Nairobi for facilitating the current research.
Conflict of interest
The authordeclares no conflict of interest.
Source of fund
The current research project was funded by the National Commission for Science Innovation and Technology (NACOSTI), Kenya.
References
- FAO. Traditional Fermented Food and Beverages for Improved Livelihoods. (Marshall E, Mejia D, eds.). Rome, Italy: Food and Agriculture Organization; 2012.
- Ogbonnaya N, Bernice Chidinma C. Studies on Akamu, a traditional fermented maize food. Rev Chil Nutr.2012;39:180-184
- Omemu AM. Fermentation dynamics during production of ogi, a Nigerian fermented cereal porridge. Rep Opin.2011; 3(4):8-17
- Aka S, Konan G, Fokou G, Dje Koffi M, Bonfoh B. Review on African traditional cereal beverages. Am J Res Commun. 2(5):103-153 :
- Gwirtz JA, Garcia-Casal MN. Processing maize flour and corn meal food products. Ann N Y Acad Sci.2014;1312(1):66-75
- Tamang JP, Watanabe K, Holzapfel WH. Review: Diversity of microorganisms in global fermented foods and beverages. Front Microbiol.2016;7(377):1-28
CrossRef - Muyanja CMBK, Narvhus JA, Treimo J, Langsrud T. Isolation, characterisation and identification of lactic acid bacteria from bushera: A Ugandan traditional fermented beverage. Int J Food Microbiol.2003;80(3):201-210
CrossRef - Mohania D, Nagpal R, Kumar M, et al. Molecular approaches for identification and characterization of lactic acid bacteria. J Dig Dis.2008;9(4):190-198
CrossRef - FAO/WHO. Taxonomy and Physiology of Lactic Acid Bacteria, Effects and Function on Nutrition. Report of a Joint FAO/WHO Expert Consultation on Evaluation on Health and Nutritional. Piacenza, Italy; 2001.
- Wang X, Ren H, Wang W, et al. Effects of Inoculation of Commercial Starter Cultures on the Quality and Histamine Accumulation in Fermented Sausages. J Food Sci. 2015;80(2):M377-M384
CrossRef - Galati A, Oguntoyinbo FA, Moschetti G, Crescimanno M, Settanni L. The Cereal Market and the Role of Fermentation in Cereal-Based Food Production in Africa. Food Rev Int.2014;30(4):317-337
CrossRef - Wood BJB. Microbiology of Fermented Foods. 2nd ed. Glagow, UK: Blackie Academic & Professional; 1998.
- Mignouna DB, Mutabazi K, Senkondo EM, Manyong VM. Adoption of a New Maize and Production Efficiency in Western Kenya. In: Joint 3rd African Association of Agricultural Economists (AAAE) and 48th Agricultural Economists Association of South Africa (AEASA) Conference. Cape town, South Africa; 2010.
- Ndwiga J, Pittchar J, Musyoka P, et al. Constraints and Opportunities of Maize Production in Western Kenya: A Baseline Assessment of Striga Extent, Severity, and Control Technologies. Ibadan, Nigeria; 2013.
- Official Methods of Analysis of AOAC International Method. 2000;996.23
- Kunyanga CN, Mbugua S, Kangethe E, Imungi J. Microbiological and Acidity Changes During the traditional production of kirario: an indigenous kenyan fermented porridge produced from green maize and millet. African J Food Agric Nutr Dev.2009;9(6):1419-1435
CrossRef - FDA. Yeasts, Molds and Mycotoxins. In: Bacteriological Analytical Manual. Center for Food Safety and Applied Nutrition; 2001.
- ISO 21528-2. Horizontal methods for the detection and enumeration of Enterobacteriaceae – Part 2: Colony-count method. In: Microbiology of Food and Animal Feeding Stuffs. Geneva, Switzerland: International Organization for Standardization; 2004.
- Mithun S, Dipak V, Sheela S. Isolation and Identification of lactobacilli from raw milk samples obtained from Aarey Milk Colony. Int J Sci Res Publ. 2015;5(1):1-5
- Oyedeji O, Ogunbanwo ST, Onilude AA. Predominant Lactic Acid Bacteria Involved in the Traditional Fermentation of Fufu and Ogi, Two Nigerian Fermented Food Products. Food Nutr Sci.2013;4(11):40-46
CrossRef - Akinleye OM, Fajolu IO, Fasure AK, Osanyinpeju OS, Aboderin AO, Salami OO. Evaluation of Microorganisms at Different Stages of Production of Ogi in Alimosho Community, Area Southwest, Lagos, Nigeria. Am J Res Commun. 2014;2(10):215-230
- Holt JH, Krieg NR, Sneath PH a., Staley JT, Williams ST. Bergey’s manual of determinative bacteriology ninth edition. Eur J Paediatr Neurol.1994;13(6):560
- Pyar H, Peh KK. Characterization And Identification Of LactobacillusAcidophilus Using Biolog Rapid Identification System. Int J Pharm Pharm Sci.2013;6(1):189-193
- Goyal R, Dhingra H, Bajpai P, Joshi N. Characterization of the Lactobacillus isolated from different curd samples. African J Biotechnol.2012;11(79):14448-14452
CrossRef - Kale PS. Isolation and identification of bacteria from curd and its application in probiotic chocolate. Eur J Exp Biol. 2014;4(6):95-97
- Reiner K. Carbohydrate Fermentation Protocol. Sterling, USA: John Wiley & Sons, Ltd; 2012.
- Bhardwaj A, Monica P, Sangu KPS, Kumar S, Dhewa T. Isolation and Biochemical Characterization of Lactobacillus species Isolated from Dahi. Res Rev A J Dairy Sci Technol. 2012;1(2):1-14
- Ramírez-chavarín norma l., Wacher-rodarte carmen, Pérez-chabela m. L. Characterization And Identification Of Thermotolerant Lactic Acid Bacteria Isolated From Cooked Sausages As Bioprotective Cultures. J Muscle Foods. 2010;21(3):585-596
CrossRef - Paéz R, Lavari L, Vinderola G, et al. Effect of heat treatment and spray drying on lactobacilli viability and resistance to simulated gastrointestinal digestion. Food Res Int.2012;48(2):748-754
CrossRef - Health Protection Agency. Guidelines for Assessing the Microbiological Safety of Ready-to-Eat Foods Placed on the Market. London, UK; 2009.
- Baylis C, Uyttendaele M, Joosten H, Davies A, Heinz HJ. The Enterobacteriaceae and Their Significance to the Food Industry. Brussels, Belgium: International Life Sciences Institute; 2011.
- Kirui MC, Alakonya AE, Talam KK, Tohru G, Bii CC. Total aflatoxin, fumonisin and deoxynivalenol contamination of busaa in Bomet county, Kenya. African J Biotechnol. 2014;13(26):2675-2678
CrossRef - Garnier L, Valence F, Mounier J. Diversity and Control of Spoilage Fungi in Dairy Products: An Update. Microorganisms.2017;5(3):42
CrossRef - Ogbonnaya N, Chidinma CB. Studies on Akamu, a traditional fermented maize food. Rev Chil Nutr. 2012;39(2):180-184
- Hoque MZ, Akter F, Hossain KM, Rahman MSM, Billah MM, Islam KMD. Isolation , Identification and Analysis of Probiotic Properties of Lactobacillus Spp. From Selective Regional Yoghurts. World J Dairy Food Sci.2010;5(1):39-46
- Ijabadeniyi AO. Microorganisms Associated with Ogi Traditionally Produced from Three Varieties of Maize. Res J Microbiol.2007;2(3):247-253
CrossRef - Adegbehingbe KT. Production of Masa Using Maize-Sorghum Blends. Int J Sci Res.2014;3(8):484-489
- Atter A, Obiri-Danso K, Amoa-Awua WK. Microbiological and chemical processes associated with the production of burukutu a traditional beer in Ghana. Int Food Res J.2014;21(5):1769-1776
- Chaves-L´opez C, Serio A, Grande-Tovar CD, Cuervo-Mulet R, Delgado-Ospina J, Paparella A. Traditional Fermented Foods and Beverages from a Microbiological and Nutritional Perspective: The Colombian Heritage. Compr Rev Food Sci Food Saf. 2014;13(5):1031-1048
CrossRef - Cousin FJ, Lynch SM, Harris HMB, et al. Detection and genomic characterization of motility in Lactobacillus curvatus: confirmation of motility in a species outside the Lactobacillus salivarius clade. Appl Environ Microbiol.2015;81(4):1297-1308
CrossRef - Cai Y, Ohmomo S, Ogawa M, Kumai S. Effect of NaCl-tolerant lactic acid bacteria and NaCl on the fermentation characteristics and aerobic stability of silage. J Appl Microbiol.1997;83:307-313
CrossRef - Abhijit Chowdhury, Nur Hossain, Nure Jannatul Mostazir, Fakruddin, Morsaline Billah, Monzur Morshed Ahmed. Screening of Lactobacillus spp. from Buffalo Yoghurt for Probiotic and Antibacterial Activity. J Bacteriol Parasitol. 2012;3(8):1-5
CrossRef - Halima Z-K, Nour-Eddine K. Statistical optimization of lactic acid production by Lactococcus lactis strain, using the central composite experimental design. African J Biotechnol.2014;13(45):4259-4267
CrossRef - Kostinek M, Specht I, Edward VA, et al. Diversity and technological properties of predominant lactic acid bacteria from fermented cassava used for the preparation of Gari, a traditional African food. Syst Appl Microbiol.2005;28(6):527-540
CrossRef - Hertzberger R, Arents J, Dekker HL, et al. H(2)O(2) production in species of the Lactobacillus acidophilus group: a central role for a novel NADH-dependent flavin reductase. Appl Environ Microbiol.2014;80(7):2229-2239
CrossRef - Hassan R, El-Kadi S, Sand M. Effect Of Some Organic Acids On Some Fungal Growth And Their Toxins Production. Int J Adv Biol. 2015;2(1):1-11

This work is licensed under a Creative Commons Attribution 4.0 International License.





