Introduction
Free radicals and Reactive Oxygen Species (ROS) has already known as the cause of oxidative cellular and molecular injuries that have been associated with initiation and development of non-communicable diseases (NCDs), including cancer.1,2 Many of studies have reported, that the intake of antioxidant-containing food materials has been correlated with a reduction in cancer and other NCDs through oxidative cellular prevention.3 Therefore, the rich of antioxidant containing in the diet were thought to be an alternative strategy to avoid NCDs.
Black Rice Bran (BRB) was known as one of the potential antioxidant sources.4 The antioxidative activity of BRB are mainly influenced by the composition of many phenolic acids (ferulic, gallic, p-coumaric, protocatechuic, caffeic, vanillic, dan syringic) and others bioactive components, such as anthocyanin, oryzanol, tocotrienol, and tocopherol.5-7 On the other hand, many BRB bioactive components were reported having the antiproliferative activity and invasion capability against cancer cells.8,9 However, to the best of our knowledge, there is novel study regarding the exploration of potentials of Indonesia BRB variety applications.
The main obstacles of BRB utilization is largely due to the low bioavailability of its bioactive components.10 The previous study revealed that 62% of BRB phenolic is insoluble forms and the aglycone phenolic bound to polysaccharides with polysaccharides.11 This makes the maximum biofunctional potentials of BRB was unreachable. According to other research, biological functionalities of compounds in BRB can be improved by using fermentation, particularly mold(s) fermentation.12 Mold(s) is a multicellular microorganism which very resistant to low water availability.13 Furthermore, the lignocellulolytic activity of molds was reported to be higher than lactic acid bacteria’s (LAB).14 Among the potential molds that can be used, Rhizopus oryzae and R. oligosporus have been long used in fermentation to produce various types of products, such as the production of tempe. These genera were highly resistant to cross contamination and high rate of digestible capability without the production of any toxic substances within controlled environment during cultivation period.15,16 Moreover, these species also have been known for their ability to produce the enzyme to be able to effectively increase chemical composition and bioactivity of white rice bran through deliberating the bioactive compounds from the cellular matrix.17 On this study, the metabolic synergy effects of those two kinds of molds in maximizing functional properties of BRB were evaluated.
Nowadays, colon cancer remains as the third highest of causes of mortality worldwide than others cancer.18 BRB bioavailability improvement through fermentation treatment are expected to decrease the prevalence of colon cancer incidence. In vitro assay, was considered to be one of the reliable scientifically approaches to evaluate the cytotoxic activity of BRB. Therefore, in the present study, the cytotoxic activity of FBRB extracts was evaluated against colon cancer cell, WiDr. The aim of this study was to improves the bioavailability of BRB bioactive components through fermentation using both R. orzyae and R. oligosporus as single and mix culture, as well as improves its antioxidative properties and cytotoxic activity against colon cancer cell, WiDr (in vitro).
Materials and Methods
Sample Preparation of Black Rice Bran
The black rice bran (hereafter BRB) used in this study were obtained from Indonesian varieties of Cempo Ireng in the form of dried unhulled rice. Rice bran samples in this study are derived from husking those dried unhulled rice grains. The milling process is performed using a Rice Machine-THU (Satake, Japan), so that the brown rice is obtained. Next, brown rice was milled using a Grain Testing Mill (Satake, Japan). Milled rice and the bran will be obtained from the process. The bran is then sieved again with a 20-mesh sieve to get rid of the chaff and groats. BRB filtrate obtained from the sieving process is ready to be fermented at a later stage.
Black Rice Bran Fermentation using R. oryzae and R. Oligosporus Cultures
The fermentation technique used is solid state fermentation (SSF). The process consists of 2 stages, namely stabilization of BRB substrate and starter inoculation on the substrate. The inoculum of R. oryzae and R. oligospurus were obtained from the culture collections of IPB Culture Collection (IPBCC) and Indonesian Culture Collection (InaCC), Indonesian Institute of Sciences (LIPI) Research Center. Preparation of fermentation sample began with the addition of water into the black rice bran substrate with water:substrate ratio = (1:2). Subsequently, the sample was stabilized at the temperature of 121 °C for 15 minutes. This process was followed by the inoculation of the mold/fungus after the sterile sample reached a temperature of 30 °C. The amount of mold culture suspension inoculated onto the substrate was 1.5 × 105 spore/g of the bran, both in single and mixed culture treatments (R. oryzae:R. oligosporus = (1:1)). Bran substrate that has been inoculated with starter were incubated for 72 and 96 hours. After incubation, the sample is lyophilized and then stored at -18 °C until they are analysed.
Preparation and Extraction of Fermented Black Rice Bran
The solvents used in the preparation of the extract are 70% ethanol and methanol. They are known to be universal and possess a broad polarity spectrum to extract potentially functional components in the fermented BRB (FBRB). In the beginning, bran substrate and solvents were prepared. A total of 10 g of FBRB were placed in a 250 mL erlenmeyer, then added with 100 mL solvent. After that, the erlenmeyer was covered with aluminum foil during the extraction process. The extraction technique used is maceration. This process were performed for 3 hours using the Innova 2300 Platform Shaker (New Brunswick Scientific, USA), then macerates were sonified using Branson 8510 Sonicator for 15 minutes. Subsequently, the extracts were filtered using Whatman Grade 1 filter paper then centrifuged at the speed of 3000 rpm for 10 minutes. Next, the filtrate were separated from the residue. The filtrate were then referred to as FBRB extract and were used in the subsequent analysis.
Total phenolic content analysis
Total phenolic content (TPC) was determined by microplate methods described by Bobo-Garcia et al.,19 Approximately 20 µL of samples were transferred into 96-well plate and then reacted with 100 µL of diluted Folin-Ciocalteu. The mixture was added with 75 µL Na2CO3 (100 gL-1) then homogenized and incubated for 2 hours at room temperature. The absorbance was measured by Epoch™ microplate spectrophotometer (BioTek®, USA) at 750 nm. Gallic Acid was used as a standard curve. TPC of samples were defined as Gallic Acid Equivalent (mg GAE) per g dry weight samples.
DPPH radical scavenging assay
The DPPH radical scavenging capacity assay was performed by 96 well plate methods adopted by Bobo-Garcia et al.,19 A 20 µL of each sample were transferred into 96 well plate then was reacted with 180 µL working solution DPPH. Then incubated for 40 min at the dark room temperature. The absorbance was measured by Epoch microplate spectrophotometer (BioTek, USA) at 515 nm. The percentage of DPPH radical scavenging was measured by the following equation:
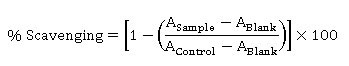
The DPPH radical scavenging capacity of samples was defined as Trolox Equivalent Antioxidant Capacity (mg TEAC per g dry weight samples).
Ferric reducing antioxidant power assay
The 96 well plate ferric reducing antioxidant power (FRAP) capacity determination method was adopted from Benzie and Strain FRAP method,20 with modification. The working FRAP solution was prepared by mixing acetic acid buffer (300 mM pH 3.6); 2,4,6-Tripyridyl-s-Triazine (TPTZ) (10 mM was diluted in 40 mM HCl); and FeCl3.6H2O (20 mM in distilled water); by the ratio of 10:1:1. Before being used, the FRAP working solution was conditioned at 37 oC for 10 min. Next, 10 µL of samples volume were transferred into 96 well plate for being analyzed. After being reacted, the analysis mixture was incubated at 37 oC for 30 min. The absorbance of the mixture was measured by Epoch™ microplate spectrophotometer (BioTek, USA) at 593 nm. The net value of absorbance of the samples and standard was measured by the following equation :
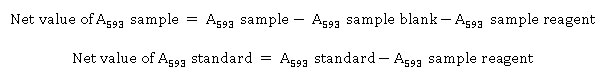
The metal complex and TPTZ reducing capacity of antioxidant samples were defined as Trolox Equivalent Antioxidant Capacity (mg TEAC per g dry weight sample).
Cytotoxic activity evaluation of BRB extracts using MTT assay
Cell growth inhibition assay was performed by colorimetric quantitative MTT (3- (4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide) assay described by Dapar et al.,21 with slight modification. The method consists of three main steps: seeding of the cells on 96-well flat-bottom microtiter plates, treatment of cells using an extract or sample of choice, and termination and absorbance reading on a multiwell scanning spectrophotometer or ELISA reader. Kidney epithellial cell from african green monkey (normal cell), Vero (ATCC CCL-81) and human colon cancer cell, WiDr (ATCC CCL-218) were obtained from PSSP-IPB culture collection.
Firstly, the NRBRB and FBRB were dissolved in phosphate-buffered saline (PBS), and the concentration was adjusted to IC50 of Vero cell baseline. The cell lines were cultured for 24 h in a 96-well plate. After adherence of the cells within 24 h of incubation at 37 oC, the cell line was treated with 100 µL aliquots of the NFBRB and FBRB extracts solution. The treated plates were then incubated for 72 h at 37 oC in a humidified incubator with 5% CO2. After incubation, 10 µL of MTT (dissolved in PBS at 5 mg/mL) was added to all the wells. After four hours of incubation at 37 °C and 5% CO2, 100 µL of 10% SDS in HCl 0.1 N was added to each well as a stopper. The absorbance was measured at 595 nm using iMark™ microplate absorbance reader (Bio-Rad, UK). Absorbance was proportional to the number of cells. The concentrations causing 50% inhibition of cell viability (IC50) were calculated.
Statistical Analysis
The experimental design used in this study is Completely Randomized Design (CRD). Research design consists of one factor (variation of microbial type) and three treatment levels (R. oryzae, R. oligosporus, and the mixture of both, respectively). The reported data is the mean ± SD for three replications of each sample (n=3). The differences between group means were evaluated by analysis of variance (ANOVA) were done by using SPSS 22.0 software (SPSS, Chicago, IL, USA) followed by the Duncan’s post hoc test (p<0.05).
Results and Discussion
Total Phenolic Content of Black Rice Bran Extract
Result of analysis indicated that there are significant difference between TPC of BRB extracted with methanol and 70% ethanol (Figure 1), where ethanolic fraction has relatively higher TPC compared to the methanolic fraction. The higher value is indicated by bioactive compounds distributed in the BRB that is dominated by phenolic components, mostly of a polar molecule. The number of extracted components is influenced by several factors including the type of solvent, its volume, temperature, and extraction period.22 Determining type of solvents becomes essential as the presence of components and their activity are affected by their solubility in solvents.23 Additionally, solvent polarity is also deemed to affect the ability of 70% ethanol solvent in extracting bioactive compounds of BRB. 70% ethanol solvent is known to have higher polarity than methanol with polarity index of 6.1 and 5.1, respectively.24 Higher polarity will increase the ability of 70% ethanol in extracting phenolic compounds compared to methanol. This assumption is further supported by Devi and Arumughan25 who revealed that generally the amount of extracted substance will increase as the polarity increases, and vice versa. In addition to that, Jun et al.,23 reported that the use of water-mixtured solvent with methanol or water with ethanol would be more efficient than absolute solvent. Thus, 70% ethanol solvent is more efficient in the extracting of phenolic components contained in BRB than methanol.
Research data also points out a significant difference (p<0.05) between NFBRB and FBRB treatment. TPC of FBRB extract has higher value than NFBRB extract. Fermentation process enhances the availability of free phenolic acids, making the amount of TPC in FBRB extract higher than NFBRB extract. An increase in the amount of free phenolic compounds allegedly occured due to microbial extracellular enzymatic hydrolytic activity during fermentation. One of the enzymes taking major role is the β-glucosidase produced by the mold.15 The β-glucosidase enzyme will act as a catalyst in hydrolysis process of the glycoside bond on the alkyl and aryl β-D-glucoside groups. In other words, the enzyme will hydrolyze the phenolic glycosides thus releasing them into free phenolic aglicons, which can have a high antioxidant activity.26
In accordance with the result from other studies, it is well known that the antioxidant activity of phenols is affected by their chemical structure, and this activity can be decreased or increased depending upon the functional group attached to a basic aglycon. The presence of glycosides attached to flavonoid aglycons, such as a flavonol or anthocyanidin, decreases the antioxidative activity of the flavonoid. In this case, the glucoside moiety interferes with the co-planarity of the flavonoid molecule, decreasing its ability to delocalize electrons and thereby decreasing the antioxidative activity of the flavonoid.27
 |
Figure 1: TPC of non-fermented black rice bran (NFBRB) extract and after fermented with R. oryzae (ORY), R. oligosporus (OLI), and mixtures (MIX) during 72 and 96-hour treatment of 2 solvent fractions: (a) Methanol and (b) 70% ethanol. The value in each bar with different letters represent a significantly difference by Duncan test (p<0.05) Click here to View figure |
The highest amount of TPC is found in MIX 96 encoded samples, both in methanolic and ethanolic fractions. Highest TPC value in MIX 96 samples indicates that 96-hour fermentation time is the most sufficient cultivation period to produce a maximum TPC. The 96-hour fermentation has been noted as the best cultivation period to produce the highest level of phenolic compounds, as no significant increase would occur afterwards.17 In addition, the high value is suspected to happen due to synergistic effect of culture combinations during fermentation. According to Holker et al.,28 a combination of cultures will produce enzymes with a wider spectrum of degradation. The multienzyme system causes a higher level of damage to the cell wall components of the BRB, resulting in larger amount of free phenolic acid compounds. Wong29 stated that the combination and synergism of some enzymes are needed to degrade one of the components of the cell wall structure.
DPPH radical scavenging Antioxidant Analysis of the Black Rice Bran Extract
In this study, FBRB antioxidant capacity is evaluated using DPPH method, where NFBRB is set to be a control group. Our data indicate that a significant difference (p<0.05) between DPPH radical scavenging score by NFBRB and FBRB extracts, where FBRB extract has higher activity tendency than the control group (Figure 2). Component degradation of the cell wall structure during fermentation is proved to be able to set phenolic bound free and enhance biological availability of phenolic components. Higher antioxidant capacity in FBRB sample is deemed to be a consequence of the increasing availability of several free phenolic components such as ferulic acid, anthocyanins, and oryzanol. According to Jun et al.,7; Razak et al.,15, phenolic components such as ferulic acid (the main phenolic compound in BRB) have been reported to act as radical scavengers against DPPH.
The result also showed that type of solvent also determines the effectiveness of DPPH radical scavenging activity. DPPH radical scavenging capacity of 70% ethanolic extract is significantly higher (p<0.05) than methanolic extract (Figure 2). It indicates that each solvent has a different effectiveness in recovering bioactive compounds in a material. Bhebhe et al.,29 showed that 30% of water (v/v) in ethanol solvents could increase water accessibility to penetrate into solid matrix of a material, and as a consequence, bioactive components have higher recovery rate, enabling the sample to have higher antioxidant activity as well.
 |
Figure 2: DPPH free radical scavenging capacity of non-fermented black rice bran (NFBRB) extract and after fermented with R. oryzae (ORY), R. oligosporus (OLI), and mixtures (MIX) during 72 and 96-hour treatment of 2 solvent fractions: (a) Methanol and (b) 70% ethanol. The value in each bar with different letters represent a significantly difference by Duncan test (p < 0.05) Click here to View figure |
Based on Figure 2, the highest DPPH radical scavenging capacity is found in 96 MIX sample for methanolic extract and ORY 72 for 70% ethanolic extract. The result showed R. oryzae’s potentials as a single culture to produce antioxidant phenolic compounds. Even though DPPH radical scavenging capacity of ORY 72 is the highest on ethanol fraction, the value does not show any significant difference (p>0.05) to the mixed culture extract (MIX 96) of ethanol fraction. It indicates that co-cultivation treatment of R. oryzae and R. oligosporus served as an effective way to obtain antioxidant phenolic compounds with the highest DPPH radical scavenging capacity.
This is in accordance with Razak et al.,’s16 findings, highlighting that the use of R. oryzae as fermentation starter could increase bran’s antioxidant activity (based on FRAP and DPPH assay). A number of phenolic compounds such as sinapic acid, vanillic acid, caffeic acid, syringic acid, protocatechuic acid, and 4-hydroxybenzoate are also reported to increase after the fermentation treatment. Based on the study it is also known that the highest growth occurred in fermented bran with mixed cultures (A. oryzae + R. oryzae), followed by A. oryzae single fermented culture, and R. oryzae respectively. The advantages of using mixed culture as starter had also been highlighted by Razak et al.,15 in which co-cultivation treatment (R. oligosporus + M. purpureus) was able to increase antioxidant capacity higher than single culture treatment. The combination of cultures during cultivation is thought to create a wider degradation spectrum on the lignocellulosic matrix through the involvement of a synergically hydrolytic enzyme system, causing an increase in free phenolics, leading to an increased antioxidant bioactivity.
Ferric Reducing Antioxidant Power (FRAP) Antioxidant Analysis of BRB Extract
In this study, antioxidant capacity of the extracts in reducing radicals is also determined by the FRAP assay. The result of FRAP analysis showed similar pattern to DPPH radical scavenging capacity analysis where antioxidant activity of methanolic extract of FBRB and 70% ethanolic extract increased significantly (p <0.05) compared to control group (Figure 3). It also illustrates the existence of antioxidant compounds on FBRB that are able to reduce free radicals through different mechanisms. The increase is allegedly caused by an increased availability of biological compounds with functional antioxidant properties, especially phenolic compounds.
 |
Figure 3: FRAP antioxidant reduction capacity of non-fermented BRB (NFBRB) extract and fermented with R. oryzae (ORY), R. oligosporus (OLI), and mixtures (MIX), respectively, during 72 and 96-hour treatment of 2 solvent fractions: (a) Methanol and (b) 70% Ethanol. The value in each bar with different letters represent a significantly difference by Duncan test (p < 0.05) Click here to View figure |
FRAP assay results also showed a difference in antioxidant effectiveness between the two extracts (70% ethanol and methanol). The antioxidant capacity of ethanolic extract tends to be higher than that of methanolic extract (Figure 3). It allegedly occurs due to differences in extracted components. Different bioactive components will result in different antioxidant activity and functional properties. Craft et al.,30 and Schmidt et al.,17 revealed that the amount of antioxidant capacity is highly dependent on chemical structure of the compound, number of hydroxyl groups, and its polarity.
In contrast to DPPH radical scavenging capacity assay, the FRAP assay describes the mechanism antioxidant actions in reducing radicals through single electron donations. The antioxidant compounds are reported to be able to prevent oxidation reaction at the initiation stage. Furthermore, this type of compound is known as a secondary antioxidant. It acts to inhibit oxidation reactions indirectly through oxygen capture mechanism and pro-oxidant binding.31
FRAP assay results also showed the ability of FBRB antioxidant compounds to act as a radical reducer apart from its role as radical damper. In other words, the bioactive compounds in FBRB extract are not only dominated by the primary antioxidant group but also the secondary antioxidants. The highest antioxidant capacity was found in the ORY 72 sample for methanolic fraction and MIX 96 for ethanolic fraction. Additionally, the results also showed that fermentation treatment using R. oryzae as a single culture with 72-hour incubation period is also potential and efficient in producing compounds with functional antioxidant properties. It is in line with Massarolo et al.,’s32 findings who reported that fermentation period using R. oryzae for 72 hours was the best cultivation time that could produce the highest antioxidant activity, however the activities in the 72-hour cultivation period do not differ significantly with that of 96-hour ones.
Antiproliferation Activities of the Black Rice Bran Extract against WiDr Colon Cancer Cells
In this study, the experiment samples used to further analyse WiDr colon cancer cells’ cytotoxic activity are samples with best quality based on TPC, DPPH, and FRAP analysis, which is the FBRB extract with 96-hour incubation period. Cytotoxic activity of FBRB extract is indicated by IC50 value. This amount of concentration could inhibit 50% of WiDr colon cancer cell growth. Lower IC50 value indicates a more effective extract ability to reduce WiDr colon cancer cell proliferation.
Table 1: IC50 value of methanolic and 70% ethanolic extracts of black rice bran*
| Type of sample | Treatment | IC50 value of black rice bran extract (µg.mL-1) | |
| Vero Cell (normal cell) | WiDr Cell | ||
| Methanolic extract of BRB | NFBRB | 1564.86 ± 146.45ab | 461.50 ± 59.09c |
| ORY 96 | 1694.27 ± 58.56bc | 163.52 ± 37.33a | |
| OLI 96 | 1509.81 ± 132.99a | 343.41 ± 41.42b | |
| MIX 96 | 1718.90 ± 38.37c | 481.73 ± 44.79c | |
| 70% ethanolic extract of BRB | NFBRB | 728.55 ± 108.15a | 425.06 ± 89.49b |
| ORY 96 | 740.75 ± 57.88a | 203.70 ± 83.43a | |
| OLI 96 | 785.39 ± 91.03a | 237.50 ± 67.00a | |
| MIX 96 | 796.89 ± 104.33a | 284.57 ± 11.38a | |
*Non-fermented black rice bran (NFBRB) extract and BRB extract after fermented with R. oryzae (ORY), R. oligosporus (OLI), and mixtures (MIX) during 72 and 96-hour treatment. Value are expressed as means ± sd. The value in each column with different superscript represent a significantly difference by Duncan test (p < 0.05).
Results of the analysis showed that cytotoxic activity of BRB extract on vero cells is considerably lower compared to those on WiDr cells (IC50 extract on vero cells > WiDr cells) (Table 1). It indicates that the BRB, both of methanolic and ethanolic fractions play a more active role in WiDr cancer cells compared to vero cells, so NFBRB or FBRB extracts could be classified safe toward normal human cells. Atjanasuppat et al.,33 stated that the index of activity of crude extract from natural ingredients could be categorized based on IC50 value, which are classified into active (≤20 μg.mL-1), moderate (>20-100 μg.mL-1), weak (>100-1000 μg.mL-1), and inactive (>1000 μg.mL-1). Thus, BRB extracts either of ethanolic or methanolic fractions potentially prevent colon carcinogenesis, although it has a weak cytotoxic activity againsts WiDr colon cancer cells. In addition to that, research findings also showed significantly different cytotoxic activities (p<0.05) between FBRB and NFBRB samples where fermentation treatment could increase cytotoxic activity higher than control treatment.
The present study also demonstrated that the sample of ORY 96 was the sample with highest cytotoxic activity in inhibiting cancer colon, WiDr growth, followed by OLI 96, MIX 96, and NFBRB for both methanolic or ethanolic extracts. The high of antiproliferative activity of ORY 96 was be assumed because of oryzanol profile content improvement. Massarolo et al.,32 conveyed that R. oryzae fermented bran will experience an increase of 2,4-cycloartanil ferulate content during cultivation. This component has been reported to have contributed to the prevention of cancer risk. Furthermore, Rashid et al.,’s34 mentioning that the bioactivity of fermented bran is strongly related to the contribution of all bioactive components in it and is not only specific to the phenolic components. Other components such as unsaturated fatty acids,32 amino acids, and peptides27 formed during the fermentation process are also reported to have contributed to the bioactivity of rice bran extract.
The increase in such activity is thought to be caused by changes in the bioactive components during fermentation process. Several in vitro studies have proved that the increased availability of bioactive compound of the bran during fermentation’s metabolic conversion process could increase its anticancer activity. Ryan et al.,10 also underlined that during fermentation process with S. cerevisiae var. boulardii, the bran will experience an increase in ferulic acid content affecting on its increased cytotoxic activity againsts lymphoma B cells. Other studies presented by Yoon et al.,35 stated that BRB’s cytotoxic activity that is fermented with B. subtilis significantly increased againsts AGS gastric cancer cells, HT-29 colon cancer cells, and MCF-7 breast cancer cells compared to the non-fermented bran. Fermentation process is alleged to degrade the polymerization of phenolic compounds, forming a new substance with an anticancer activity.
The involvement of phenolic compound antioxidants such as ferulic acid, p-coumaric acid, caffeic acid, and anthocyanins in inhibiting cancer cells has been widely reported. Rosa et al.,36 argued that ferulic acid, p-coumaric acid, caffeic acid could inhibit proliferation and superoxide production of HT29-D4, HCT-15, and HT-29 cancer colon cells. Additionally, the phenolic compounds are also reported to cause apoptosis in cancer cells through oxidant mediation mechanisms.
The combination of R. oryzae and R. oligosporus cultures and 96-hour incubation time (MIX 96) during fermentation are proved to be more effective than single culture in maximizing the availability of phenolic antioxidant compounds of BRB which acts as a reductant/reducing agent of free radical compounds. The potential use of R. oryzae as a single culture is indicated by the high cytotoxic activity in WiDr colon cancer cells compared to other treatment. Therefore, fermentation could increase the activity of functional properties of antioxidant and cytotoxic derived from BRB extract in WiDr colon cancer cells.
Acknowledgements
This work was supported by a grant from Directorate of Higher Education, Ministry of Research, Technology and Higher Education of Indonesia.
Conflict of interest
The authors have declared no conflict of interest associated with this publication.
References
- Chi HY, Lee CH, Kim KH, Kim SL, Chung IM. Analysis of Phenolic Compounds and Antioxidant Activity with H4IIE Cells of Three Different Rice Grain Varieties. Eur Food Res Technol; 225(5-6): 887-893: (2007)
CrossRef - Goufo P, Trindade H. Rice antioxidants: phenolic acids, flavonoids, anthocyanins, proanthocyanidins, tocopherols, tocotrienols, γ-oryzanol, and phytic acid. Food Sci Nutr; 2(2): 75-104: (2014)
CrossRef - Butsat S, Siriamornpun S. Antioxidant Capacities and Phenolic Compounds of The Husk, Bran and Endosperm of Thai Rice. Food Chem; 119(2): 606-613: (2010)
CrossRef - Muntana N, Prasong S. Study on Total Phenolic Contents and Their Antioxidant Activities Of Thai White, Red, and Black Rice Bran Extracts. Pak J Biol Sci; 13(4): 170-174: (2010)
CrossRef - Kong S, Lee J. Antioxidant in Milling Fractions of Black Rice Cultivars. Food Chem; 120(1): 278-281: (2010)
CrossRef - Zhang MW, Zhang RF, Zhang FX, Liu RH. Phenolic Profiles and Antioxidant Activity of Black Rice Bran of Different Commercially Available Varieties. J Agric Food Chem; 58: 7580-7587: (2010)
CrossRef - Jun HI, Shin JW, Song GS, Kim YS. Isolation and Identification of Phenolic Antioxidants in Black Rice Bran. J Food Sci; 80(2): C262-C268: (2015)
CrossRef - Forster GM, Raina K, Kumar A, Kumar S, Agarwal R, Chen M-H, Bauer JE, McClung AM, Ryan EP. Rice Varietal Differences in Bioactive Bran Components for Inhibition of Colorectal Cancer Cell Growth. Food Chem; 141(2): 1545-1552: (2013)
- Suttiarporn P, Chumpolsri W, Mahatheeranont S, Luangkamin S, Teepsawang S, Leardkamolkarn. Structures of Phytosterols and Triterpenoids with Potential Anti-Cancer Activity in Bran of Black Non-Glutinous Rice. Nutrients; 7(3): 1672-1678: (2015)
CrossRef - Ryan EP, Heuberger AL, Weir TL, Barnett B, Broeckling CD, Prenni J. Rice Bran Fermented with Saccharomyces boulardii Generates Novel Metabolite Profile with Bioactivity. J Agric Food Chem; 59: 1862-1870: (2011)
CrossRef - Adom KK, Liu RH.. Antioxidant Activity of Grains. J Agric Food Chem; 50(21): 6182-6187: (2002)
CrossRef - Oliveira MD, Cipolatti EP, Furlong EB, Soares LDS. Phenolic Compounds and Antioxidant Activity in Fermented Rice (Oryza sativa) Bran. Cienc Technol Aliment Campinas; 32(3): 531-537: (2012)
CrossRef - Raghavarao KSMS, Ranganathan TV, Karanth NG. Some Engineering Aspects of Solid-State Fermentation. Biochem Eng J; 13(2-3): 127-135: (2003)
CrossRef - Setiarto RHB, Saskiawan I. Seleksi dan Karakterisasi Mikroba Lignoselulolitik yang Diisolasi dari Limbahgergaji sebagai Media Tanam Jamur Tiram (Pleurotus ostreatus). Berita Biol; 12(1): 19-27: (2013)
- Razak DLA, Rashid NYAR, Jamaluddin A, Sharifudin SA, Long K. Enhancement of Phenolic Acid Content and Antioxidant Activity of Rice Bran Fermented with Rhizopus oligosporus and Monascus purpureus. Biocatal Agric Biotechnol; 4: 33-38: (2015)
- Razak DLA, Rashid NYA, Jamaluddin A, Sharifudin SA, Long K. Cosmeceutical potensials and bioactive compounds of rice bran fermented with single and mix cilture of Aspergillus oryzae and Rhizopus oryzae. J Saudi Soc For Agric Sci; 16(2): 127-134: (2017)
- Schmidt CG, Goncalves LM, Prietto L, Hackbart HS, Furlong EB. Antioxidant Activity and Enzyme Inhibition of Phenolic Acids from Fermented Rice Bran with Fungus Rizhopus oryzae. Food Chem; 146: 371-377: (2014)
- [WHO] World Health Organization. Cancer. Available at: http://www.who.int/mediacentre/factsheets/fs297/en/#: (2015)
- Bobo-Garcia G, Daidov-Pardo G, Arroqui C, Virseda P, Marin-Arroyo MR, Navarro M. Intra-Laboratory Validation of Microplate Methods for Total Phenolic Content and Antioxidant Activity on Polyphenolc Extracts, and Comparison with Conventional Spectrophotometric Methods. J Sci Food Agric; 95(1): 204-209: (2015)
- Benzie I, Strain J. The Ferric Reducing Ability of Plasma (FRAP) as a Meausure of “Antioxidant Power”: the FRAP Assay. Anal Biochem; 239(292): 70-76: (1996)
- Dapar MLG, Garzon JF, Demayo CG. Cytotoxic Activity and Antioxidant Potentials of Hexane and Methanol Extracts of IR-64 Rice Bran against Human Lung (A549) and Colon (HCT116) Carcinomas. Int Res J Biological Sci; 2(5): 19-23: (2013)
- Pedro AC, Granato D, Rosso ND. Extraction of Anthocyanins and Polyphenols from Black Rice (Oryza sativa) by Modeling and Assesing Their Reversibility and Stability. Food Chem; 191: 12-20: (2016)
- Jun HI, Song GS, Yang EI, Youn Y, Kim YS. Antioxidant Activities and Phenolic Compounds of Pigmented Rice Bran Extracts. J Food Sci; 77(7): 1-6: (2012)
- Snyder L. Classification of The Solvent Properties of Common Liquids. J Chromatogr Sci; 16(6): 223-234: (1978)
- Devi RR, Arumughan C. Phytochemical Characterization of Defatted Rice Bran and Optimization of a Process for Their Extraction and Enrichment. Bioresour Technol; 98(16): 3037-3043: (2007)
- Martins S, Mussatto SI, Martinez-Avila G, Montanez-Saenz. Bioactive Phenolic Compounds: Production and Extraction by Solid-State Fermentation. a Review. Biotechnol Adv; 29(3): 365-373: (2011)
- Hur SJ, Lee SY, Kim Y, Choi I, Kim G. Effect of Fermentation on The Antioxidant Activity in Plant-Based Foods. Food Chem; 160: 346-356: (2014)
- Holker U, Hofer M, Lenz J. Biotechnological Advantages of Laboratory-Scale Solid-State Fermentation with F Appl Microbiol Biotechnol; 64(2): 175-186: (2004)
- Bhebhe M, Fuller T, Chipurura B, Muchuweti M. Effect of Solvent Type on Total Phenolic Content and Free Radical Scavenging Activity of Black Tea and Herbal Infusions. Food Anal Methods; 9(4): 1060-1067: (2016)
- Craft BC, Kerrihard AL, Amarowicz R, Pegg RB. Phenol-Based Antioxidants and The In Vitro Methods used for Their Assessment. Compr Rev Food Sci Food Saf; 11: 148-173: (2012)
- Gupta D. Methods for Determination of Antioxidant Capacity: a Review. IJPSR; 6(2): 546-566: (2015)
- Massarolo KC, Souza TD, Collazzo CC, Furlong EB. The Impact of Rhizopus oryzae Cultivation on Rice Bran : Gamma-Oryzanol Recovery and Its Antioxidant Properties. Food Chem; 228: 43-49: (2017)
- Atjanasuppat K, Wongkham W, Meepowpan P, Kittakoop P, Sobhon P, Bartlett A, Whitfield P. In vitro Screening for Anthelmintic and Antitumour Activity of Ethnomedicinal Plants from Thailand. J Ethnopharmacol; 123: 475-482: (2009)
- Rashid NYA, Razak DLA, Jamaluddin A, Sharifuddin SA, Long Kamariah. Bioactive Compounds and Antioxidant Activity of Rice Bran Fermented with Lactic Acid Bacteria. MJM; 11(2): 156-162: (2015)
- Yoon HJ, Lee JH, Jin HJ, Kim HJ, Kim KT, Paik HD. Effect of Fermentation by Bacillus subtilis on Antioxidant and Cytotoxic Activities of Black Rice Bran. Int J Food Sci Technol; 50(3): 612-618: (2015)
- Rosa LS, Silva NJA, Soares NCP, Monteiro MC, Teodoro AJ. Anticancer Properties of Phenolic Acids in Colon Cancer – a Review. J Nutr Food Sci; 6(2): 1-7: (2016).

This work is licensed under a Creative Commons Attribution 4.0 International License.





