Introduction
Coffee is a member of the Rubiaceae family, comprising around 70 species. Two of these species are internationally significant: Arabica (Coffea Arabica) and Robusta (Coffea Canephora).1 Robusta originated mainly from lowlands of Central and West Africa as well as South Asia primarily Vietnam, whereas Arabica frequently hails from South America (primarily from Brazil) and the upland and mountainous regions of East Africa.2 Coffee beans are well known for their antioxidant properties.3,4
Antioxidants, found naturally in many foods and beverages. It aims to prevent cellular damage triggered by free radicals, that is associated with diseases including cancer and heart disease.5 Free radicals are reactive oxygen species that circulate throughout the body and are produced as byproducts of a variety of processes. Under normal conditions, antioxidant systems remove them from the body but when these normal mechanisms are interrupted, radicals build in excess, contributing to the development of oxidative stress.6 Oxidative stress occurs when the body experiences an imbalance between free radicals’ production and antioxidant defenses.7
Green Coffee Beans (GCB) are unroasted coffee seeds from coffea plant. Carbohydrates (55.0–65.5%), fats (10.0–18.0%), nitrogen containing compounds (11.0–15.0%), purine alkaloids (0.8–4.0%), chlorogenic acids (CGA) (6.7-9.2%), and minerals (3.3–5.4%) make up the majority of the content of GCB. Rich in bioactive substances with antioxidant qualities, green coffee extract contains trigonelline, diterpenes, caffeine, and CGA.8,9 The key phenolic chemicals present in green coffee bean extract are caffeic acid, caffeine, and CGA. These compounds exhibit antioxidant, antimutagenic, anticancer, and antimicrobial.10-12
Green coffee beans contain more 5-O-caffeoylquinic acid 13 (5-CQA) than roasted coffee, often up to twice more depending on the roasting time. Caffeine suppresses oxidative stress and strengthens the antioxidant system. In human skin fibroblasts, caffeine inhibits hydrogen peroxide-induced lipid peroxidation products, and inhibits tissue lipid peroxidation and lowers Reactive Oxygen Species (ROS).2
Considering the respective concentrations, Robusta has 2.2% of caffeine and 7.0–10.0% of chlorogenic acid, while Arabica typically has 1.2% of caffeine and 5.5–8.0% of chlorogenic acid.14-15
Solvent extraction is a popular method for isolating antioxidants. The solvent has a significant impact on the yield, phenolic content, and antioxidant activity of extracts, due to the varying antioxidant potentials of compounds with different polarities.16 The extraction and purification of phytochemicals and antioxidants from plant material is influenced by an array of factors, including temperature, time, solvent concentration, and solvent polarity. Different phytochemicals are extracted in solvents with varying polarity based on their chemical composition, as no particular solvent may be dependable enough to recover all the phytochemical and antioxidant substances present in the plant material.17-18
It was found that using polar and non-polar solvents in combination enhances the efficiency of phytochemicals with high antioxidant content being extracted from bean and other legume seeds.17 In comparison to other extracts, it was discovered in the investigation that T. garganica ethyl acetate extracts showed potent antioxidant and radical scavenging properties.19 Green coffee water and ethanol extracts demonstrated potent antioxidant characteristics as well as a significant capacity to bring down transition metal ions.11
Although the effect of solvent polarity on plant secondary metabolites and antioxidant activity is a known fact but it is different for each plant species. The effect of solvent polarity on the secondary metabolites and antioxidant activity of green coffee is still a matter of curiosity. Green coffee has been the subject of previous research based on the influence of polarity, but previous studies on the impact of polarity on the antioxidant activity and polyphenolic content of green coffee have not examined the range of eight solvents, which ranges from low to high polarity. Therefore, the purpose of this study was to assess the manner in which extracting solvents affected the entire polyphenolic content and antioxidant capacity of the crude extracts obtained from green coffee beans.
Material and Methods
Chemicals
Folin-Ciocalteu (Phenol Reagent), 2,2-diphenyl-1-picrylhydrazyl (DPPH), 2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS), 2,4,6-Tripyridyl-S-triazine (TPTZ), Sodium Carbonate, Ferric chloride, Potassium per sulphate, Sodium Nitrate, Sodium Hydroxide, Aluminium Chloride were purchased from Sigma– Aldrich Company, USA and Sisco Research Laboratories Pvt. Ltd, India. Any other chemicals were of analytical grade.
Sampling
A mature and sundried green coffee bean (Coffea Robusta) was purchased in Kannur, Kerela, India. The hard seed coat and silver skin were scraped away with a keen, clean blending knife. The material was then processed with a kitchen milling machine and passed through a 60-mesh filter to produce the sample powder for analysis.
Extract Preparation and % Extraction Yield
Extract preparation was performed using conventional dry method of extraction based on water bath, for which 2 g sample was ground and dissolved in 20 mL of an appropriate solvent. Mixture was left on vortex and the polyphenols were extracted for 1 h at room temperature, then centrifuged at 6000rpm for 5 min. Filtration was done using whatmann No.1 in a pre-constant beaker and placed on water bath until the extract was evaporated to dryness.20-22 Final weight of the beaker taken and the total extractable components (TEC) were calculated using:17
TEC % = Wt. of Extract/ Wt. of Sample X 100
The residues obtained after extraction were then dissolved in the respective solvent and used for phytochemical and antioxidant analysis. The solvents used for extraction were arranged in Table 1, on the basis of their polarity from least to most polar.
Table 1: Polarity of the organic solvents used for extraction of Phenolic compounds
|
S.No. |
Solvents | Polarity Index |
| 1 | Hexane |
0.0 |
|
2 |
Dichloromethane (DCM) | 3.1 |
| 3 | Chloroform (CHF) |
4.1 |
|
4 |
Ethanol | 5.2 |
| 5 | Methanol |
5.1 |
|
6 |
Acetone | 5.1 |
| 7 | Dimethylsulphoxide (DMSO) |
7.2 |
|
8 |
Aqueous (Aq.) |
10.2 |
Source:23
Polyphenolic Content Analysis
Total Polyphenolic Content (TPC)
The TPC quantification was performed by using the phenol reagent method.24 At initial, 1mL of distilled water was added with 20µL of prepared extract. Into the reaction mixture100µL of phenol reagent was incorporated and kept in incubator at 38oC for 3 minutes. 280 µL of 25% w/v sodium carbonate solution and 600 µL of distilled water were introduced to the mixture. Following one hour incubation at room temperature in the dark, the absorbance was taken at 765 nm using a UV-Visible Spectrophotometer against a blank containing phenol reagent and distilled water with no extract. The total phenolic amount was calculated as gallic acid equivalent (GAE).
Total Flavonoid Content (TFC)
The TFC was calculated using the colorimetric method with minor modifications in which 1mL of prepared extract was added in 10 mL test tube containing 4 mL of distilled water. In the same test tube, 0.3 mL of 5% NaNO2 and 10% AlCl3 (After 5 minutes) were added. After 5 minutes of incubation, 2 mL of 1 M NaOH was added, and subsequently the total volume was made up to 10 mL using distilled water. The absorbance was quantified at 510 nm against a prepared reagent blank using a UV-visible spectrophotometer.25
Antioxidant Activity
DPPH free radical scavenging assay
In the beginning 100μL of extract was placed in test tube. Hereafter, 150 μL of DPPH solution (2.5 mg DPPH in 100 ml of methanol) was added in the same. Then after, entire solution mixture was kept in dark for 20 minutes after providing 3mL methanol. The measurement of absorbance was performed at 515 nm.26 A DPPH free radical scavenging activity was calculated as follows:
Scavenging activity (%) = [(Acontrol- Asample) /Acontrol] x 100
Where,
Acontrol = absorbance without extract
Asample = absorbance with the extract.
ABTS free radical scavenging assay
The ABTS assay was carried out employing the technique described below. Distilled water was used to prepare an ABTS diammonium salt solution of 7.6 mM and a potassium persulfate solution of 2.5 mM. Each solution was kept in a dark room for 12 hours. After 30 minutes of incubation, the two solutions were mixed and refrigerated for 24 hours before being diluted in ethanol.27 Using a UV-Visible spectrophotometer, the absorbance was calculated at 670 nm in percentage using the given formula:
ABTS Scavenging activity (%) = [(Acontrol- Asample) /Acontrol] * 100
Acontrol = absorbance without extract
Asample = absorbance with the extract.
Ferric Reducing Antioxidant Power (FRAP)
The FRAP Activity of the sample was evaluated spectrophotometrically technique. The FRAP reagent was made by combining 300 mM acetate buffer, 10 mL 2,4,6-tris(2-pyridyl)-1,3,5-triazine (TPTZ) in 40 mM HCl, and 20 mM FeCl3 in a 10:1:1 ratio at 37°. To produce the FRAP reagent, use a 1-5 mL variable micropipette (3.995 mL) to combine it with 5 µL of diluted plant sample. After 30 minutes of incubation at 37°C, the absorbance at 593 nm was measured against a blank reagent (3.995 mL FRAP reagent + 5 µL distilled water). The FRAP values were calculated by comparing the absorbance change in the test mixture to those obtained from increasing Fe3+ concentrations and expressed as mmol Fe II eq/g.28
Statistical Analysis
The statistics gathered were displayed as mean + standard deviation. Using SPSS version 21.0, a one-way analysis of variance (ANOVA) test and Duncan’s multiple range method were used to compare the significant differences. The values of P ˂0.05 were considered statistically significant. Using Rstudio version 4.3.2, Pearson’s correlation coefficients (P <0.01) were also determined.
Results
Total Extractable Component (TEC)
The results entail that among the solvents, the highest extracting yield was obtained in acetone (3.9%), followed by methanol (3.5%), ethanol (2.9%), CHF (2.7%), DCM (2.5), aqueous (2.4%), hexane (1.5%) and DMSO (1.5%) shown in Table 2. The results show a substantial (p< 0.05) effect of extracting solvent on extraction yield content, with acetone, methanol and ethanol (polarity- 5.1 to 5.2) being more effective to extract the greatest amount of secondary metabolites also indicates abundance of low polar bioactive compounds in green coffee beans. Similar results were obtained in other studies.31 A very low correlation (r2 = 0.138) has been found between solvent polarity and TEC content which simply means there is no effect or limited effect of solvent polarity on the total extractable yield of the GCB as shown in table 3.
Table 2: TEC content of GCB in different solvents
|
Solvent |
% TEC |
| Acetone |
3.9 |
|
Methanol |
3.5 |
| Ethanol |
2.9 |
|
CHF |
2.7 |
| DCM |
2.5 |
|
Aq. |
2.4 |
| Hexane |
1.5 |
|
DMSO |
1.5 |
Total Polyphenolic Content (TPC) and Total Flavonoid content (TFC)
TPC was found significantly high Figure 1 in methanol (114.36mg/g) followed by CHF (60.66mg/g) hexane (60.36mg/g) water (58.03) acetone (53.33) DCM (33.80mg/g) DMSO (27.50mg/g) and ethanol (26.80mg/g). Similar result was also found in the study of low-grade coffee beans.1 TFC content was also significantly higher Figure 2 in methanol (9.88mg/g) followed by acetone (8.17mg/g), hexane (7.853mg/g), CHF (4.66mg/g), aqueous (3.49mg/g), DCM (2.39mg/g), DMSO (1.59mg/g) and ethanol (1.45mg/g). A negative correlation (Table 3) was found between the polarity of solvent and total flavonoid content (r2 = -0.35) and total polyphenolic content (r2 = -0.06).
 |
Figure 1: Shows the Total Phenolic Content (TPC) of GCB. |
 |
Figure 2: Shows the Total Flavonoid Content (TFC) of GCB. |
Table 3: Correlation coefficient (r2) of TEC, TPC, %Antiradical, ABTS and FRAP versus Polarity of the solvent
| %TEC | TPC | TFC | %Antiradical | %ABTS Scavenging activity | FRAP | |
| Solvent Polarity | 0.138 | -0.06 | -0.35 | 0.62 | 0.81 | 0.65 |
| Correlation is significant at P <0.01 | ||||||
Total Antioxidant Activity
Table 4 shows that the values from various polarity solvents were significantly different (p< 0.05) and the highest DPPH scavenging activity (94.22±0.45%) was attained by ethanol, followed by DCM, DMSO, aqueous, acetone, methanol, CHF and hexane (Figure 3). Thus, it is stated that ethanol is the most suitable solvent (among all the other tested pure solvents) for DPPH free radical activity of GCB. The range of the DPPH free radical scavenging activity is 94.22±0.45 to 16.80±0.50%.
It was again reported in table 4 that ABTS activity and FRAP activity of all the extracts was significantly different (p <0.05). Among all the seven solvents, aqueous extract has the highest ABTS activity (96.51%) and FRAP activity (0.13 mmol Fe II/g) as shown in Figure 4 & Figure 5. This indicates that the aqueous is the most suitable solvent for the ABTS and FRAP activity of GCB.
A positive very strong correlation (Table 3) was found between the polarity of solvent and ABTS Activity (r2 = 0.81) and FRAP Activity (r2 = 0.65) whereas, strong correlation (r2=0.62) was found between the polarity of the solvent and DPPH scavenging activity.
Table 4: Total Polyphenolic Content and Antioxidant Activity of GCB among the tested solvents
|
|
POLYPHENOLS | ANTIOXIDANTS | |||
| Solvents | TPC (mg GAE/g) | TFC (mgQE/g) | %Antiradical Activity (%) | % ABTS (%) |
FRAP (mmol Fe II/g) |
|
Aqueous |
58.03±0.25c | 3.59±0.01e | 88.53±0.54d
|
96.51±0.79a | 0.13±0.002a |
| CHF | 60.66±0.57b | 4.66±0.05d | 28.52±0.42g | 2.34±0.55g |
0.03±0.007d |
|
DCM |
33.80±0.26e | 2.39±0.01f | 93.70±0.21a | 3.80±0.89f | 0.03±0.003d |
| Acetone | 53.33±1.52d | 8.17±0.15b | 65.78±0.062e | 32.83±0.67e |
0.07±0.005c |
|
DMSO |
27.50±0.55f | 1.59±0.03g | 89.88±0.11c
|
40.56±0.75d | 0.03±0.006d |
| Hexane | 60.73±0.64b
|
7.85±0.05c | 16.80±0.50h | 2.29±0.53g |
0.03±0.001d |
|
Methanol |
114.36±0.25a | 9.88±0.05a | 88.40±0.87f | 44.24±0.99c | 0.07±0.001c |
|
Ethanol |
26.80±0.65f | 1.45±0.03g | 94.22±0.45a | 74.63±0.57b |
0.11±0.002b |
Mean values with the same letter in the same column (a, b, c, d, e, f, g, h) for a specific concentration and extract type do not differ significantly at p < 0.05.
 |
Figure 3: Shows the DPPH scavenging activity of GCB. |
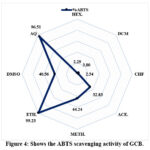 |
Figure 4: Shows the ABTS scavenging activity of GCB. |
 |
Figure 5: Shows the Ferric Reducing Antioxidant Power (FRAP) of GCB. |
Discussion
Coffee is a functional food containing two most biologically active compounds; CGA and caffeine, responsible for flavor formation, possess massive health effects with antioxidant properties.43 While extracting such bioactive compounds from green coffee beans, different solvents had shown a considerable effect on the extractable solids yield. The relationship between the solvent types and its polarity, emerged as significant at p<0.05, showing that the polarity of the solvent is an important variable to consider in the estimation of TEC.29-30 The structure of a solute, including its phenolic content, has a significant role in determining its solubility in solvents.32 Many studies found that methanol, acetone and water are the better solvents for higher yield and extractability.33-34 Despite Water and DMSO being more polar, TEC content was lower which signifies the presence of less highly polar compounds in GCB.
Polyphenols are plant secondary metabolites, essential for various physiological functions of plants, and participate in defense mechanisms against stress and stimuli.44 In GCB, Chlorogenic acid (CGA) is the major phenolic compound present, with redox properties that make them excellent antioxidants and act as reducing agents, singlet oxygen quenchers, and hydrogen donors.45 Furthermore, studies render an inverse association between polyphenolic-rich food intake and the risk of chronic human diseases.46 However, the differences observed between the polyphenolic content in different solvents were mainly related to their different characteristics, such as polarity, and thus, solubility, of individual compounds in the extraction solvent.35 Eventually, it has been demonstrated that alcohol, despite of having less polarity than aqueous, dissolves cell walls and seeds more effectively than water, resulting in more polyphenol release from the cells.36 Methanol is the optimal solvent for extracting polyphenol compounds due to its ability to inhibit polyphenol oxidase reactions and its ease of evaporation compared to water.37 A study on polyphenolic content of GCB from different region of Ethiopia reported total soluble polyphenols in GCB ranges from 21.86 mgGAE/g to 43.6 mgGAE/g, cell wall bound polyphenols range from 8.6-15.3 mgGAE/g and flavonoid content from 3.3-6.2 mgCE/g.38 Another study reported polyphenols content 90.95 mgGAE/g to 287.54 mgGAE/g in roasted and green coffee beans of Thailand.39 Several findings revealed that the aqueous extracts having the maximum antioxidant activity effect because of its high polar nature, followed by the methanol and acetone extracts.41-42 However, a research work has also support that DPPH radical scavenging activity was recorded highest in ethanol extract of GCB.29 It is important to note activity of coffee beans, can be influenced by factors such as the degree of maturation, coffee species, geographical origin, and the preparation process.40,2
Conclusion
The polarity of solvent has a significant influence on the extraction, polyphenols and antioxidant activities. In this study, it was found that the total extraction yield of acetone, methanol and ethanol (3.9>3.5>2.9) were significantly different despite of having polarity index almost similar (5.1<5.1<5.2). On the contrary, methanol being less polar (5.1) compared to aqueous (10) has the extensive polyphenolic content (TPC-114.6> 58.03 mg GAE/g; TFC- 9.88>3.59 mg QE/g respectively), implying that methanol is an efficient solvent for polyphenol extraction. Similar result was found as in case of DPPH free radical scavenging activity, found greatest in ethanol (94.22%) with respect to high polar aqueous (88.53%). At the same time, ABTS and FRAP activity was highest in aqueous (96.51% and 0.13mmol Fe II/g respectively) followed by ethanol (74.63% and 0.11mmol Fe II/g respectively). This study reveals that medium polar solvent such as acetone, methanol and ethanol were the best solvents for the extraction of polyphenolic content and antioxidant activity in GCB. The correlation coefficient also indicates that polyphenolic content and antioxidants are not just completely dependent on solvent polarity.
Generally, the trend of recent studies tends to believe that only high polar solvents can extract high antioxidant value and polyphenolic content. However, it is occasionally found that low polar and mid polar solvents can also produce excellent extraction outcomes in terms of antioxidant and polyphenolic content. Consequently, this research suggests that future investigations should give equal emphasis to low polar, mid polar and high polar solvent types because the value of the antioxidant and polyphenolic content depends not only on polarity but also on other factors such as plant material, phenols present in sample, methodology, temperature and time duration of extraction.
Acknowledgement
The authors are grateful to the Department of Family and Community Sciences, University of Allahabad, Prayagraj, Uttar Pradesh, India for providing the laboratory facilities throughout the experimental work.
Conflict of Interest
The authors declare no conflict of interest.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
This statement does not apply to this article.
Ethics Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed Consent Statement
The authors give their consent for the publication of identifiable details to be published in the Journal.
Permission to Reproduce Material from other Sources
Not applicable
Clinical Trial Registration
This research does not involve any clinical trials.
Author Contributions
- Shraddha Tripathi: Writing – review &editing.
- Shirshika Singh: Writing – original draft.
- Neha Mishra: Reviewed the manuscript.
- Neetu Mishra: Conceptualization, review, editing & finalizing.
References
- Ramalakshmi K, Kubra IR, Rao LJ. Antioxidant potential of low-grade coffee beans. Food Research International. 2008 Jan 1;41(1):96-103.
CrossRef - Jeszka-Skowron M, Sentkowska A, Pyrzyńska K, De Peña MP. Chlorogenic acids, caffeine content and antioxidant properties of green coffee extracts: influence of green coffee bean preparation. European Food Research and Technology. 2016 Aug;242:1403-9.
CrossRef - Gligor O, Clichici S, Moldovan R, Muntean D, Vlase AM, Nadăș GC, Matei IA, Filip GA, Vlase L, Crișan G. The effect of extraction methods on phytochemicals and biological activities of green coffee beans extracts. Plants. 2023 Feb 6;12(4):712.
CrossRef - van der Werf R, Marcic C, Khalil A, Sigrist S, Marchioni E. ABTS radical scavenging capacity in green and roasted coffee extracts. LWT-Food Science and Technology. 2014 Sep 1;58(1):77-85.
CrossRef - Zeb A. Concept, mechanism, and applications of phenolic antioxidants in foods. Journal of Food Biochemistry. 2020 Sep;44(9):e13394.
CrossRef - Chaudhary P, Janmeda P, Docea AO, Yeskaliyeva B, Abdull Razis AF, Modu B, Calina D, Sharifi-Rad J. Oxidative stress, free radicals and antioxidants: Potential crosstalk in the pathophysiology of human diseases. Frontiers in chemistry. 2023 May 10;11:1158198.
CrossRef - Jomova K, Raptova R, Alomar SY, Alwasel SH, Nepovimova E, Kuca K, Valko M. Reactive oxygen species, toxicity, oxidative stress, and antioxidants: Chronic diseases and aging. Archives of toxicology. 2023 Oct;97(10):2499-574.
CrossRef - Macheiner L, Schmidt A, Schreiner M, Mayer HK. Green coffee infusion as a source of caffeine and chlorogenic acid. Journal of Food Composition and Analysis. 2019 Dec 1;84:103307.
CrossRef - Brzezicha J, Błażejewicz D, Brzezińska J, Grembecka M. Green coffee VS dietary supplements: A comparative analysis of bioactive compounds and antioxidant activity. Food and Chemical Toxicology. 2021 Sep 1;155:112377.
CrossRef - Duangjai A, Rawangkan A, Siriphap A, Kiddee A, Yosboonruang N, Yosboonruang A. A Promising Functional Food for Diabetes Prevention, Antioxidation, and Anti-inflammation of Green Coffee Bean Extract. Journal of Human, Earth, and Future. 2024 Mar 1;5(1):100-10.
CrossRef - Masek A, Latos-Brozio M, Kałużna-Czaplińska J, Rosiak A, Chrzescijanska E. Antioxidant properties of green coffee extract. Forests. 2020 May 15;11(5):557.
CrossRef - Palmieri MG, Cruz LT, Bertges FS, Húngaro HM, Batista LR, da Silva SS, Fonseca MJ, Rodarte MP, Vilela FM, do Amaral MD. Enhancement of antioxidant properties from green coffee as promising ingredient for food and cosmetic industries. Biocatalysis and agricultural biotechnology. 2018 Oct 1;16:43-8.
CrossRef - Clifford MN. Chlorogenic acids and other cinnamates–nature, occurrence and dietary burden. Journal of the Science of Food and Agriculture. 1999 Mar 1;79(3):362-72.
CrossRef - Raseetha Siva RS, Noratikah Rajikin NR, Zaibunnisa Abdul Haiyee ZA, Wan Iryani WI. Assessment of antioxidant activity and total phenolic content from green coffee Robusta sp. beans.
- Garrett R, Schwab NV, Cabral EC, Henrique BV, Ifa DR, Eberlin MN, Rezende CM. Ambient mass spectrometry employed for direct analysis of intact arabica coffee beans. Journal of the Brazilian Chemical Society. 2014;25:1172-7.
CrossRef - Barchan A, Bakkali M, Arakrak A, Pagán R, Laglaoui A. The effects of solvents polarity on the phenolic contents and antioxidant activity of three Mentha species extracts. Int. J. Curr. Microbiol. Appl. Sci. 2014 Dec 10;3(11):399-412.
- Nawaz H, Shad MA, Rehman N, Andaleeb H, Ullah N. Effect of solvent polarity on extraction yield and antioxidant properties of phytochemicals from bean (Phaseolus vulgaris) seeds. Brazilian Journal of Pharmaceutical Sciences. 2020 Mar 16;56:e17129.
CrossRef - Iloki-Assanga SB, Lewis-Luján LM, Lara-Espinoza CL, Gil-Salido AA, Fernandez-Angulo D, Rubio-Pino JL, Haines DD. Solvent effects on phytochemical constituent profiles and antioxidant activities, using four different extraction formulations for analysis of Bucida buceras L. and Phoradendron californicum. BMC research notes. 2015 Dec;8:1-4.
CrossRef - Athmouni K, BELGHITH T, BELLASSOUAD K, El Feki A, AYADI H. Effect of solvent polarity on the content of biomolecules and antioxidant activity of Thapsia garganica (Apiaceae). Algerian Journal of Natural Products. 2015;3(3):194-208.
- Abubakar AR, Haque M. Preparation of medicinal plants: Basic extraction and fractionation procedures for experimental purposes. Journal of Pharmacy and Bioallied Sciences. 2020 Jan 1;12(1):1-0.
CrossRef - Pandey A, Tripathi S. Concept of standardization, extraction and pre phytochemical screening strategies for herbal drug. Journal of Pharmacognosy and phytochemistry. 2014;2(5):115-9.
- Ujang ZB, Subramaniam T, Diah MM, Wahid HB, Abdullah BB, Abd Rashid AH, Appleton D. Bioguided fractionation and purification of natural bioactives obtained fromalpinia conchigera water extract with melanin inhibition activity.
- Ricciutelli M, Di Martino P, Barboni L, Martelli S. Evaluation of rapamycin chemical stability in volatile-organic solvents by HPLC. Journal of pharmaceutical and biomedical analysis. 2006 Jun 7;41(3):1070-4.
CrossRef - Priftis A, Stagos D, Konstantinopoulos K, Tsitsimpikou C, Spandidos DA, Tsatsakis AM, Tzatzarakis MN, Kouretas D. Comparison of antioxidant activity between green and roasted coffee beans using molecular methods. Molecular medicine reports. 2015 Nov 1;12(5):7293-302.
CrossRef - Ribarova F, Atanassova M, Marinova D, Ribarova F, Atanassova M. Total phenolics and flavonoids in Bulgarian fruits and vegetables. JU Chem. Metal. 2005;40(3):255-60.
- Mansouri A, Embarek G, Kokkalou E, Kefalas P. Phenolic profile and antioxidant activity of the Algerian ripe date palm fruit (Phoenix dactylifera). Food chemistry. 2005 Feb 1;89(3):411-20.
CrossRef - Farooq S, Sehgal A. Antioxidant activity of different forms of green tea: Loose leaf, bagged and matcha. Current Research in Nutrition and Food Science Journal. 2018 Apr 20;6(1):35-40.
CrossRef - Benzie IF, Strain JJ. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: the FRAP assay. Analytical biochemistry. 1996 Jul 15;239(1):70-6.
CrossRef - Oliveira ÉR, Silva RF, Santos PR, Queiroz F. Potential of alternative solvents to extract biologically active compounds from green coffee beans and its residue from the oil industry. Food and bioproducts processing. 2019 May 1;115:47-58.
CrossRef - Haminiuk CW, Plata-Oviedo MS, de Mattos G, Carpes ST, Branco IG. Extraction and quantification of phenolic acids and flavonols from Eugenia pyriformis using different solvents. Journal of Food Science and Technology. 2014 Oct;51:2862-6.
CrossRef - Truong DH, Nguyen DH, Ta NT, Bui AV, Do TH, Nguyen HC. Evaluation of the use of different solvents for phytochemical constituents, antioxidants, and in vitro anti‐inflammatory activities of Severinia buxifolia. Journal of food quality. 2019;2019(1):8178294.
CrossRef - Alara OR, Abdurahman NH, Ukaegbu CI. Extraction of phenolic compounds: A review. Current research in food science. 2021 Jan 1;4:200-14.
CrossRef - Borges A, José H, Homem V, Simões M. Comparison of Techniques and Solvents on the Antimicrobial and Antioxidant Potential of Extracts from Acacia dealbata and Olea europaea. Antibiotics. 2020 Jan 28;9(2):48.
CrossRef - Liu SC, Lin JT, Wang CK, Chen HY, Yang DJ. Antioxidant properties of various solvent extracts from lychee (Litchi chinenesis Sonn.) flowers. Food Chemistry. 2009 May 15;114(2):577-81.
CrossRef - Iloki-Assanga SB, Lewis-Luján LM, Lara-Espinoza CL, Gil-Salido AA, Fernandez-Angulo D, Rubio-Pino JL, Haines DD. Solvent effects on phytochemical constituent profiles and antioxidant activities, using four different extraction formulations for analysis of Bucida buceras L. and Phoradendron californicum. BMC research notes. 2015 Dec;8:1-4.
CrossRef - Maryana E, Saepudin E, Laily N. The potency of chlorogenic acid extract from green coffee beans on inhibition pancreatic lipase activity. InAIP Conference Proceedings 2021 Sep 1 (Vol. 2370, No. 1). AIP Publishing.
CrossRef - Yao L, Jiang Y, Datta N, Singanusong R, Liu X, Duan J, Raymont K, Lisle A, Xu Y. HPLC analyses of flavanols and phenolic acids in the fresh young shoots of tea (Camellia sinensis) grown in Australia. Food Chemistry. 2004 Feb 1;84(2):253-63.
CrossRef - Mehari B, Chandravanshi BS, Redi-Abshiro M, Combrinck S, McCrindle R, Atlabachew M. Polyphenol contents of green coffee beans from different regions of Ethiopia. International Journal of Food Properties. 2021 Jan 1;24(1):17-27.
CrossRef - Kiattisin K, Nantarat T, Leelapornpisid P. Evaluation of antioxidant and anti-tyrosinase activities as well as stability of green and roasted coffee bean extracts from Coffea arabica and Coffea canephora grown in Thailand. Journal of Pharmacognosy and Phytotherapy. 2016 Oct 31;8(10):182-92.
CrossRef - Hao K, Liu X, Wang X, Fei L, Liu L, Jie F, Li Y, Yang Q, Shan Y. Optimizing shade cultivation method and irrigation amount to improve photosynthetic characteristics, bean yield, and quality of coffee in a subtropical monsoon climate. Frontiers in Plant Science. 2022 Apr 29;13:848524.
CrossRef - Li H, Ge Y, Luo Z, Zhou Y, Zhang X, Zhang J, Fu Q. Evaluation of the chemical composition, antioxidant and anti-inflammatory activities of distillate and residue fractions of sweet basil essential oil. Journal of Food Science and Technology. 2017 Jun; 54:1882-90.
CrossRef - Ghanimi R, Ouhammou A, El Atki Y, Bouchari ME, Cherkaoui M. The Antioxidant Activities of Ethanolic, Methanolic, Ethyl Acetate, and Aqueous Extracts of the Endemic Species, Lavandula mairei Humbert (A Comparative Study between Cold and Hot Extraction). Ethiopian Journal of Health Sciences. 2022 Nov 1;32(6).
CrossRef - Jeszka-Skowron M, Zgoła-Grześkowiak A, Grześkowiak T. Analytical methods applied for the characterization and the determination of bioactive compounds in coffee. European Food Research and Technology. 2015;240(1):19-31.
CrossRef - Daglia M. Polyphenols as antimicrobial agents. Current Opinion in Biotechnology.2012;23(2):174-181.
CrossRef - Hossain MA, Shah MD, Gnanaraj C, Iqbal M. In-vitro total phenolics, flavonoids contents and antioxidant activity of essential oil, various organic extracts from the leaves of tropical medicinal plant ‘Tetrastigma’ from Sabah. Asian Pacific Journal of Tropical Medicine. 2011;4(9):717-721.
CrossRef - Scalbert A, Manach C, Morand C, Remesy C, Jimenez L. Dietary polyphenols and the prevention of diseases. Critical Reviews in Food Science and Nutrition. 2005;45(3):287-306.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





