Introduction
The lipoprotein lipase (LPL) gene, located on chromosome 8p22, plays an important role in the hydrolysis of chylomicrons and VLDL leading to the enhancement and maturation of HDL. (Sprecher et al., 1996; Goldberg et al., 1996). Deficiency of lipoprotein lipase is associated with low HDL-C levels in both homozygous and heterozygous states (Kaser et al. 2003; Klos et al. 2007). It is also known to play a key role in lipid metabolism associated with obesity, dyslipidemias, hypertension (HTN) and type 2 diabetes (T2D; Munoz-Barriosa et al., 2012). Lipoprotein lipase is the critical enzyme responsible for the hydrolysis of triacylglycerol-rich lipoproteins (TG-RL) in peripheral circulation (Goldberg & Merkel, 2001) and in metabolic syndrome, defined by impaired conditions that include obesity, insulin resistance, dyslipidemia (including elevated triacylglycerol levels), and hypertension (Eckel, Grundy, & Zimmet, 2005). In addition, cells such as cardiac and skeletal myocytes, macrophages, and adipocytes synthesize and secrete LPL which is transported across the endothelial cells to its site of action on the lumen of the endothelium. Obesity, metabolic syndrome and T2D are the most common conditions associated with hypertriglyceridemia, and are often the result of increased plasma concentrations of the very low density lipoproteins (VLDL), free fatty acids, and/or decreased LPL (Eckel et al., 2005; Yuan et al., 2007), which also accounts for insulin resistance.
Although many studies have examined the gene expression and regulation of LPL in obesity and diabetes, few studies have investigated the role of dietary fat in relation to the expression and activity of the enzyme. Impairment and inhibition of the activity or function of LPL can lead to hypertriglyceridemia, a condition known to be a major risk factor for cardiovascular disease (Goldberg & Merkel, 2001; Merkel, Eckel, & Goldberg, 2002). Fatty acids are a major component of cell membranes and serve as long-term energy storage in the body. Some fatty acids also act as cell signaling molecules and hormone precursors. Each of the three common variants of the LPL gene (S477X, Pvu II, and Hind III) mildly affect LPL activity, but are all associated with increased TG and decreased HDL levels in response to high-fat diets. The mechanism by which saturated and unsaturated fatty acids affect LPL activity is also not well known. In this study, a known LPL raising agent was used to compare the effects of dietary fatty acids on adipose tissue LPL, plasma glucose, triacylglycerol, HDL-C concentration and IRS as a means of reducing the risk for developing heart disease.
Methods
Animals and Diet. Ninety day old NIH strain of the spontaneously hypertensive stroke- prone (SHRSP) male rats (average systolic BP ~160 mmHg) weighing ∼3250 g, were used in this study. Animals were housed in suspended stainless steel cages (40 × 24 × 18 cm), in a temperature-controlled room (20–23°C) with a 12-h light/dark cycle. All rats were fed a pelleted commercial nonpurified diet (Purina Rodent Chow, #500; TMI Nutrition, St. Louis, MO). They were then randomly assigned to 3 groups (8/group), to which either triolein (TO; ∼92% oleic acid, maximum peroxide value 0.84 meq/kg), or tripalmitin (TFA; ∼98% palmitic acid, maximum peroxide value 0.48 meq/kg) was fed by gavage (5 g/kg b. wt), and Control (CON; no additional fat). The animals were fed Chow and force-fed either triolein (TO) or Trans-fat (TFA) daily by gavage (5 mg/kg b. wt.), for 7 days. On days 5, 6 and 7, the animals also received NO-1886 (25 mg/kg b. wt.). Animals were sacrificed 4 hours after their last dosage on day 7. NO-1886 a gift from Dr. Kazuhiko Tsutsumi of Otsuka Pharmaceutical Factory, Japan NO-1886 is a novel drug that up-regulates LPL at the level of transcription. On day 7 all animals were anesthetized (50 mg/Kg Nembutal) and injected via the tail vein with 100 IU heparin/kg b. wt.) and sacrificed 15 minutes later. Blood and tissues were collected for outcome analyses. Protocols for animal use were approved by WSU Institutional Animal Investigation Committee and the study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals (NRC of the National Academies, 2011).
Measurement of Plasma Lipids. Total cholesterol (TC), triacylglycerol (TG) in serum in addition to individual lipoprotein fractions, including HDL (d = 1.059–1.23 kg/L) were determined using enzymatic reagents (TC and TG; DMA, Arlington, TX; PL, Wako Pure Chemicals, Richmond, VA) as described previously (Kudchodkar, Lacko, Dory, & Fungwe, 2000).
Measurement of Hepatic apoB100. The induction of hepatic apoB100 in SHRSP rats fed triolein or trans-fat with/without N0-1886 were measured. Total RNA was isolated from the liver and ApoB100 mRNA was determined by RT-PCR using sense and antisense specific primers.
Glucose Tolerance Test. Glucose tolerance test was performed by challenging a separate group of animals (CON and TO fed) with 25mg glucose per g of body weight. Blood glucose was measured at 30mins intervals using a kit from sigma (Kako, Masse, Huang, Tall, & Goldberg, 2002).
Measurement of insulin. Blood insulin was determined with an Insulin Radioimmunology kit (Beijing Institute of Atomic Research, Beijing, China). (Yin, Tsutsumi, Yuan, & Yang, 2002). Lipoprotein lipase activity was measured in heparinized blood with a lipid emulsion containing [3H] Triolein. (Kako et al., 2002). Relative insulin resistance was measured using the Measurement of HOMA-IR Index. Insulin resistance was calculated by the HOMA-IR-formula: [fasting insulin (mU/ml). fasting glucose (mmol/l)]/22.5 (Matthews et al. 1985).
Statistical Analysis. All results were subjected to statistical analysis using SPSS statistical package version 17.0 (IBM Corporation, Armonk, NY, USA).
Results
Plasma Lipids. The results in Figure 1 show the effect of fatty acids and NO-1886 on plasma TG concentration. The TFA fed group maintained TG levels similar to the CON group not fed fatty acids. TG levels on the other hand, were significantly higher in animals fed with TO (p<0.05). All the dietary groups receiving NO-1886 exhibited reduced plasma TG concentrations (p<0.05). HDL-C levels were not significantly affected by fat type (Figure 2). However, administration of NO-1886 dramatically increased HDL-C concentrations in the control and TO fed groups (p<0.05), but not with the TFA group (p>0.05) suggesting that as TG levels decline HDL-C levels tend to increase. As shown in Figure 3, the ratio of TG: HDL-C considered a risk indicator for CVD, was significantly reduced in all groups following administration of NO-1886 which stimulated LPL expression. Elevating the activity of LPL reduces the risk of reduce HDL-C regardless of plasma TG levels.
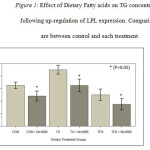 |
Figure 1: Effect of Dietary Fatty acids on TG concentrations following up-regulation of LPL expression. |
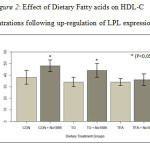 |
Figure 2: Effect of Dietary Fatty acids on HDL C concentrations following up-regulation of LPL |
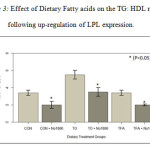 |
Figure 3: Effect of Dietary Fatty acids on the TG: HDL ratio following up-regulation of LPL expression. |
Insulin levels. Mean Insulin levels after 7 days of feeding and 3 days following administration of NO-1886 are summarized in Figure 4. Insulin levels were higher in the CON animals and declined by 25% with the LPL raising drug. Feeding TO reduced Insulin levels by 50% of Con and increasing LPL activity had no additional effect. Feeding TFA reduced Insulin by 25% of Con and stimulating LPL reduced it further down to 50% of Con. Triolein (95% oleic acid) had the most potent effect on lowering insulin levels. These data suggest that fatty acids can singly or in combination with other therapy be hypoinsulemic.
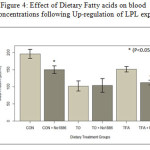 |
Figure 4: Effect of Dietary Fatty acids on blood insulin concentrations following Up-regulation of LPL expression. |
Postprandial glucose concentration. Figure 5 shows significant reduction in postprandial glucose levels in Control fed SHRSP (Con), TO fed SHRSP, TO+NO-1886, TFA fed SHRSP, and TFA+NO-1886 fed rats (p<0.05). Glucose concentrations were reduced by 10-15% after 7 days of fat feeding or 3 days of drug administration. The HOMA indices (insulin resistance) obtained were: Con= 66+4, Con+ NO1886=46+2, TO=36+8, TO+NO-1886= 31+7, TFA= 64+15, and TFA + NO-1886= 39+4. TO was effective in lowering insulin resistance (IR), but not different from use of the drug NO-1886. TFA had no effect. Stimulation of LPL lowered IR index and was most effective in combination with TO. These results show that in conditions of insulin resistance with hypertension, fat feeding not only decrease insulin levels as shown earlier, but also reduce plasma glucose levels as well. However, the effect of NO-1886 was marginal, suggesting that the effect of LPL on plasma glucose concentration may depend on the type of fat.
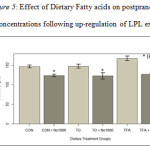 |
Figure 5: Effect of Dietary Fatty acids on postprandial glucose concentrations following up-regulation of LPL expression. |
Adipose Tissue Lipoprotein Lipase. Figure 6 shows LPL activity in adipose tissue from Sprague Dawley (SD, Wild Type) and SHRSP rat. In this experiment we used the SD rat (Wild Type) to compare the expression of LPL in the SHRSP rat. LPL activity in the SHRSP rats was 50% less than in the SD rat (p<0.05). Con+NO-1866 group had LPL activity 4-fold higher. TO or TFA raised LPL activity to the same levels as seen with the SD rats and 2-fold higher than Con rats. NO-1886 in combination with TO or TFA dramatically raised LPL activity 2-10 fold higher. These results show that fat feeding raises LPL activity and can be more potent when combined with other forms of therapy for insulin resistance. Specific LPL activities were 50% less in the SHRSP rats compared to a normal SD rat. Con rats fed NO-1886 had LPL specific activities increased by 3-fold. In TO or TFA fed rats, LPL specific activities were raised to the same level or slightly higher than for SD rats, but were 2-fold higher than Con rats.
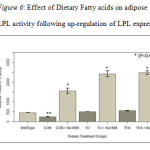 |
Figure 6: Effect of Dietary Fatty acids on adipose tissue LPL activity following up-regulation of LPL expression. |
Expression of Hepatic apoB100. As shown in Figure 7, dietary fatty acids stimulate, while induction of LPL simultaneously down regulate hepatic apoB mRNA levels depending on the fatty acid species. Both TO and TFA stimulated induction of apoB100. Administration of NO-1886 down regulated apoB100 in Controls, and Trans fat, but not in the TO fed animals. The data suggest that these fatty acids have similar regulatory effects on apoB100, but differ in their response when LPL is stimulated.
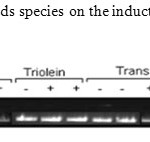 |
Figure 7: Effect of Dietary fatty acids species on the induction of LPL and hepatic apoB mRNA concentration. |
Glucose Tolerance Test. Figure 8 shows Glucose tolerance test (GTT) administered to animals that were fed TO as 30% of total caloric intake, and the respective control animals fed regular control chow diet for two weeks, after which the animals were challenged with 25mg glucose per g of body weight. Blood glucose was measured at 30mins intervals and the means were calculated. Calculating the area under the curve revealed no difference between the two diet groups, however each of the group had a different response. The control diet had a sharp increase in blood glucose levels, followed by a sharp decrease, on the other hand the TO-fed group had a slower increase that was followed by a slower decrease in blood glucose levels.
 |
Figure 8: Effect of feeding Triolein or Control diet for two weeks on Glucose Tolerance Test in the SHSHP rat. |
Plasma Insulin and HOMA-IR. As shown in Table 1, levels animals in the TO group had higher levels of phospholipids and lower levels of insulin. NO-1866 lowered HOMA-IR by almost 40% in the control and the TFA group, however far less reduction was observed in the Triolein group. The control and Trans fatty acids groups had up to 45% higher HOMA-IR than the Triolein group. Phospholipids levels were elevated with TO group when compared with control.
Discussion
Findings from this study suggests that monounsaturated dietary FA can individually or in combination with an LPL raising drug increase plasma HDL-C levels. These data fit well with our overall hypothesis that dietary components can raise HDL-C levels if the rate of lipolysis of TG-rich lipoproteins are raised to make available sufficient HDL apolipoprotein (either derived from the chylomicron/VLDL lipolysis itself or pre-existing in the plasma) to bind and maintain the excess phospholipid also generated in the process.
Fat feeding stimulates production of chylomicrons and VLDL and lipoproteins containing apolipoprotein E and A-I, both of which are required for formation of HDL. We previously demonstrated that species of dietary fatty acids vary in how they stimulate phospholipid rich precursor particles (VLDL/VHDL) for HDL (Hatahet et al., 2003; Kudchodkar et al., 2000). TG-rich lipoproteins can be a source of apoA-I, and if there is impairment of chylomicron/VLDL secretion, the process of lipolysis cannot generate this important source of HDL apolipoprotein. Therefore, the levels of apolipoprotein will be determined by the abundance of free or HDL-associated apolipoproteins, the efficiency of lipolysis, or even the rate of VLDL synthesis. Mechanistically lipolysis of VLDL or chylomicrons, will generate surface material for nascent HDL particle formation that can become associated with pre-existing HDL, resulting in larger HDL particles with a reduced clearance time (Blanchette-Mackie & Scow, 1976; Tall, Green, Glickman, & Riley, 1979). Decreased rates of lipolysis can be due to a variety of systemic changes that may include reduced peripheral-bound LPL which is supposed to ‘tether’ TG-rich particles to muscle and/or adipose tissue, or elevated apoC-III that may reduce active LPL and thus slow down or reduce the ability to enzymatically hydrolyze the neutral lipid core. Similarly, phospholipids (PL) and cholesterylester (CE) transfer are increased in LPL deficiency which may partly explain low HDL-levels in LPL-deficient subjects (Kaser et al. 2003). In the current study, feeding triolein increased phospholipid and HDL-C, a finding which is in agreement with Kaser and colleagues (2003). Therefore, studies that determine the rates of chylomicrons/VLDL and HDL catabolism or production rates will aid in the delineation of the mechanism for low HDL in the insulin resistant and hypertensive states.
In the present study, NO-1886 administration resulted in significant elevation of HDL-C concentration in the TO and CON groups but not in the TFA group. These findings were similar to Kusunoki et al., 2002 and Yin et al., 2002, who used diet induced diabetic Male New Zealand White rabbits and found that NO-1886 caused an elevation in HDL-C concentration through promoting lipolytic enzymes expression (Yin et al., 2002). Kusunoki et al., 2002 used diet-induced insulin resistant male Sprague-Dawley rats to demonstrate that NO-1886 also successfully improved HDL-C concentration. The effect of oleic acid is similar to Gilmore et al., (2011) who reported that high oleic acid meat increased HDL-C concentrations in humans. The type and species of fatty acids administered was found to play a key role in the activation of LPL in studies by who reported that a diet rich in omega 3 fatty acids seemed to protect from atherosclerosis even among insulin resistant models in agreement with the findings of the present study. Moreover, our findings indicted that treatment with NO-1886 and oleic acid caused significant reductions in triacylglycerol concentration only in the control and TO groups in accordance with the findings of Lacroix et al., 2012 and Mohammadifard et al., 2013, in which TFA were found to be detrimental to serum and plasma lipids.
In studies reviewed by Pape and Bisgaier (1998) reviewed the discovery of HDL-elevating therapy and how that impacted TG-rich lipoproteins, and suggested that the reduction in apoC-III allows for more rapid uptake of remnants by the liver, resulting to insufficient time to generate surface material. In addition, they reported that if LCAT-derived cholesterol is removed rapidly from the HDL by cholesteryl ester transfer protein (CETP), then there may not be a detectable increase in steady-state HDL levels, given the increased rate of surface material production (Bisgaier & Pape, 1998). The model in this case is the insulin resistant SHRSP rat, which like all rodents do not have CETP, but may have other lipid transfer proteins. This however, could be the case in species such as monkey, humans and rabbits, which have CETP in their plasma. Humans in general, have significantly less CETP activity (approximately 10 to 25% of the level), compared to rabbits and other non-human primates, which suggests that the CETP-mediated transfer of LCAT-derived HDL cholesteryl esters will be less likely to reduce HDL in humans by the above mechanism.
There was no significant increase in HDL-C concentration with the TFA fed group following the introduction of NO-1886. This can be attributed to the documented role of TFA in reducing HDL-C as reported by (Lacroix et al., 2012) in humans. The current study shows that the TO group had the lowest HOMA IR index which could highlight the role of healthy fatty acids in improving insulin resistance. Although many mechanisms linking insulin resistance and FAs remain not well understood, Wang et al., 2012 reported that FFAs might contribute to further the progression of insulin resistance which would counter the protective role of NO-1886 as seen in the HOMA IR findings for the Control and TFA groups.
Table 1: Effect of fatty acids, Triolein (TO), Trans Fatty Acid (TFA) with/without NO-1886 on plasma phospholipids (PL), Insulin and Insulin resistance (HOMA-IR. abcMeans with similar superscripts between Diet ± NO-1886 are not different (P>0.05).
|
Diet |
Weight (g) |
Adipose tissue (g) |
PL mg/dL |
Insulin µIU/ml |
HOMA-IR |
| CON |
328.5±37 |
16±5 |
112±4 |
195±7b |
66±4a |
| CON + NO-1866 |
358.7±19 |
15±1 |
92±29b |
153±12 |
46±2b |
| TO |
363.5±28 |
15±6 |
141±12a |
105±25a |
36±8c |
| TO + NO-1866 |
333.6±19 |
14±2 |
142±11a |
112±29a |
31±7c |
| TFA |
339.2±28 |
17±1 |
124±11a |
162±5.1b |
64±15a |
| TFA + NO-1866 |
333.8±21 |
16±3 |
130±6a |
117±7 |
39±4c |
Conclusion
In Conclusion, findings from this study, suggest that dietary fat supplementation alone or in combination with NO-1886 raised LPL activity significantly in the SHRSP rat. Although feeding TO and TFA increased plasma triacylglycerol, TO feeding reduced insulin levels by 50% and by 25% respectively when compared to Control group. Similarly, the TO group had the lowest HOMA-IR index with or without NO-1886 which corresponded to the slower glycemic response to the GTT compared to the Control diet. Over all, our results indicate that stimulation the activity of lipoprotein lipase by dietary means, including the feeding of monounsaturated fat may increase HDL-C, reduce plasma triacylglycerol and symptoms of MetS risk, and thus may decrease vascular complications through the normalization of lipid metabolism associated with metabolic syndrome.
Acknowledgements
This work was supported by in part, by an institutional research fellowship and by a National Institutes of Health grant #HL03389 from the U.S. Public Health Service to Dr. Fungwe.
References
- Bisgaier, C. L., & Pape, M. E. (1998). High density lipoprotein: Are elevated levels desirable and achievable? Current Pharmaceutical Design, 4(1), 53-70.
- Blanchette-Mackie, E. J., & Scow, R. O. (1976). Retention of lipolytic products in chylomicrons incubated with lipoprotein lipase: Electron microscope study. Journal of Lipid Research, 17(1), 57-67.
- Chang, C. L., Seo, T., Du, C. B., Accili, D., & Deckelbaum, R. J. (2010). N-3 fatty acids decrease arterial low-density lipoprotein cholesterol delivery and lipoprotein lipase levels in insulin-resistant mice. Arteriosclerosis, Thrombosis, and Vascular Biology, 30(12), 2510-2517.}
CrossRef - Eckel, R. H., Grundy, S. M., & Zimmet, P. Z. (2005). The metabolic syndrome. The Lancet, 365(9468), 1415-1428.
CrossRef - Gilmore LA1, Walzem RL, Crouse SF, Smith DR, Adams TH, Vaidyanathan V, Cao X, Smith SB. (2011) Consumption of high-oleic acid ground beef increases HDL-cholesterol concentration but both high- and low-oleic acid ground beef decrease HDL particle diameter in normocholesterolemic men. J Nutr 141(6):1188-94.
CrossRef - Goldberg, I. J., & Merkel, M. (2001). Lipoprotein lipase: Physiology, biochemistry, and molecular biology. Front Biosci, 6(4), 388-405.
- Hatahet, W., Cole, L., Kudchodkar, B. J., & Fungwe, T. V. (2003). Dietary fats differentially modulate the expression of lecithin:Cholesterol acyltransferase, apoprotein-A1 and scavenger receptor b1 in rats. The Journal of Nutrition, 133(3), 689-694.
- Kako, Y., Masse, M., Huang, L. S., Tall, A. R., & Goldberg, I. J. (2002). Lipoprotein lipase deficiency and CETP in streptozotocin-treated apoB-expressing mice. Journal of Lipid Research, 43(6), 872-877.
- Kaser S, Sandhofer A, Hölzl B, Gander R, Ebenbichler CF, Paulweber B, Patsch JR. (2003) Phospholipid and cholesteryl ester transfer are increased in lipoprotein lipase deficiency. J Intern Med. 253(2):208-16.
CrossRef - Klos KL, Kullo IJ. Genetic determinants of HDL: monogenic disorders and contributions to variation. Curr Opin Cardiol. 2007; 22: 344-351
CrossRef - Kudchodkar, B. J., Lacko, A. G., Dory, L., & Fungwe, T. V. (2000). Dietary fat modulates serum paraoxonase 1 activity in rats. The Journal of Nutrition, 130(10), 2427-2433.
- Kusunoki, M., Tsutsumi, K., Hara, T., Ogawa, H., Nakamura, T., Miyata, T., Kakumu, S. (2002). Correlation between lipid and glycogen contents in liver and insulin resistance in high-fat fed rats treated with the lipoprotein lipase activator NO-1886. Metabolism, 51(6), 792-795.
CrossRef - Lacroix, E., Charest, A., Cyr, A., Baril-Gravel, L., Lebeuf, Y., Paquin, P., Lamarche, B. (2012). Randomized controlled study of the effect of a butter naturally enriched in trans fatty acids on blood lipids in healthy women. The American Journal of Clinical Nutrition, 95(2), 318-325. doi:10.3945/ajcn.111.023408
CrossRef - Matthews, D., Hosker, J., Rudenski, A., Naylor, B., Treacher, D., & Turner, R. (1985). Homeostasis model assessment: Insulin resistance and β-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia, 28(7), 412-419.
CrossRef - Merkel, M., Eckel, R. H., & Goldberg, I. J. (2002). Lipoprotein lipase: Genetics, lipid uptake, and regulation. Journal of Lipid Research, 43(12), 1997-2006.
CrossRef - Mohammadifard, N., Hosseini, M., Sajjadi, F., Maghroun, M., Boshtam, M., & Nouri, F. (2013). Comparison of effects of soft margarine, blended, ghee, and unhydrogenated oil with hydrogenated oil on serum lipids: A randomized clinical trail. ARYA Atherosclerosis, 9(6), 363-371.
- Munoz-Barriosa S, Paola Guzman-Guzmana, I., FranciscoMunoz-Valleb, J., Salgado-Bernab B., Salgado-Goytiaa, L. and Parra-Rojasa. I. (2012) Association of the HindIII and S447X polymorphisms in LPL gene with hypertension and type 2 diabetes in Mexican families. Disease Markers 33: 313–320
CrossRef - Tall, A. R., Green, P. H., Glickman, R. M., & Riley, J. W. (1979). Metabolic fate of chylomicron phospholipids and apoproteins in the rat. The Journal of Clinical Investigation, 64(4), 977-989.
CrossRef - U.S. Department of Health and Human Services. (1985). National research council guide for the care and use of laboratory animals. (No. Publication No. 85-23). Bethesda, MD.: National Institutes of Health.
- Wang, Z., Zeng, H., Wei, H., Yi, G., Yu, J., Wang, Y., Yin, W. (2012). NO-1886 ameliorates glycogen metabolism in insulin-resistant HepG2 cells by GSK-3ß signaling. The Journal of Pharmacy and Pharmacology, 64(2), 293-301. doi:10.1111/j.2042-7158.2011.01402.x
CrossRef - Yin, W., Tsutsumi, K., Yuan, Z., & Yang, B. (2002). Effects of the lipoprotein lipase activator NO-1886 as a suppressor agent of atherosclerosis in aorta of mild diabetic rabbits. Arzneimittelforschung, 52(08), 610-614.
- Yuan, G., Al-Shali, K. Z., & Hegele, R. A. (2007). Hypertriglyceridemia: Its etiology, effects and treatment. Canadian Medical Association Journal. 176(8), 1113-1120.
CrossRef.

This work is licensed under a Creative Commons Attribution 4.0 International License.




