Introduction
Food consists of a diverse range of lipids and oils, with fatty acids (FA) being the main components of diet and a source of energy for humans.1 Different types of FA are found in the blood, cells, and tissues of nearly all animals. They play a major role in many biological processes relevant to metabolism, hormone regulation, and many pathways in cells such as β-oxidation to produce energy and to depot fat storage. 2 They also regulate additional crucial activities such as the membrane permeability, transcription factor activity, expression levels of genes, and generation of bioactive lipid mediators.2 FAs are carboxylic acids characterized by the presence of chemical double bonds, saturated or unsaturated. They consist of an even number of carbon chains ranging from two to thirty-six carbon atoms. Polyunsaturated fatty acids (PUFA) are a major group that is structurally constituted of double bonds arranged in a pentadiene configuration.3 The occurrence of FA in the body is tissue and species-specific, for example, the most prevalent FA in animal and plant tissues with sixteen or eighteen carbon atoms, including palmitic, stearic, oleic, and linolenic acids. 4 Selectivity in mammals, FA are twelve to twenty-four carbon atoms long with up to six double bonds. Moreover, FAs longer than 22 carbon atoms or shorter than 14 atoms are present in very small amounts. Generally, saturated fatty acids are predominant in animal fats, whereas PUFA are more abundant in vegetable fats, with the exception of palm and coconut oils.5
In general, saturated fatty acids are predominant in animal fats, whereas polyunsaturated fatty acids are more abundant in vegetable fats, with the exception of palm and coconut oils. Normally, humans ingest 20 different kinds of FA daily with saturated, monounsaturated, or polyunsaturated carbon chains. Among them few are essential fatty acids (EFA) such as Omega-3 FAs (n−3 FAs) and Omega-6 FAs (n−3 FAs) are not synthesized within the human body and therefore need to be incorporated into the diet.6
n−3 FAs have unsaturated hydrocarbon chains with a double bond at the third and fourth positions, whereas in n−3 FAs, the double bond is situated at the carbon at position six.7 The carboxyl end is where most chemical changes take place, while the methyl group and its adjacent double bond, such as in n-3 and n-6, remain unchanged during physiological transitions in the body. n−3 FAs are generally PUFA, with cis double bonds such as ALA, EPA, and DHA, are classified as functional foods.8 These FAs are found in a wide range of diet items. ALA is present in plant-based items like soybeans, flaxseed, chia seeds, and oils from canola. Alternatively, DHA and EPA, are mostly found in marine-based foods such as oily fish. Intake of long-chain n−3 FAs in humans is preferred because the biological conversion of ALA to EPA and DHA is either incomplete or extremely little in vivo. 9 Since the human body cannot produce ALA, it must be obtained from foods or supplements. n−3 FAs play a major role in cell membrane fluidity. Levels of DHA are particularly high in sperm, brain, and retinal (eye) cells. In addition, n−3 FAs play a regulatory role in numerous functions within the various organs like the heart and lungs; blood vessels; and immune and endocrine systems.10,11
In recent years, there has been a notable gain in the market for fish oil-based food supplements, driven by the expectation of gaining similar health benefits as those derived from consuming fish. The global n-3 supplement market, valued at USD 5.18 billion in 2019, is projected to witness a compound annual growth rate of 8.4% from 2020 to 2027.12 Despite the substantial turnover in this market, there is a noticeable scarcity of scientific studies dedicated to assessing the quality of these products.13 Ensuring the quality of products containing n-3 FAs is a critical step in the manufacturing process. This is typically achieved through the implementation of quantitative chemical tests to verify compliance with international standards.14 Ensuring the effectiveness and safety of n-3 supplements relies on crucial quality control measures, primarily focusing on the absence of heavy metals (such as mercury) and trace metals (sodium, potassium, magnesium, iron, and calcium), as well as assessing the quality and quantity of docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA). 15,16 Numerous projects are underway to scrutinize the quality of commercial omega-3 fish oil brands. These assessments encompass the examination of key quality attributes, including uniformity of weight, disintegration time, and DHA and EPA content. Additionally, they involve monitoring the presence of sodium, potassium, calcium, iron, magnesium, and mercury, even when not explicitly stated on product labels. Through risk assessment, particularly using the hazard quotient, it has been revealed that consumers using these products over an extended period may be at risk of potential adverse health effects.17 The findings of these studies underscore significant concerns regarding the quality and safety of commercially available omega-3 supplements. This emphasizes the critical necessity for enhanced regulatory measures and increased consumer awareness to address and rectify these issues.
In this review, we have highlighted the general properties and manufacturing process for n-3 FAs namely chemistry, dietary sources, extraction, and encapsulating techniques. Further, we also compiled the major literature evidence relevant to the dosage, quality, and health advantages of n−3 FAs. Collectively, we attempted to leverage the potential risks associated with the use of these supplements while providing some recommendations for regulations sought for these dietary aids. We conducted a thorough search of research databases, including Pubmed, Medline, and Google Scholar, to gather peer-reviewed articles for writing this article.
Chemistry
Structurally, n−3 FAs contain double bond at the carbon three from the methyl (-CH3) end, called as omega where the alpha end consists of carboxylic acid functional group (-COOH) shown in Figure 1. Its chemical formula is C60H92O6 with a molecular weight of 909.39 g/mol. Some of the most common n−3 FAs are α-linolenic acid (ALA; 18:3 n-3), stearidonic acid (SDA; 18:4 n-3), eicosapentaenoic acid (EPA;20:5 n-3), docosapentaenoic acid (DPA; 22:5 n-3), and docosahexaenoic acid (DHA; 22:6 n-3). Specifically, the three most important in terms of human physiology are ALA (all-cis-9,12,15-octadecatrienoic acid), EPA (all-cis-5,8,11,14,17-eicosapentaenoic acid) DHA (all-cis-4,7,10,13,16,19-docosahexaenoic acid) with eighteen, twenty , and twenty two carbon atoms with three, five , or six double bonds, respectively. Of note, all double bonds are in the cis-configuration, meaning that the two hydrogen atoms are on the same side of the double bond. Methylene bridges (-CH2) also act as double bond breaks, leaving two single bonds between each pair of adjacent double bonds.7 ALA is the parent n-3 FA which desaturases and lengthens to produce n−3 PUFAs. Due to the limitations of the enzyme delta-12 and delta-15 desaturase that inserts cis double bonds at carbon-carbon positions 12 and 15, n−3 PUFAs is not synthesised in body. ALA is a necessary 18-carbon unsaturated FA that undergoes both chain elongation and desaturation processes to yield EPA or DPA.18
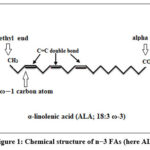 |
Figure 1: Chemical structure of n−3 FAs (here ALA) |
Natural n−3 FAs sources
Major dietary source of n−3 FAs comes from aquatic organisms especially the liver of cod and halibut, and the flesh from mackerel, menhaden, and salmon.19 Moreover, essential FAs are present in seeds like flaxseed and chia seeds, as well as nuts, particularly walnuts. Plant oils, including flaxseed oil 20, soybean oil 21, and canola oil 22, are also rich sources of these beneficial FAs. Additionally, fortified foods like specific brands of eggs, yogurt, juices, and milk provide sufficient amounts of these essential nutrients. 23 (Figure 2).
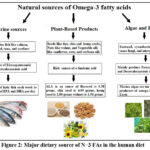 |
Figure 2: Major dietary source of N−3 FAs in the human diet. |
Marine sources
Aquatic organisms are as a valuable reservoir of PUFA, notably n-3 FA, which the human body does not naturally produce.24 EPA and DHA are the two key n−3 PUFAs derived from marine sources, whereas DPA is found at relatively low concentrations. Cold-water fish like salmon, mackerel, tuna, and sardines are rich sources of natural n−3 FAs. In comparison to freshwater fish, marine fish contain high amounts of MUFA and PUFA. 25.26, Furthermore, cod flesh, halibut, and skipjack tuna are identified as having a substantial concentration of DHA, while flounder species and haddock exhibit a noteworthy level of EPA. Additionally, n−3 PUFA can be found in crustaceans, bivalves, and cephalopods. When compared to other cooked fish, salted mackerel had the highest concentration of EPA and DHA. Joint FAO/WHO Expert recommended having 2-3 servings of fatty fish each week to attain 250 mg of EPA and DHA per day.27
Plant-Based Products
Plants are the main source of ALA, mostly present in seeds, nuts, and vegetable oils.28 Moreover, flaxseed, chia, and echium seed oils in addition to walnuts as excellent sources of ALA. On the other hand, safflower, sunflower, corn, and soybean oils boast high linoleic acid content. A single ounce of flaxseed, chia seeds, hemp seeds, or walnuts supplies a sufficient quantity of ALA, meeting the recommended intake of 1.1 g/day for females and 1.6 g/day for males. In an ounce, flaxseed has 6.38 grams, chia seeds have 4.99 grams, hemp seed has 2.80 grams and walnuts have 2.54 grams of ALA, respectively.28,29
Microalgae, Seaweeds (Macroalgae), and Fungi
As a nutritional supplement, seaweed is becoming more and more popular all over the world in cookery.30 Seaweed stands out as a functional food, offering a rich source of n3-PUFAs, protein, and minerals.31 Notably, commercially available biomass extracts like Spirulina and Chlorella, derived from cyanobacteria and green algae, serve as excellent suppliers of n3-PUFAs. Furthermore, certain fungi and microalgae also contain n-3 PUFAs.32 According to Senanayake and Fichtali 33 marine algae play a major role as the primary producers of long-chain -3 PUFA on Earth. DHA, a significant component, is found in various algae types such as Crypthecodinium cohnii and Schizochytrium spp., with the latter contributing to 55% and the former to 40% of the total FAs. Additionally, phytoplankton and algae generate -3 PUFAs, particularly EPA and DHA, which are then transferred through the food chain and accumulated in the lipids of fish and marine animals.34
Commercial production of n−3 FAs
Just 5% of the global fish oil production is dedicated to extracting n−3 FAs for use in diet supplements.35 The remaining fractions that are produced are fed to animals. However, in the future, fishmeal and oils will be used in diets for aquaculture at lower levels, leading to rise in the availability of these crucial essential FAs for human consumption. The quality and composition of FAs isolated from any source will likely depend on the environmental factors and the implemented extraction techniques.36
Traditional solvent Extraction
Various techniques are being used to extract n−3 FAs. Whole fish or tissue that is rich in lipids is used to extract oils through traditional solvent-based techniques using an organic solvent with a mixture of methanol and chloroform with a ratio of 2:1. This technique can isolate both polar and non-polar oils from the tissue. At first, tissue is ground with the solvent followed by the addition of water to generate two distinct layers. Polar and nonpolar lipids are collected at the bottom chloroform layer and the residue is collected from the top methanolic and water layers. Finally, chloroform is evaporated leaving the lipids behind. This technique is mostly used to extract n−3 PUFAs from fish liver and muscle tissue.37, 38
Conventional Extraction
Usually, the wet pressing method is used to extract omega-3 fish oil. The process involves cooking the fish initially, followed by pressing, decanting, and centrifuging to separate the fatty and aqueous components. The collected oil fraction, rich in n−3 FAs, undergoes refinement stages, including neutralization, bleaching, degumming or water refining, winterization, and deodorization.39 Quality of oil is maintained by reducing oil acidity, absorbing impurities or pollutants, separating phospholipids, and removing odorous. Finally, the yield of the n−3 FAs is increased by the molecular distillation process.35
Green extraction method
Although the yield of n−3 FAs with standard extraction techniques is acceptable, it has several disadvantages e.g. harmful organic solvent residues may persist in the last fraction, high temperatures that can destroy the heat-sensitive n−3 PUFAs and lastly environmental pollution caused by organic solvents and time-consuming procedures.40-43 To ensure the purity and stability of these foodstuffs, more mild, secure, and ecologically friendly green chemical extraction techniques are being developed by industries. These methods are based on the idea of “green” extraction utilizing supercritical fluid, enzyme, microwave, pulsed electric fields, and ultrasound technology.40,42 Supercritical fluid extraction is widely employed for extracting n−3 FAs due to its economic production of high-purity oils with substantial yields, all achieved without elevated temperatures or organic solvents.44-46
Biotechnological approach for synthesizing n-3 fatty acids.
As a consequence of drawbacks associated with fish oil production, there is a continual quest for alternative 47 and sustainable sources of n‐3 FAs like engineering plants to produce n-3 FAs in their seed.48 However, this method requires months for harvesting and purifying the n-3 FA from the plant oil. Although there has been notable advancement in the biotechnological production of n-3 FA, significant challenges persist in overcoming the current supply shortage to meet the growing demand for this product. While genetic manipulation techniques in plants have led to the development of transgenic plants as potential sources of n‐3 FAs, these processes are both expensive and labor-intensive when implemented on a large scale.49
Some cost-effective, time-efficient, environmentally friendly alternatives for n‐3 FAs synthesis are being developed like industrial extraction of n‐3 FAs from microalgae and other potential oleaginous microorganisms.50 These microbial oils, also known as single-cell oils are produced by unicellular microorganisms, constituting 20–80% lipid per dry biomass. The production of single-cell oils is enhanced when organisms are grown at lower temperatures, promoting the process of fatty acid synthesis. Precision fermentation utilizes microorganisms, such as yeast or bacteria, as a production host to create specific molecules of interest. This approach holds promise for the animal-free production of PUFA. Oleaginous yeast, like Yarrowia lipolytica, is suitable candidate for large-scale fermentation process to synthesizing EPA due to its ability to accumulate significant amounts of intracellular lipids and utilize sugars from agricultural feedstock.50
The use of several microbes as cell factories for n-3 FAs production presents an avenue for improvement. Enhancing biosynthesis by optimizing carbon yield, balancing cofactors, and minimizing by-product formation holds the promise of elevating product quality and reducing bio-manufacturing costs. Exploring alternative carbon sources, such as economical sugars, waste oils/fats, offers an opportunity for increased product yield and cost efficiency. Advanced molecular biology tools can be leveraged to refine metabolic engineering, thereby boosting the yield of bio-manufactured n-3 FA. Additionally, cutting-edge technologies like continuous fermentation and AI-controlled fermentation can enhance downstream process engineering, increasing productivity while lowering operational and capital expenses. Collectively, these integrated strategies present a comprehensive roadmap for efficiently, cost-effectively, and sustainably elevating the production of omega-3 fatty acids.47
Encapsulation system of omega-3 FA
n−3 FAs are biologically active molecules and very prone to oxidation because of unsaturation in the long chain. The rate of lipid oxidation depends on the degree of unsaturation; thus DHA/EPA are more vulnerable to oxidation. Primary oxidation produces hydroperoxides and then subsequently lipid peroxides, which further generate a lot of free radicals (aldehydes/ketones).51 These oxidation products are toxic it has negligible biological activity.52 For instance, 4-hydroxyalkyl, one of the oxidized products of n−3 PUFAs, is toxic to the brain and retina.53 Moreover, certain oxidized end products of n−3 PUFAs pose a risk of atherosclerosis, induce inflammation through the activation of pro-inflammatory molecules, and adversely impact the body’s cholesterol levels.54 To address these concerns, various encapsulation systems, including gels, emulsions, and powders, have been developed and innovated. These systems aim to encapsulate these FAs so as to enhance their stability, dispensability, and bioavailability. (Figure 3).
Liposomes
Liposomes are extensively used in encapsulating n−3 PUFAs with a double-layer vesicle which usually consists of phospholipids arranged in concentric rings.55,56 In terms of structure, liposomes encompass both polar and non-polar regions, allowing them to encapsulate substances that are both hydrophilic and hydrophobic. Liposomes are prepared and assembled through solvent evaporation, injection, and micro fluidization techniques each of which has a different commercial potential.57 Liposomes enhance the oxidative stability of FAs while being stored however the high cost of the phospholipids and the low physical stability of liposomes due to fragile phospholipid membranes in complex matrices are some drawbacks of this technology.58 To increase the stability of liposomes, phospholipid bilayers have been coated with wall material mainly for the pharmaceutical and food industries. Nanoliposomes is thought to be a useful technique for stabilizing and increasing the bioavailability of bioactive substances that are encapsulated.59 Consequently, a variety of lipid-based nanocarriers, including nanoliposomes, were created by some researches to boost the bioavailability of n−3 FA. 60,61 Soy lecithin is used as a wall material for EPA and DHA nanoliposomes with increased encapsulation efficiency to 100%.62 Researchers have reported β-sitosterol63 and Poly-D, L-lactide-co-glycolide chitosan64 for DHA nanoliposomes where chitosan/gelatine (CH/GE)65 was attempted for n−3 PUFAs nanoliposomes with increased encapsulation efficiency. Recently a thin-film hydration was used to encapsulate chia oil nanoparticles with 88.31% efficiency.66 The encapsulated nanoliposome exhibited improved thermal stability and a prolonged release of n-3 FAs. Hence can be used as a supplement for both dietary and medicinal purpose.66
Emulsions
Emulsification is the process of combining two immiscible liquids with a smaller droplet of one liquid dispersed in the other liquid.67, 68 Specifically, hydrophobic bioactive substances are encapsulated using traditional single-layer interface emulsions using water-in-oil and oil-in-water techniques. The encapsulation system used for n−3 PUFAs is usually either oil-in-water emulsion or nano-emulsion due to their hydrophobic nature. In emulsions, the average droplet diameter exceeds 100 nm, while it is less than 100 nm for nanoemulsions.69 The use of nanoemulsions significantly enhances the capability and stability of the orally ingested n-3 oil. In this procedure, n-3 oil and water are homogenized with high-shear mixers, colloid mills, high-pressure valve homogenizers, microfluidizers, and sonicators in the presence of an emulsifier.69 The emulsion acts as a physical barrier for reactive elements and protects n−3 PUFAs from oxidation.70 The stabilization of the interface layer through emulsification can help alleviate the effects of adverse environmental factors on the oxidation of n−3 PUFAs. To address health risk concerns, chemical and synthetic emulsifiers are currently being avoided and substituted with natural interface stabilizers, including proteins, polysaccharides, and phospholipids. Recently, the stability of n−3 PUFAs-rich emulsions was examined using two milk proteins, namely sodium casein and whey protein isolate. Both proteins create an interface on the oil droplet film, resisting droplet breakage through robust repulsive interactions.71 Furthermore, the stabilizing capabilities of hydrolysed whey protein (WPH), blue cod protein, and soy protein were also evaluated for n−3 PUFAs emulsions.72 Recently Jagtap et al 73 has created an innovative formulation for the stabilized n-3 FA fortification and micronutrient enrichment with flaxseed oil, water, and sucrose ester (an emulsifier) 1. Important minerals, antioxidants, and plant-based n-3 FA were all included in the designed formulation. This product has the potential to improve the nutritional quality and bioavailability of n-3 FA in food and nutraceutical industries 73. Zinc, vitamin E, magnesium, vitamin B3 and vitamin B6 were added by considering their metabolic effects1. The bioavailability of n−3 is significantly influenced by micronutrients and co-factors.74-76. Fundamental aspects of emulsion formula and their subsequent use in different industries are being assessed in order to improve product quality and its potential for industrial applications 77
Microencapsulation
Microencapsulation is a process of converting an emulsion to powder form through drying and subsequently encapsulation to form a solid functional ingredient to improve its handling, storage, or stability.78,79 Normally, oil containing n−3 PUFAs is emulsified with water and then dissolved in materials used for microencapsulation. The mixture is then dehydrated to powder form as a microcapsule of oil droplets surrounded by microencapsulated material.80 The choice of wall material relies on factors such as packing, encapsulation efficiency, and chemical stability. It must be water-soluble and capable of forming a low-viscosity fluid, facilitating pumping during processes like spray drying or electro-spraying. Additionally, it should hinder gas diffusion and unwanted chemical reactions, particularly oxidation, and possess a suitable glass transition temperature. This temperature is crucial as the material is rigid and brittle below this point and soft and rubbery above it. Recently, Solomando et al 81 used chitosan and maltodextrine as a wall material to created fish oil microcapsules to be added in various commercial meat products. These microcapsules enhanced the amount of EPA and DHA in cooked and dry-cured meat products without changing the physico-chemical properties or oxidative stability of n-3 FA.
Spray drying
The most common method employed for the industrial-scale microencapsulation of n−3 PUFAs is spray drying.82 This process commences with the homogenization of oil-in-water to create an emulsion, which is then blended with wall materials such as casein, sugar beet pectin, lactose, dextrose, maltodextrin, starch, glucose syrup, gum Arabic and gelatine.83,84 Subsequently, the mixture is pumped through a slender nozzle into a chamber that is heated, generating small droplets that swiftly undergo drying and are ultimately collected in powder form. The average diameters and morphology of the spray-dried powder had an impact on the stabilities n-3 FA. One of the crucial element in determining the surface-to-oil ratio in the spray-dried powder is the ratio of the oil droplet’s diameter to the particle’s diameter. Recently, Sultana et al 85 employs a kinetic model featuring fundamental mass transfer phenomena and chemical reactions to examine the stability of PUFAs in spray-dried powder. Degradation or oxidation kinetic constant and mechanism number can be used in the Avrami equation to evaluate the stability of n-3 FA in powder form 85. Some of the major disadvantages of this method are the use of high temperatures and chemicals which can degrade n−3 PUFAs and reduce the nutrients. Also, the drying of the emulsion from the outside may leave the final product with some internal humidity.
Microfluidic jet spray drying technology
This technique produces microcapsules with uniform and larger particles of n−3 PUFAs emulsions. The method involves the preparation of whey protein isolate microcapsules of stabilized DHA oil, yielding uniform and consistent droplets. This is achieved using an airflow rate of 15 L/min at 160◦C through a microfluidic aerosol nozzle with a diameter of 75 μm. Whey protein isolate serves to shield DHA from elevated temperatures by creating a heat-induced cross-linked network over the oil droplet surface during the spray drying process 86 The electrospray technology based on the electrohydrodynamics of the solution and dispersed materials, is being used to produce n-3 PUFAs microcapsules in low temperature to decrease the impact of hot air flow on these particles . García-Moreno et al., 87 used charged polymer carriers for the n−3 PUFAs and sprays them onto the opposing electrode in the electric field to disperse into smaller droplets polymer forms of n−3 PUFAs microcapsules after solvent evaporates.
Electro-Spray and Electro-Spin technology
Derived from the electrohydrodynamics of the solution, the electrospray technology uses low temperatures to prepare n−3 PUFAs microcapsules to reduce the effect of flow of heated air.on n−3 PUFAs. This method is considered a mild technique and is more suitable for n−3 PUFAs due to their unstable functional materials. n−3 PUFAs are carried by a charged polymer and sprayed in the electric field towards the other electrode to disperse into smaller droplets. During this process, the n−3 PUFAs disperse into smaller droplets while transitioning from one electrode to another, resulting in the formation of n−3 PUFA microcapsules. These microcapsules can take the form of particles (through electro-spraying) or fibers (via electro-spinning), depending on the specific operating conditions.88-90 Rahmani-Manglano et al 91 used low molecular weight carbohydrates as encapsulating agents during the monoaxial or coaxial electrospraying and spray-drying processes to create the n-3 FA capsules. Electrospraying created encapsulates were reported with lower retention qualities as compared to spray-drying method with higher encapsulation efficiency.91
Freeze Drying
This method converts omega-3 oil into a powder by freezing it at -90 to -40 °C followed by dehydrating it under a vacuum.92 This method is impracticable than spray drying for the reasons that it is costly, time taking, and providing less efficient outputs.93
Extrusion
This technology uses extrusion to convert omega-3 oils into solids. The molten wall material is mixed with emulsified oil subjected to elevated pressure and then passed through a slender nozzle to produce hardened microcapsules.94 Although this technique produce commercial microcapsules on a large scale, the use of high temperatures promotes the oxidation of the fish oil during the manufacturing process with significant capital and energy expenses.95
 |
Figure 3: Encapsulation systems designed to encapsulate n−3 PUFAs to overcome oxidation and deterioration during storage. |
Quality System uses to maintain omega 3 supplements for commercial use
Quality control and the pharmaceutical ingredients of dietary omega-3 supplements have been well defined by pharmacopeia monographs and typically address the fatty-acid composition, oxidation states, and controlling the presence of any environmental pollutants.96 n−3 FAs available these days are either non-prescription (e.g., fish oils) or prescription (Rx) medications that are not necessary the equivalent on the basis of their contents.97 The United States Food and Drug Administration (USFDA) oversees the production of prescription pharmaceuticals and sets criteria for consistency and quality control.98,99 It is crucial for both consumers and medical professionals to verify the qualitative and quantitative differences between FDA-approved prescription formulations and the dietary supplements that are commercially available.
Oxidative status
n−3 FAs are among the best labile supplements due to high susceptibility to oxidation and deterioration over time during storage.100 Exposure of this FA to oxygen in the air can lead to rancidity, deterioration issues, and change of taste and odour.101 Upon prolonged storage, the nutritional value of the supplement decreases with the formation of unhealthy components such as 4‐hydroxy‐2‐alkenal and 4‐hydroxy‐2‐hexanal.102 Some of the implemented precautionary procedures to prevent this kind of spoilage are to store the supplement at possible low temperatures, to encapsulate in safe polymer matrices, and to protect it from light.103 This supplement is commonly available as a soft gel capsule that are mixed with antioxidants in order to reduce lipid oxidation. In addition, these supplements are manufactured using specialized techniques to assist in controlling the oxidation process, such as employing fresh raw ingredients of high quality and a low-temperature extraction method. Specifically, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), tert-butyl hydroquinone (tBHQ), propyl gallate, tocopherol, and ascorbyl palmitate are among the antioxidants commonly used in theses supplements to reduce the oxidation process. Such antioxidants can reduce the oxidation process but will likely not prevent it while the speed of oxidization is mostly depending on the composition of FAs, oxygen/ light contact, temperature, and the presence of water and heavy metals. The oxidation process in these FAs significantly affects the composition and produces lipid hydroperoxides as the primary oxidation by-product, which then degrades into secondary oxidation by-products such as aldehydes, ketones, and alcohols.104,105 The level of secondary oxidation is determined by the para-anisidine value (AV), which provides a measurement of aldehydic compounds (predominantly 2-alkenes and 2,4-alkadienals) while hydroperoxide levels are determined by the peroxide value (PV) in the oxidized oil. The quality of n−3 FAs is determined by calculating the total oxidation (TOTOX) levels by measuring both PV and AV (primary and secondary oxidation levels.106 Although the maximum recommended levels for TOTOX for this supplement have been set by many organizations these standards are based on palatability rather than health.107
Actual versus claimed content
EPA and DHA quantity and oxidative status in n−3 FAs supplements are the main indicator of their quality.108 Many studies have reported the presence of a significant amount of saturated fat in commercially available N−3 FAs capsules which do not comply with the label.104,109-115 The main reason may be the lack of a proper purification process for removing the impurities originally present in the fish oil. Many countries have extensively examined the composition of n−3 FAs supplements available in their market. Some samples from the US did not meet the stated label claim of EPA or DHA.114,115 Furthermore, samples from Poland showed lower levels of FA content compared to the claim stated in the label.113 Moreover, Brazil reported large amounts of low-cost soybean oil added in EPA and DHA supplements.109 Another study from New Zealand has found that only 3 out of 32 samples contained the exact or surplus amount of the label claim in terms of EPA and DHA.116 Alghamdi and co-workers have recently analyzed the commercially available Omega-3 Supplements in three Arab Gulf Countries. Samples from the market of Saudi Arabia, Bahrain and Kuwait were measured for EPA and DHA and did not match the stated label amount.117 The main reason was attributed to the negligence in monitoring for the quality of fish oil supplements by the manufacturers or companies. Preparation of omega-3 supplements involves impeccable purification levels to remove contaminations like free FAs, heavy metals, and coloured compounds present in the raw material. Accurate purification steps are potentially expensive, and are circumvented by the companies. To that end, the incompliance evidence observed in the literature emphasizes the importance of the post-market surveillance programs conducted by regulators in order to ensure the integrity for those supplements and to provide any corrections/notifications plans to manufacturers.
Harmful contaminants
Omega 3 supplements are mainly extracted from marine organisms. These organisms are exposed to the dangerous contaminants present in water such as heavy metals and residual pesticides. Bio-accumulation of these harmful chemicals poses a higher risk of toxins in larger fish types. In fish oil harvested for such organisms, toxins in trace amounts remain in the final products.118 Some of the toxins found in these products include lead, mercury arsenic, cadmium, dieldrin, dichlorodiphenyltrichloroethane, selenium, and polychlorinated biphenyls.118 Although studies have not reported any significant amount of toxins in these supplements, prolonged exposure to it can result in enduring health complications.119
Encapsulation
Encapsulating the oil supplement is one of the best ways to protect it from atmospheric oxygen to avoid oxidation.117 Rancid odor and flavor are not detected in capsulated samples as compared to bottle-stored oil supplements due to the protective effect of packaging against oxidation. It is recommended to encapsulate omega-3 oils in polymer matrices such as Sunflower phospholipids, whey protein concentrate, maltodextrin, β-lactoglobulin, fibrils, and chitosan. Omega-3 products are commonly available in capsules, chewables, and syrups. Syrup forms are more susceptible to oxidation than capsules as capsules are better protected during the storage period. Common materials used to encapsulate n-3 oils are mentioned in section 5 of the present article.
Medical use, dosage, and risk factors
Currently, the US Food and Drug Administration (FDA) approved two types of prescription n-3 FA medications, one containing both EPA and DHA and with EPA only. Prescription n-3 FA medications are approved only after ample clinical safety and efficacy studies. Available n-3 FA products are icosapent ethyl (the ethyl ester of EPA) contains only EPA, n-3-acid ethyl esters containing both ethyl esters of EPA and DHA, n-3-carboxylic acids contain free FAs mainly EPA and DHA.120 FDA has approved n-3 FA medications only for the treatment of hypertriglyceridemia with a dose of 2–4 g/day depending on the severity of the disease.112,113 Currently, a lot of research on the possible role of this medication in treating conditions and ailments such as cardiovascular disease, type 2 diabetes, cancer depression, brain development, rheumatoid arthritis, asthma, diabetic retinopathy, and non-alcoholic fatty liver disease is at the peak.8,121-125 These medications are generally safe with mild side effects such as eructation, stomachache, diarrhea, nausea, and joint pain.126,127 In some extreme cases adverse reactions such as arthralgia and oropharyngeal pain, increased ALT/AST, pruritus, and rash are also reported.126-128 Prescription omega-3 FA medications should not be confused with dietary supplements of n−3 FAs which do not require FDA approval and don’t need any safety trial or efficacy before marketing. n-3 FA dietary supplements are available without prescription with lower concentrations of EPA and DHA and are not designed to treat any disease.120 Such supplements don’t need FDA approval, hence EPA and DHA levels might differ widely between different brands and products and are derived from different sources mainly from fish and other marine organisms. Studies have reported the therapeutic and protective role of these supplements against hypertension,129 coronary heart diseases,130 atherosclerosis,131 memory loss, and neurodegenerative disorders such as autism spectrum disorder (ASD), Alzheimer’s and Parkinson’s diseases.132-134 EPA and DHA supplements decrease oxidative stress due to their anti-inflammatory and antioxidant properties.135,136 Mild side effects like unpleasant taste, bad breath, bad-smelling sweat, headache, and gastrointestinal symptoms are reported. However, patients on anticoagulants or nonsteroidal anti-inflammatory drugs should consult a physician before using them as omega-3 dietary supplements may prolong the time for bleeding to stop after a wound. 137,138
Regulation of Omega-3 at Representative Jurisdictions
Globally, counties have different regulatory requirements for licensing and registration of omega-3 products. In addition, the specifications for the quality and safety of those products may vary according to the laboratory testing requested by a given regulatory authority. Consequently, these two factors may alter the regulatory process for importing countries as well as it could hinder consumer access to such products. We will highlight three representative examples of leading regulatory authorities in this area namely the Health Science Authority (HSA, Singapore), Health Canada (Canada), and Therapeutic Goods Administration (TGA, Australia). HSA, Health Canada, and TGA are classifying omega-3 supplements as health supplements, natural health products, and complementary medicines, respectively.129,132 According to Health Canada, health supplements are subjected to product licensing to be legally sold in Canada.140 Also, TGA requires health supplements to be registered in the Australian Register of Therapeutic Goods (ARTG).142,143 However, HSA does not require licensing and it is the dealers’ option to notify HSA of supplying their health supplements in Singapore.139
Regarding the quality, safety/efficacy, claims requirements and restrictions, authorities necessitate stringent requirements for manufacturers. Table 1 – summarize authorities’ criteria for the aforementioned elements to guarantee omega-3 products compliance to regulations set by each country.
Table 1: Summary of Each Authoritie’s Criteria for Quality, Safety/Efficacy and Claims.139-143
|
Requirements Aspect |
Regulatory Authority Criteria |
||
|
HSA |
Health Canada |
TGA |
|
|
Quality |
Stability and longevity of the product. Conditions for storage and distribution. Manufacturing, packaging, and assembly environments |
Precise labeling and packaging specifications. Adherence to Good Manufacturing Practices (GMP). |
All complementary medicines must adhere to the principles of Good Manufacturing Practice (GMP). Must undergo a full TGA pre-market evaluation of quality, safety, and efficacy by the TGA, if at least one of the following applies: · The products contain ingredients beyond those allowed for use in listed medicines or assessed listed medicines. · They carry higher-level indications not permitted for use in listed or assessed listed medicines. · They are required to be sterile |
|
Safety and Efficacy |
Prohibited substances in the health supplement (e.g., ingredients sourced from human parts, undisclosed active substances, active ingredients known to possess inherent pharmacological properties). The product should adhere to specified heavy metal limits (As 5ppm, Cd: 0.3 ppm, Pb: 10ppm, Hg: 0.5ppm). The product should comply with designated microbial limits: (total aerobic microbial count: NMT 105, yeast and mold: NMT 5×102, absence of Escherichia coli, salmonellae, and staphylococcus aureus). |
Applicants are required to furnish comprehensive information to the authority about the product, encompassing details on medicinal ingredients, their source, dosage, potency, non-medicinal ingredients, and recommended use Substantiating the safety and efficacy of the product requires valid evidence, such as clinical trial data, references to published studies, journals, pharmacopeias, and traditional sources. |
|
|
Claims |
Products should not be labeled, advertised, or promoted for any explicit medicinal intent, including claims that imply the treatment or prevention of any disease, disorder, or related conditions |
While proper evidence is necessary, the specific type and quantity of supporting evidence depend on both the proposed health claim of the product and its overall associated risks. |
Registered complementary medicines have the option to include a ‘TGA assessed claim’ on their label and other advertising indicating the TGA has assessed the evidence for the medicine’s indications. |
Quality control of Omega-3 supplements
Quality control for manufacturing finished products containing n−3 FAs is crucial process, which is typically achieved through performing quantitative chemical tests to ensure compliance to the pharmacopeias and/or international standards. Generally, there are a list of chemical tests required to assessment quality of products which fall under three main categories. Firstly, the assay of active ingredients (EPA & DHA) and the total N−3 FAs . Secondly, oxidation tests which can be measured and calculated by Peroxide value (PV) and Anisidine value (AV). Thirdly, the environmental contaminants particularly polychlorinated biphenyls (PCBs), dioxin (e.g. PCBs) and heavy metals tests.144
The determination and quantification of EPA, DHA and total n−3 FAs are heavily depending on source and type of dosage form of active substances (i.e. ethyl ester, triglyceride, free FA, or phospholipids). This fact is clearly shown through observing that fish and krill oil are characterized by a high EPA content, while DHA content is higher in algae oil.117 Monitoring adherence to declared EPA and DHA levels on product labels is essential, ensuring compliance with regulatory thresholds. In New Zealand and Australia, for instance, regulations stipulate that a product must possess a minimum of 92% of the claimed content.. In contrary, in the USA where n -FAs are classified as natural products, their assay need to be at least 80% of the label claim. In addition, in the EU, a 20% tolerance for the nutrient is considered acceptable.135 Discrepancies in the regulations between different jurisdictions might explain the regulatory violations in terms of incompliance to the label claim of FAs mentioned earlier in this review.113-115,117 Consequently, establishing global harmonization and/or common agreement regards the acceptable label claim for those supplements is demanded.
According to different pharmacopeias, the oxidation tests should be performed to satisfy the good quality of products containing n−3 FAs . For example, in British Pharmacopeia (BP) and European pharmacopeia (Ph.Eur), the acid value (AV) acceptable limit is NMT 2.0 while in the United States Pharmacopeia (USP) the limit is NMT 3.0.146,148 Moreover, the maximum limit of oxidations test of Anisidine value (AV), Peroxide value (PV) and Total oxidation value (TOTOX) is variable in different pharmacopoeias. Specifically, in the USP and BP, the peroxide value (PV) limit is NMT 10 mEq/Kg while in Health Canada and Australian TGA guidelines the maximum limit must be 5 mEq/Kg.149,150 Likewise, the Anisidine value (AV) in BP the maximum limit is 20.0 while in USP it is stated to be NMT 25.0. The slight variations within regulations might alter the quality attributes for those supplements and hence consumers could potentially affect particularly products authorized for prescription use.
Conclusion
n−3FAs supplements are manufactured using multiple industrial techniques with varied ingredients. In addition, such supplements are most sensitive to oxidation and can deteriorate if stored for a long period. Its quality mainly depends on its total oxidative level and concentration of both EPA and DHA. Some preventative measures include storing the supplement at a low temperature, encapsulating it in secure polymer matrices, and protection against direct sunlight. There are some discrepancies among the quality control tests and the regulations pertaining to the marketing of these supplements, which perhaps raise the flag for the need for global regulatory harmonization. While compliance criteria may differ between different jurisdictions, consumers and healthcare professionals should verify the qualitative and quantitative attributes of n-3 FA dietary supplements before consumption.
Disclaimer
The views expressed in this paper are those of the author(s) and do not necessarily reflect those of the SFDA or its stakeholders. Guaranteeing the accuracy and validity of the data is the sole responsibility of the research team.
Authors’ Contribution
Conceptualization, FSD. Writing—original draft preparation, RSB. writing—review and editing, ASA; FSB. Visualization, RSB. Funding acquisition FSD. All authors have read and agreed to the published version of the manuscript.
Conflict of Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper
Funding Sources
There is no funding sources.
Data Availability Statement
This statement does not apply to this article.
Ethics statement
This research did not involve human participants, animal, subjects, or any material that requires ethical approval.
References
- Meijaard E, Abrams JF, Slavin JL, Sheil D. Dietary Fats, Human Nutrition and the Environment: Balance and Sustainability. Front Nutr. 2022;9. doi:10.3389/fnut.2022.878644
CrossRef - Calder PC. Functional Roles of Fatty Acids and Their Effects on Human Health. Journal of Parenteral and Enteral Nutrition. 2015;39(1_suppl):18S-32S. doi:10.1177/0148607115595980
CrossRef - Mariamenatu AH, Abdu EM. Overconsumption of Omega-6 Polyunsaturated Fatty Acids (PUFAs) versus Deficiency of Omega-3 PUFAs in Modern-Day Diets: The Disturbing Factor for Their “Balanced Antagonistic Metabolic Functions” in the Human Body. J Lipids. 2021;2021:1-15. doi:10.1155/2021/8848161
CrossRef - Shramko VS, Polonskaya Y V., Kashtanova E V., Stakhneva EM, Ragino YI. The Short Overview on the Relevance of Fatty Acids for Human Cardiovascular Disorders. Biomolecules. 2020;10(8):1127. doi:10.3390/biom10081127
CrossRef - Tvrzicka E, Kremmyda LS, Stankova B, Zak A. FATTY ACIDS AS BIOCOMPOUNDS: THEIR ROLE IN HUMAN METABOLISM, HEALTH AND DISEASE – A REVIEW. PART 1: CLASSIFICATION, DIETARY SOURCES AND BIOLOGICAL FUNCTIONS. Biomedical Papers. 2011;155(2):117-130. doi:10.5507/bp.2011.038
CrossRef - Kaur N, Chugh V, Gupta AK. Essential fatty acids as functional components of foods- a review. J Food Sci Technol. 2014;51(10):2289-2303. doi:10.1007/s13197-012-0677-0
CrossRef - Shahidi F, Ambigaipalan P. Omega-3 Polyunsaturated Fatty Acids and Their Health Benefits. Annu Rev Food Sci Technol. 2018;9(1):345-381. doi:10.1146/annurev-food-111317-095850
CrossRef - Behl T, Kotwani A. Omega-3 fatty acids in prevention of diabetic retinopathy. Journal of Pharmacy and Pharmacology. 2017;69(8):946-954. doi:10.1111/jphp.12744
CrossRef - Zhang AC, Singh S, Craig JP, Downie LE. Omega-3 Fatty Acids and Eye Health: Opinions and Self-Reported Practice Behaviors of Optometrists in Australia and New Zealand. Nutrients. 2020;12(4):1179. doi:10.3390/nu12041179
CrossRef - Colussi G, Catena C, Novello M, Bertin N, Sechi LA. Impact of omega-3 polyunsaturated fatty acids on vascular function and blood pressure: Relevance for cardiovascular outcomes. Nutrition, Metabolism and Cardiovascular Diseases. 2017;27(3):191-200. doi:10.1016/j.numecd.2016.07.011
CrossRef - Gutiérrez S, Svahn SL, Johansson ME. Effects of Omega-3 Fatty Acids on Immune Cells. Int J Mol Sci. 2019;20(20):5028. doi:10.3390/ijms20205028
CrossRef - Omega 3 Supplements Market Size, Share & Trends Analysis Report By Source (Fish Oil, Krill Oil), By Form (Soft Gels, Capsules), By Functionality, By End-User, By Distribution Channel, By Region, And Segment Forecasts, 2020–2027. [(accessed on 10 June 2021)]; Available online: https://www.grandviewresearch.com/industry-analysis/omega-3-supplement-market
- Nevigato T, Masci M, Caproni R. Quality of Fish-Oil-Based Dietary Supplements Available on the Italian Market: A Preliminary Study. Molecules. 2021 19;26(16):5015. doi: 10.3390/molecules26165015.
CrossRef - Kim EJ, Kim JH, Kim MS, Jeong SH, Choi DH. Process Analytical Technology Tools for Monitoring Pharmaceutical Unit Operations: A Control Strategy for Continuous Process Verification. Pharmaceutics. 2021 Jun 21;13(6):919. doi: 10.3390/pharmaceutics13060919.
CrossRef - Rifat MA, Wahab AM, Rahman MA, Nahiduzzaman M, Mamun AA, Nutritional value of the marine fish in Bangladesh and their potential to address malnutrition: A review, Heliyon, 9, 2,2023,e13385 https://doi.org/10.1016/j.heliyon.2023.e13385.
CrossRef - Lurie P. Ensuring the Safety and Value of Supplements. JAMA Netw Open. 2023;6(7):e2323832. doi:10.1001/jamanetworkopen.2023.23832
CrossRef - Zhou X-R, Yao ZJ, Benedicto K, Nichols PD, Green A, Singh S. New Sustainable Oil Seed Sources of Omega-3 Long-Chain Polyunsaturated Fatty Acids: A Journey from the Ocean to the Field. Sustainability. 2023; 15(14):11327. https://doi.org/10.3390/su151411327
CrossRef - Lee J, Lee H, Kang S, Park W. Fatty Acid Desaturases, Polyunsaturated Fatty Acid Regulation, and Biotechnological Advances. Nutrients. 2016;8(1):23. doi:10.3390/nu8010023
CrossRef - Jenkins DJA, Josse AR. Fish oil and omega-3 fatty acids. Can Med Assoc J. 2008;178(2):150-150. doi:10.1503/cmaj.071754
CrossRef - Al-Madhagy S, Ashmawy NS, Mamdouh A, Eldahshan OA, Farag MA. A comprehensive review of the health benefits of flaxseed oil in relation to its chemical composition and comparison with other omega-3-rich oils. Eur J Med Res. 2023;28(1):240.
CrossRef - Deol P, Fahrmann J, Yang J, Evans JR, Rizo A, Grapov D, Salemi M, Wanichthanarak K, Fiehn O, Phinney B, Hammock BD, Sladek FM. Omega-6 and omega-3 oxylipins are implicated in soybean oil-induced obesity in mice. Sci Rep. 2017 Oct 2;7(1):12488. doi: 10.1038/s41598-017-12624-9.
CrossRef - Lin XL, Baisley J, Bier A, Vora D, Holub B. Transgenic Canola Oil Improved Blood Omega-3 Profiles: A Randomized, Placebo-Controlled Trial in Healthy Adults. Front Nutr. 2022;9:847114 doi:10.3389/fnut.2022.847114
CrossRef - Alcorta A, Porta A, Tárrega A, Alvarez MD, Vaquero MP. Foods for Plant-Based Diets: Challenges and Innovations. Foods. 2021;10(2):293. doi: 10.3390/foods10020293.
CrossRef - Shahidi F. Omega-3 fatty acids and marine oils in cardiovascular and general health: A critical overview of controversies and realities. J Funct Foods. 2015;19:797-800. doi:10.1016/j.jff.2015.09.038
CrossRef - Shahidi F. Marine oils: compositional characteristics and health effects. In: Shahidi F, ed. In Nutraceutical and Specialty Lipids and Their Co-Products, Ed. CRC Press; 2006:227-250. doi:10.1201/9781420015911
CrossRef - Orozco Colonia BS, Vinícius de Melo Pereira G, Soccol CR. Omega-3 microbial oils from marine thraustochytrids as a sustainable and technological solution: A review and patent landscape. Trends Food Sci Technol. 2020;99:244-256. doi:10.1016/j.tifs.2020.03.007
CrossRef - Agostoni C, Berni Canani oberto, Fairweather-Tait S, et al. Scientific Opinion on health benefits of seafood (fish and shellfish) consumption in relation to health risks associated with exposure to methylmercury. EFSA Journal. 2014;12(7):3761. doi:https://doi.org/10.2903/j.efsa.2014.3761
CrossRef - Santos HO, Price JC, Bueno AA. Beyond Fish Oil Supplementation: The Effects of Alternative Plant Sources of Omega-3 Polyunsaturated Fatty Acids upon Lipid Indexes and Cardiometabolic Biomarkers—An Overview. Nutrients. 2020;12(10):3159. doi:10.3390/nu12103159
CrossRef - Shahidi F, Miraliakbari H. Omega-3 ( n -3) Fatty Acids in Health and Disease: Part 1—Cardiovascular Disease and Cancer. J Med Food. 2004;7(4):387-401. doi:10.1089/jmf.2004.7.387
CrossRef - Khan MI, Shin JH, Kim JD. The promising future of microalgae: current status, challenges, and optimization of a sustainable and renewable industry for biofuels, feed, and other products. Microb Cell Fact. 2018;17(1):36. doi:10.1186/s12934-018-0879-x
CrossRef - Panahi Y, Darvishi B, Jowzi N, Beiraghdar F, Sahebkar A. Chlorella vulgaris: A Multifunctional Dietary Supplement with Diverse Medicinal Properties. Curr Pharm Des.2015;22(2):164-173. doi:10.2174/1381612822666151112145226
CrossRef - Andrade LM. Chlorella and Spirulina Microalgae as Sources of Functional Foods, Nutraceuticals, and Food Supplements; an Overview. MOJ Food Processing & Technology. 2018;6(1). doi:10.15406/mojfpt.2018.06.00144
CrossRef - Senanayake.P.J.N. S, Fichtali J. Marine oils: single cell oil as a source of nutraceuticals and specialty lipids: processing technologies and application. In: In Nutraceutical and Speciality Lipids and Their Co-Products,Ed. ; 2006:251-280.
CrossRef - Alasalvar C, Shahidi F, Quantick P. Food and Health Applications of Marine Nutraceuticals: a Review. In: Seafoods — Quality, Technology and Nutraceutical Applications. Springer Berlin Heidelberg; 2002:175-204. doi:10.1007/978-3-662-09836-3_15
CrossRef - Ciriminna R, Meneguzzo F, Delisi R, Pagliaro M. Enhancing and improving the extraction of omega-3 from fish oil. Sustain Chem Pharm. 2017;5:54-59. doi:10.1016/j.scp.2017.03.001
CrossRef - Gulzar S, Raju N, Chandragiri Nagarajarao R, Benjakul S. Oil and pigments from shrimp processing by-products: Extraction, composition, bioactivities and its application- A review. Trends Food Sci Technol. 2020;100:307-319. doi:10.1016/j.tifs.2020.04.005
CrossRef - Venugopalan VK, Gopakumar LR, Kumaran AK, et al. Encapsulation and Protection of Omega-3-Rich Fish Oils Using Food-Grade Delivery Systems. Foods. 2021;10(7):1566. doi:10.3390/foods10071566
CrossRef - Basuru GMVT, Kariyawasam MGTR, Alakolanga AGAW, Abeyrathne EDNS. Development of a Simple Nontoxic Method to Extract Crude Fish Oil from Yellowfin Tuna (Thunnus albacares) Offal. In: International Research Conference of Uva Wellassa University. ; 2019.
- Šimat V, Vlahović J, Soldo B, Skroza D, Ljubenkov I, Generalić Mekinić I. Production and Refinement of Omega-3 Rich Oils from Processing By-Products of Farmed Fish Species. Foods. 2019;8(4):125. doi:10.3390/foods8040125
CrossRef - Adeoti IA, Hawboldt K. A review of lipid extraction from fish processing by-product for use as a biofuel. Biomass Bioenergy. 2014;63:330-340. doi:10.1016/j.biombioe.2014.02.011
CrossRef - Rubio-Rodríguez N, Beltrán S, Jaime I, de Diego SM, Sanz MT, Carballido JR. Production of omega-3 polyunsaturated fatty acid concentrates: A review. Innovative Food Science & Emerging Technologies. 2010;11(1):1-12. doi:10.1016/j.ifset.2009.10.006
CrossRef - Al Khawli F, Pateiro M, Domínguez R, et al. Innovative Green Technologies of Intensification for Valorization of Seafood and Their By-Products. Mar Drugs. 2019;17(12):689. doi:10.3390/md17120689
CrossRef - Chemat F, Abert Vian M, Fabiano-Tixier AS, et al. A review of sustainable and intensified techniques for extraction of food and natural products. Green Chemistry. 2020;22(8):2325-2353. doi:10.1039/C9GC03878G
CrossRef - Fiori L, Manfrini M, Castello D. Supercritical CO2 fractionation of omega-3 lipids from fish by-products: Plant and process design, modeling, economic feasibility. Food and Bioproducts Processing. 2014;92(2):120-132. doi:10.1016/j.fbp.2014.01.001
CrossRef - Rahimi MA, Omar R, Ethaib S, Siti Mazlina MK, Awang Biak DR, Nor Aisyah R. Microwave-assisted extraction of lipid from fish waste. IOP Conf Ser Mater Sci Eng. 2017;206:012096. doi:10.1088/1757-899X/206/1/012096
CrossRef - Pateiro M, Domínguez R, Varzakas T, Munekata PES, Movilla Fierro E, Lorenzo JM. Omega-3-Rich Oils from Marine Side Streams and Their Potential Application in Food. Mar Drugs. 2021;19(5):233. doi:10.3390/md19050233
CrossRef - Qin J, Kurt E, LBassi T, Sa L, Xie D. Biotechnological production of omega-3 fatty acids: current status and future perspectives. Front Microbiol. 2023 Nov 7;14:1280296. doi: 10.3389/fmicb.2023.1280296
CrossRef - Cai Y, Yu X-H and Shanklin J .A toolkit for plant lipid engineering: Surveying the efficacies of lipogenic factors for accumulating specialty lipids. Front. Plant Sci.2022 13:1064176. doi: 10.3389/fpls.2022.1064176
CrossRef - Khan, W.A., Chun-Mei, H.U., Khan, N., Iqbal, A., Lyu, S.-W. and Shah, F. Bioengineered plants can be a useful source of omega-3 fatty acids. Biomed Res Int 2017 1-9 .https://doi.org/10.1155/2017/7348919
CrossRef - Patel A, Karageorgou D, Rova E, Katapodis P, Rova U, Christakopoulos P, Matsakas L. An Overview of Potential Oleaginous Microorganisms and Their Role in Biodiesel and Omega-3 Fatty Acid-Based Industries. Microorganisms. 2020 Mar 19;8(3):434. doi: 10.3390/microorganisms8030434
CrossRef - Zhang J, Freund MA, Culler MD, et al. How To Stabilize n-3 Polyunsaturated Fatty Acids (PUFAs) in an Animal Feeding Study?—Effects of the Temperature, Oxygen Level, and Antioxidant on Oxidative Stability of n-3 PUFAs in a Mouse Diet. J Agric Food Chem. 2020;68(46):13146-13153. doi:10.1021/acs.jafc.9b08298
CrossRef - Wang W, Yang H, Johnson D, Gensler C, Decker E, Zhang G. Chemistry and biology of n-3 PUFA peroxidation-derived compounds. Prostaglandins Other Lipid Mediat. 2017;132:84-91. doi:10.1016/j.prostaglandins.2016.12.004
CrossRef - Sottero B, Leonarduzzi G, Testa G, Gargiulo S, Poli G, Biasi F. Lipid Oxidation Derived Aldehydes and Oxysterols Between Health and Disease. European Journal of Lipid Science and Technology. 2019;121(1):1700047. doi:10.1002/ejlt.201700047
CrossRef - Yakubenko VP, Byzova T V. Biological and pathophysiological roles of end-products of DHA oxidation. Biochimica et Biophysica Acta (BBA) – Molecular and Cell Biology of Lipids. 2017;1862(4):407-415. doi:10.1016/j.bbalip.2016.09.022
CrossRef - Xu M, Lian Z, Chen X, et al. Effects of resveratrol on lipid and protein co-oxidation in fish oil-enriched whey protein isolate emulsions. Food Chem. 2021;365:130525. doi:10.1016/j.foodchem.2021.130525
CrossRef - Sedaghat Doost A, Nikbakht Nasrabadi M, Kassozi V, Nakisozi H, Van der Meeren P. Recent advances in food colloidal delivery systems for essential oils and their main components. Trends Food Sci Technol. 2020;99:474-486. doi:10.1016/j.tifs.2020.03.037
CrossRef - Amoabediny G, Haghiralsadat F, Naderinezhad S, et al. Overview of preparation methods of polymeric and lipid-based (niosome, solid lipid, liposome) nanoparticles: A comprehensive review. International Journal of Polymeric Materials and Polymeric Biomaterials. 2018;67(6):383-400. doi:10.1080/00914037.2017.1332623
CrossRef - Gulzar S, Benjakul S. Characteristics and storage stability of nanoliposomes loaded with shrimp oil as affected by ultrasonication and microfluidization. Food Chem. 2020;310:125916. doi:10.1016/j.foodchem.2019.125916
CrossRef - Aman Mohammadi M, Farshi P, Ahmadi P, Ahmadi A, Yousefi M, Ghorbani M, Hosseini SM. Encapsulation of Vitamins Using Nanoliposome: Recent Advances and Perspectives. Adv Pharm Bull. 2023 13(1):48-68. doi: 10.34172/apb.2023.005
CrossRef - Aryasree Sukumar, P. Gurumoorthi, K. A. Athmaselvi, Effect of ultrasonication on emulsion formulation, encapsulation efficiency, and oxidative stability of spray dried chia seed oil, Journal of Food Science and Technology,2023 10.1007/s13197-023-05716-0, 60, 6, (1761-1771
CrossRef - C. Anandharamakrishnan, Sayantani Dutta, J.A. Moses, Introductory overview on liposomes, Liposomal Encapsulation in Food Science and Technology, 2023 10.1016/B978-0-12-823935-3.00012-6, (1-14),
CrossRef - Liu W, Hou Y, Jin Y, Wang Y, Xu X, Han J. Research progress on liposomes: Application in food, digestion behavior and absorption mechanism. Trends Food Sci Technol. 2020;104:177-189. doi:10.1016/j.tifs.2020.08.012
CrossRef - Han C, Yang C, Li X, Liu E, Meng X, Liu B. DHA loaded nanoliposomes stabilized by β-sitosterol: Preparation, characterization and release in vitro and vivo. Food Chem. 2022;368:130859. doi:10.1016/j.foodchem.2021.130859
CrossRef - Liu E, Zhao S, Li X, Meng X, Liu B. Preparation, characterization of PLGA/chitosan nanoparticles as a delivery system for controlled release of DHA. Int J Biol Macromol. 2021;185:782-791. doi:10.1016/j.ijbiomac.2021.06.190
CrossRef - Hosseini SF, Soofi M, Rezaei M. Enhanced physicochemical stability of n-3 PUFAs concentrates-loaded nanoliposomes decorated by chitosan/gelatin blend coatings. Food Chem. 2021;345:128865. doi:10.1016/j.foodchem.2020.128865
CrossRef - Choudhary P, Dutta S, Moses JA, Anandharamakrishnan C (2021) Nanoliposomal encapsulation of chia oil for sustained delivery of α-linolenic acid. Int J Food Sci Technol 56(9):4206–4214
CrossRef - Du Q, Zhou L, Lyu F, Liu J, Ding Y. The complex of whey protein and pectin: Interactions, functional properties and applications in food colloidal systems – A review. Colloids Surf B Biointerfaces. 2022;210:112253. doi:10.1016/j.colsurfb.2021.112253
CrossRef - Walker R, Decker EA, McClements DJ. Development of food-grade nanoemulsions and emulsions for delivery of omega-3 fatty acids: opportunities and obstacles in the food industry. Food Funct. 2015;6(1):41-54. doi:10.1039/C4FO00723A
CrossRef - Jiang H, Sheng Y, Ngai T. Pickering emulsions: Versatility of colloidal particles and recent applications. Curr Opin Colloid Interface Sci. 2020;49:1-15. doi:10.1016/j.cocis.2020.04.010
CrossRef - Walker RM, Gumus CE, Decker EA, McClements DJ. Improvements in the formation and stability of fish oil-in-water nanoemulsions using carrier oils: MCT, thyme oil, & lemon oil. J Food Eng. 2017;211:60-68. doi:10.1016/j.jfoodeng.2017.05.004
CrossRef - Cofrades S, Garcimartín A, Gómez-Estaca J, et al. The Effect of Emulsifying Protein and Addition of Condensed Tannins on n-3 PUFA Enriched Emulsions for Functional Foods. Foods. 2020;9(11):1589. doi:10.3390/foods9111589
CrossRef - Padial-Domínguez M, Espejo-Carpio FJ, García-Moreno PJ, Jacobsen C, Guadix EM. Protein derived emulsifiers with antioxidant activity for stabilization of omega-3 emulsions. Food Chem. 2020;329:127148. doi:10.1016/j.foodchem.2020.127148
CrossRef - Jagtap AA, Badhe YS, Hegde MV, Zanwar AA. Development and characterization of stabilized omega-3 fatty acid and micronutrient emulsion formulation for food fortification. J Food Sci Technol. 2021 Mar;58(3):996-1004. doi: 10.1007/s13197-020-04614-z. Epub 2020 Jul 3.
CrossRef - Ued FV, Mathias MG, Toffano RBD, Barros TT, Almada MORV, Salomão RG, Coelho-Landell CA, Hillesheim E, Camarneiro JM, Camelo-Junior JS, Aragon DC, Moco S, Kussmann M, Kaput J, Monteiro JP. Vitamin B2 and folate concentrations are associated with ARA, EPA and DHA fatty acids in red blood cells of Brazilian children and adolescents. Nutrients. 2019;11(12):2918. doi: 10.3390/nu11122918.
CrossRef - Yary T, Voutilainen S, Tuomainen TP, Ruusunen A, Nurmi T, Virtanen JK. Omega-6 polyunsaturated fatty acids, serum zinc, delta-5- and delta-6-desaturase activities and incident metabolic syndrome. J Hum Nutr Diet. 2017;30(4):506–514. doi: 10.1111/jhn.12437
CrossRef - Saunders AV, Davis BC. Garg M L (2012) Omega-3 polyunsaturated fatty acids and vegetarian diets. MJA Open. 2012;1(2):22–26. doi: 10.5694/mjao11.11507
CrossRef - Zhu Q, Pan Y, Jia X, Li J, Zhang M, Yin L. Review on the stability mechanism and application of water-in-oil emulsions encapsulating various additives. Compr Rev Food Sci F. 2019;18(6):1660–1675. doi: 10.1111/1541-4337.12482.
CrossRef - Ozkan G, Franco P, De Marco I, Xiao J, Capanoglu E. A review of microencapsulation methods for food antioxidants: Principles, advantages, drawbacks and applications. Food Chem. 2019;272:494-506. doi:10.1016/j.foodchem.2018.07.205
CrossRef - Charles AL, Abdillah AA, Saraswati YR, et al. Characterization of freeze-dried microencapsulation tuna fish oil with arrowroot starch and maltodextrin. Food Hydrocoll. 2021;112:106281. doi:10.1016/j.foodhyd.2020.106281
CrossRef - Strobel SA, Hudnall K, Arbaugh B, Cunniffe JC, Scher HB, Jeoh T. Stability of Fish Oil in Calcium Alginate Microcapsules Cross-Linked by In Situ Internal Gelation During Spray Drying. Food Bioproc Tech. 2020;13(2):275-287. doi:10.1007/s11947-019-02391-y
CrossRef - Solomando JC,Vázquez F, Antequera T, Folgado C, Perez-Palacios T ,Addition of fish oil microcapsules to meat products – Implications for omega-3 enrichment and salt reduction,Journal of Functional Foods, 105,2023,105575, https://doi.org/10.1016/j.jff.2023.105575.
CrossRef - Geranpour M, Assadpour E, Jafari SM. Recent advances in the spray drying encapsulation of essential fatty acids and functional oils. Trends Food Sci Technol. 2020;102:71-90. doi:10.1016/j.tifs.2020.05.028
CrossRef - Boerekamp DMW, Andersen ML, Jacobsen C, Chronakis IS, García-Moreno PJ. Oxygen permeability and oxidative stability of fish oil-loaded electrosprayed capsules measured by Electron Spin Resonance: Effect of dextran and glucose syrup as main encapsulating materials. Food Chem. 2019;287:287-294. doi:10.1016/j.foodchem.2019.02.096
CrossRef - García-Moreno PJ, Pelayo A, Yu S, et al. Physicochemical characterization and oxidative stability of fish oil-loaded electrosprayed capsules: Combined use of whey protein and carbohydrates as wall materials. J Food Eng. 2018;231:42-53. doi:10.1016/j.jfoodeng.2018.03.005
CrossRef - Sultana A, Adachib S, Yoshii H. Encapsulation of fish oil and essential fatty acids by spray drying Sustainable Food Technol., 2023,1, 827-836
CrossRef - Wang Y, Liu W, Chen XD, Selomulya C. Micro-encapsulation and stabilization of DHA containing fish oil in protein-based emulsion through mono-disperse droplet spray dryer. J Food Eng. 2016;175:74-84. doi:10.1016/j.jfoodeng.2015.12.007
CrossRef - García-Moreno, P. J., Mendes, A. C., Jacobsen, C., & Chronakis, I. S. (2018). Biopolymers for the nano-microencapsulation of bioactive ingredients by electrohydrodynamic processing. In T. J. Gutiérrez (Ed.), Polymers for food applications (pp. 447-479). Springer International Publishing.
CrossRef - García-Moreno PJ, Mendes AC, Jacobsen C, Chronakis IS. Biopolymers for the Nano-microencapsulation of Bioactive Ingredients by Electrohydrodynamic Processing. In: Polymers for Food Applications. Springer International Publishing; 2018:447-479. doi:10.1007/978-3-319-94625-2_17
CrossRef - Saifullah Md, Shishir MRI, Ferdowsi R, Tanver Rahman MR, Van Vuong Q. Micro and nano encapsulation, retention and controlled release of flavor and aroma compounds: A critical review. Trends Food Sci Technol. 2019;86:230-251. doi:10.1016/j.tifs.2019.02.030
CrossRef - Shishir MRI, Xie L, Sun C, Zheng X, Chen W. Advances in micro and nano-encapsulation of bioactive compounds using biopolymer and lipid-based transporters. Trends Food Sci Technol. 2018;78:34-60. doi:10.1016/j.tifs.2018.05.018
CrossRef - Rahmani-Manglano, N.E.; Guadix, E.M.; Jacobsen, C.; García-Moreno, P.J. Comparative Study on the Oxidative Stability of Encapsulated Fish Oil by Monoaxial or Coaxial Electrospraying and Spray-Drying. Antioxidants 2023, 12, 266. https://doi.org/10.3390/antiox12020266
CrossRef - Fan K, Zhang M, Mujumdar AS. Recent developments in high efficient freeze-drying of fruits and vegetables assisted by microwave: A review. Crit Rev Food Sci Nutr. 2019;59(8):1357-1366. doi:10.1080/10408398.2017.1420624
CrossRef - Chen Q, Zhong F, Wen J, McGillivray D, Quek SY. Properties and Stability of Spray-Dried and Freeze-Dried Microcapsules Co-Encapsulated with Fish Oil, Phytosterol Esters, and Limonene. Drying Technology. 2013;31(6):707-716. doi:10.1080/07373937.2012.755541
CrossRef - Lucía C, Marcela F, Ainhoa L. Encapsulation of Almond Essential Oil by Co-Extrusion/Gelling Using Chitosan as Wall Material. Journal of Encapsulation and Adsorption Sciences. 2017;07(01):67-74. doi:10.4236/jeas.2017.71004
CrossRef - Bamidele OP, Emmambux MN. Encapsulation of bioactive compounds by “extrusion” technologies: a review. Crit Rev Food Sci Nutr. 2021;61(18):3100-3118. doi:10.1080/10408398.2020.1793724
CrossRef - Phung AS, Bannenberg G, Vigor C, et al. Chemical Compositional Changes in Over-Oxidized Fish Oils. Foods. 2020;9(10):1501. doi:10.3390/foods9101501
CrossRef - Hilleman DE, Wiggins BS, Bottorff MB. Critical Differences Between Dietary Supplement and Prescription Omega-3 Fatty Acids: A Narrative Review. Adv Ther. 2020;37(2):656-670. doi:10.1007/s12325-019-01211-1
CrossRef - Collins N, Tighe AP, Brunton SA, Kris-Etherton PM. Differences between Dietary Supplement and Prescription Drug Omega-3 Fatty Acid Formulations: A Legislative and Regulatory Perspective. J Am Coll Nutr. 2008;27(6):659-666. doi:10.1080/07315724.2008.10719743
CrossRef - Wallace TC. Twenty Years of the Dietary Supplement Health and Education Act—How Should Dietary Supplements Be Regulated? ,. J Nutr. 2015;145(8):1683-1686. doi:10.3945/jn.115.211102
CrossRef - Suzan AJ, Garcia PHD, Furlan CPB, et al. Oxidative stability of fish oil dietary supplements and their cytotoxic effect on cultured human keratinocytes. NFS Journal. 2022;29:1-7. doi:10.1016/j.nfs.2022.09.002
CrossRef - Othón-Díaz ED, Fimbres-García JO, Flores-Sauceda M, et al. Antioxidants in Oak (Quercus sp.): Potential Application to Reduce Oxidative Rancidity in Foods. Antioxidants. 2023;12(4):861. doi:10.3390/antiox12040861
CrossRef - Albuquerque TG, Costa HS, Oliveira MBPP. 4-hydroxy-2-alkenals in foods: a review on risk assessment, analytical methods, formation, occurrence, mitigation and future challenges. Crit Rev Food Sci Nutr. 2022;62(13):3569-3597. doi:10.1080/10408398.2020.1867499
CrossRef - Reis DR, Ambrosi A, Luccio M Di. Encapsulated essential oils: A perspective in food preservation. Future Foods. 2022;5:100126. doi:10.1016/j.fufo.2022.100126
CrossRef - Mason RP, Sherratt SCR. Omega-3 fatty acid fish oil dietary supplements contain saturated fats and oxidized lipids that may interfere with their intended biological benefits. Biochem Biophys Res Commun. 2017;483(1):425-429. doi:10.1016/j.bbrc.2016.12.127
CrossRef - Ayala A, Muñoz MF, Argüelles S. Lipid Peroxidation: Production, Metabolism, and Signaling Mechanisms of Malondialdehyde and 4-Hydroxy-2-Nonenal. Oxid Med Cell Longev. 2014;2014:1-31. doi:10.1155/2014/360438
CrossRef - Bannenberg G, Rice HB, Bernasconi A, et al. Ingredient label claim compliance and oxidative quality of EPA/DHA omega-3 retail products in the U.S. Journal of Food Composition and Analysis. 2020;88:103435. doi:10.1016/j.jfca.2020.103435
CrossRef - Yenipazar H, Şahin‐Yeşilçubuk N. Effect of packaging and encapsulation on the oxidative and sensory stability of omega‐3 supplements. Food Sci Nutr. 2023;11(3):1426-1440. doi:10.1002/fsn3.3182
CrossRef - Bhargava R, Pandey K, Ranjan S, Mehta B, Malik A. Omega-3 fatty acids supplements for dry eye – Are they effective or ineffective? Indian J Ophthalmol. 2023;71(4):1619. doi:10.4103/IJO.IJO_2789_22
CrossRef - Galuch M, Carbonera F, Magon T, et al. Quality Assessment of Omega-3 Supplements Available in the Brazilian Market. J Braz Chem Soc. Published online 2017. doi:10.21577/0103-5053.20170177
CrossRef - Damerau A, Ahonen E, Kortesniemi M, Puganen A, Tarvainen M, Linderborg KM. Evaluation of the composition and oxidative status of omega-3 fatty acid supplements on the Finnish market using NMR and SPME-GC–MS in comparison with conventional methods. Food Chem. 2020;330:127194. doi:10.1016/j.foodchem.2020.127194
CrossRef - Bannenberg G, Mallon C, Edwards H, et al. Omega-3 Long-Chain Polyunsaturated Fatty Acid Content and Oxidation State of Fish Oil Supplements in New Zealand. Sci Rep. 2017;7(1):1488. doi:10.1038/s41598-017-01470-4
CrossRef - Kolanowski W. Omega-3 LC PUFA Contents and Oxidative Stability of Encapsulated Fish Oil Dietary Supplements. Int J Food Prop. 2010;13(3):498-511. doi:10.1080/10942910802652222
CrossRef - Kowalski R, Kałwa K, Wilczyński K, Kobus Z. The fatty acids composition of selected fish oils used as dietary supplements. Polish Journal of Natural Sciences. 2019;34:115-126.
- Kleiner AC, Cladis DP, Santerre CR. A comparison of actual versus stated label amounts of EPA and DHA in commercial omega-3 dietary supplements in the United States. J Sci Food Agric. 2015;95(6):1260-1267. doi:10.1002/jsfa.6816
CrossRef - Ritter JCS, Budge SM, Jovica F. Quality analysis of commercial fish oil preparations. J Sci Food Agric. 2013;93(8):1935-1939. doi:10.1002/jsfa.5994
CrossRef - Albert BB, Derraik JGB, Cameron-Smith D, et al. Fish oil supplements in New Zealand are highly oxidised and do not meet label content of n-3 PUFA. Sci Rep. 2015;5(1):7928. doi:10.1038/srep07928
CrossRef - Alghamdi F, Embaby MG, G�nal M, AbuGhazaleh AA. Analysis of the Omega-3 Fatty Acids Content in Commercial Omega-3 Supplements in Arab Gulf Countries. Am J Anim Vet Sci. 2020;15(4):315-322. doi:10.3844/ajavsp.2020.315.322
CrossRef - Burca N, Watson RR. Fish Oil Supplements, Contaminants, and Excessive Doses. In: Omega-3 Fatty Acids in Brain and Neurological Health. Elsevier; 2014:447-454. doi:10.1016/B978-0-12-410527-0.00036-3
CrossRef - Jansson P, Kay B. Aldehydes identified in commercially available n-3 supplements via 1 H NMR spectroscopy. Nutrition. 2019;60:74-79. doi:10.1016/j.nut.2018.10.004
CrossRef - Fialkow J. Omega-3 Fatty Acid Formulations in Cardiovascular Disease: Dietary Supplements are Not Substitutes for Prescription Products. American Journal of Cardiovascular Drugs. 2016;16(4):229-239. doi:10.1007/s40256-016-0170-7
CrossRef - Yang Y, Deng W, Wang Y, et al. The effect of omega-3 fatty acids and its combination with statins on lipid profile in patients with hypertriglyceridemia: A systematic review and meta-analysis of randomized controlled trials. Front Nutr. 2022;9. doi:10.3389/fnut.2022.1039056
CrossRef - Li JJ, Dou KF, Zhou ZG, et al. Role of omega-3 fatty acids in the prevention and treatment of cardiovascular Diseases: A consensus statement from the Experts’ Committee Of National Society Of Cardiometabolic Medicine. Front Pharmacol. 2022;13. doi:10.3389/fphar.2022.1069992
CrossRef - Calder PC, Deckelbaum RJ. Editorial: Omega-3 fatty acids and cardiovascular outcomes: an update. Curr Opin Clin Nutr Metab Care. 2019;22(2):97-102. doi:10.1097/MCO.0000000000000543
CrossRef - Brigham EP, Woo H, McCormack M, et al. Omega-3 and Omega-6 Intake Modifies Asthma Severity and Response to Indoor Air Pollution in Children. Am J Respir Crit Care Med. 2019;199(12):1478-1486. doi:10.1164/rccm.201808-1474OC
CrossRef - Rosenberg K. Omega-3 Fatty Acid Intake Lowers Risk of Diabetic Retinopathy. AJN, American Journal of Nursing. 2017;117(1):60-61. doi:10.1097/01.NAJ.0000511570.42940.9e
CrossRef - Brinton EA, Mason RP. Prescription omega-3 fatty acid products containing highly purified eicosapentaenoic acid (EPA). Lipids Health Dis. 2017;16(1):23. doi:10.1186/s12944-017-0415-8
CrossRef - Fabian CJ, Kimler BF, Hursting SD. Omega-3 fatty acids for breast cancer prevention and survivorship. Breast Cancer Research. 2015;17(1):62. doi:10.1186/s13058-015-0571-6
CrossRef - Krupa K, Fritz K, Parmar M. Omega-3 Fatty Acids.; 2023.
- Musazadeh V, Kavyani Z, Naghshbandi B, Dehghan P, Vajdi M. The beneficial effects of omega-3 polyunsaturated fatty acids on controlling blood pressure: An umbrella meta-analysis. Front Nutr. 2022;9. doi:10.3389/fnut.2022.985451
CrossRef - Arca M, Borghi C, Pontremoli R, et al. Hypertriglyceridemia and omega-3 fatty acids: Their often overlooked role in cardiovascular disease prevention. Nutrition, Metabolism and Cardiovascular Diseases. 2018;28(3):197-205. doi:10.1016/j.numecd.2017.11.001
CrossRef - Liu QK. Triglyceride-lowering and anti-inflammatory mechanisms of omega-3 polyunsaturated fatty acids for atherosclerotic cardiovascular risk reduction. J Clin Lipidol. 2021;15(4):556-568. doi:10.1016/j.jacl.2021.05.007
CrossRef - Dighriri IM, Alsubaie AM, Hakami FM, et al. Effects of Omega-3 Polyunsaturated Fatty Acids on Brain Functions: A Systematic Review. Cureus. Published online October 9, 2022. doi:10.7759/cureus.30091
CrossRef - Li P, Song C. Potential treatment of Parkinson’s disease with omega-3 polyunsaturated fatty acids. Nutr Neurosci. 2022;25(1):180-191. doi:10.1080/1028415X.2020.1735143
CrossRef - Ferreira I, Rauter AP, Bandarra NM. Marine Sources of DHA-Rich Phospholipids with Anti-Alzheimer Effect. Mar Drugs. 2022;20(11):662. doi:10.3390/md20110662
CrossRef - Alfawaz H, Al-Onazi M, Bukhari SI, et al. The Independent and Combined Effects of Omega-3 and Vitamin B12 in Ameliorating Propionic Acid Induced Biochemical Features in Juvenile Rats as Rodent Model of Autism. Journal of Molecular Neuroscience. 2018;66(3):403-413. doi:10.1007/s12031-018-1186-z
CrossRef - Meital LT, Windsor MT, Perissiou M, et al. Omega-3 fatty acids decrease oxidative stress and inflammation in macrophages from patients with small abdominal aortic aneurysm. Sci Rep. 2019;9(1):12978. doi:10.1038/s41598-019-49362-z
CrossRef - Oppedisano F, Macrì R, Gliozzi M, et al. The Anti-Inflammatory and Antioxidant Properties of n-3 PUFAs: Their Role in Cardiovascular Protection. Biomedicines. 2020;8(9):306. doi:10.3390/biomedicines8090306
CrossRef - Mickleborough TD. Omega-3 Polyunsaturated Fatty Acids in Physical Performance Optimization. Int J Sport Nutr Exerc Metab. 2013;23(1):83-96. doi:10.1123/ijsnem.23.1.83
CrossRef - Health Science Authority (HSA). Regulatory overview of health supplements. Published December 31, 2018. https://www.hsa.gov.sg/health-supplements/overview
- Health Canada. About Natural Health Product Regulation in Canada. Published July 6, 2022. https://www.canada.ca/en/health-canada/services/drugs-health-products/natural-non-prescription/regulation.html
- Therapeutic Goods Administration (TGA). Complementary medicines. https://www.tga.gov.au/topics/complementary-medicines
- Therapeutic Goods Administration (TGA). Overview of the regulation of listed medicines and registered complementary medicines. Published July 2021. https://www.tga.gov.au/sites/default/ files/overview -regulation-listed-medicines-and-registered-complementary-medicines.pdf
- Therapeutic Goods Administration (TGA). Registered complementary medicines. https://www.tga.gov.au/products/medicines/non-prescription-medicines/registered-complementary-medicines
- The Global Organization for EPA and DHA Omega-3s (GOED). The Global Organization for EPA and DHA Omega-3s (GOED) Guidance Document.; 2018. Accessed July 25, 2023. https://goedomega3 .com/storage/app/ media/uploaded-files/GOED%20TGD%20-%202018%2007%2019.pdf
- Bannenberg G. EPA/DHA Dietary Supplement Compliance: Content Claims and Quality. In: Omega-3 Summit . ; 2019.
- United States Pharmacopeia (USP). (401) FATS AND FIXED OILS. Published online 2022. Accessed July 25, 2023. https://doi.usp.org/USPNF/USPNF_M99160_04_01.html
- European Pharmacopoeia. Omega -3-Marine Triglycerides.
- European Pharmacopoeia. Appendix X P. Oils Rich in Omega -3-acids.
- Australian Government D of health, TGAT. Compositional Guideline for DHA/EPA rich Schizochytrium algal oil. Accessed July 30, 2023. https://www.tga.gov.au/sites/default/files/cm-cg-dha-epa-rich-schizochytrium-algal-oil.pdf
- Health Canada. Fish Oil . Published online March 15, 2023. Accessed July 30, 2023. https://webprod.hc-sc.gc.ca/nhpid-bdipsn/monoReq.do?id=88&lang=eng

This work is licensed under a Creative Commons Attribution 4.0 International License.






