Introduction
Cereal grains, including rice, wheat, and maize, constitute the main source of dietary carbohydrates and substantially contribute to human energy needs and health.1 Glucose, a breakdown product of carbohydrate metabolism, is central to energy intake. At the same time, the incidence of diabetes mellitus (higher blood glucose levels) has been on the rise in the worldwide2 with an expected prevalence of 7079 individuals per 100,000 by 2030.3 In addition, many regions of the world especially Asian and Indian diet is rich in carbohydrate 4. Therefore, the quantity and quality of carbohydrate intake can be crucial in mitigating public health challenges.
The scientific literature has extensively reviewed four components to assess carbohydrate quality: dietary fibre content/intake, whole grain content/intake, free sugar content/intake, and glycemic index/load.5 The concept of the glycemic index (GI) has been introduced to rank carbohydrates based on their effect on postprandial glycemia.6 A recent meta-analysis provided evidence that a higher dietary GI and glycemic load increases the risk of diabetes.7,8 At the same time, low-GI foods have also been reported to improve glycemic control in diabetes, increase insulin sensitivity9, and enhance pancreatic β-cell function8, highlighting the importance of considering GI in food choice.
The identification of food ingredients that can mitigate the high glycemic response induced by high-GI foods has garnered significant attention in recent years.9,10 In addition, convenience foods with low GI are required in the era of increasing prevalence of noncommunicable diseases, such as diabetes.
Several food ingredients11-13 and strategies14-16 have been used in recent years to reduce the high glycemic response induced by high-GI foods. Although grain-based foods are consumed for energy requirements, they often lack adequate protein and dietary fibre. In addition, the urgent need for novel and natural functional food ingredients to control postprandial blood glucose surge to manage chronic metabolic diseases has been expressed in the consensus statement of the International Carbohydrate Quality Consortium (ICQC).17
One such potential food ingredient is fenugreek (Trigonella foenum graecum L., Fabaceae family) seed, a spice commonly used in India, which has robust scientific evidence for its blood glucose-lowering effects in diabetic conditions18.
Several studies incorporating fenugreek seed powder into food preparations have demonstrated promising GI-lowering effects. A reduction in glycemic index (GI) values was observed when fenugreek seed powder was added to common Indian dishes, such as dhokla, upma, and laddu.19 Similarly, replacing 10% refined wheat flour with fenugreek seed powder in buns and flatbreads significantly reduced GI values.20 Moreover, adding fenugreek seed powder to fried rice and white bread with jam resulted in a substantial decrease in the incremental area under the curve (IAUC) compared with the consumption of these foods alone.21 The dietary fibres (mucilage and galactomannan) of fenugreek seeds are believed to cause GI lowering effects by reducing the rate of carbohydrate digestion and absorption, and postprandial blood glucose.22 However, the precise composition of fenugreek seed responsible for GI-lowering effects is unknown. In addition, global regulatory standards require standardized products with consistent quality and quantity of bioactive compounds.
In addition, the incorporation of fenugreek seeds into the daily diet is often hindered by their inherent bitterness. Recently, defatted fenugreek seed flakes (Fenuflakes™) were developed as palatable and standardized forms of fenugreek seeds that are safe for human consumption.23 Fenuflakes hold promise as a novel ingredient for lowering the GI of high-carbohydrate food reparations because the defatting process enriches seeds with fibres and proteins and removes fats.24 and further reduces the bitterness of fenugreek.25. Therefore, the present study evaluated the GI-lowering capacity of Fenuflakes to grain-based Indian food preparations in healthy human subjects using an open-label randomized control study.
Materials and Methods
Trial Design
The present study was conducted following a randomized, controlled, and open-label design to assess the effect of Fenuflakes on the glycemic response in healthy human participants with six food preparations, with and without fenugreek seed flakes. The research protocol adhered to the guidelines of the “Food and Agriculture Organization”(FAO)/ World Health Organization (WHO)”26-28 and “ISO:26642:2010” standard29 for recommendations of GI determination and food classification. The study was conducted following the ethical principles of the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of the “Madras Diabetes Research Foundation (MDRF) (No: ECR/194/Inst/TN/2013/RR-19). All participants provided written informed consent prior to participation and the study was prospectively registered in the Clinical Trial Registry of India (CTRI) (No: CTRI/2022/02/040940).
Participants
The study recruited 15 healthy participants, (7 females and 8 males) at the Glycemic Index Testing Centre. A sample size of 15 was determined based on the reported guidelines for determining the GI of food where ten individuals can provide a sufficient degree of power and precision.28
The inclusion criteria were as follows: healthy males and females, aged 18 to 45 years, body mass index (BMI) between 18.5 and 22.9 kg/m2, willingness to consume test foods, without known food allergies or intolerances, not on medications known to affect glucose tolerance, on a stable dose of oral contraceptives, acetylsalicylic acid, thyroxin, vitamins, and mineral supplements or drugs to treat hypertension or osteoporosis.
Individuals with diabetes mellitus or the use of anti-hyperglycaemic drugs or insulin to treat diabetes and related conditions, pregnant or lactating women, those who experienced a major medical or surgical event requiring hospitalization within the preceding 3 months, those with any disease or drug use that could impact digestion and absorption of nutrients, or those using steroids or antipsychotics, which can significantly impact glucose metabolism and body fat distribution, were excluded from the study. In addition, the participants were allocated to the sequence of food preparation in a randomized manner, with Fenuflakes (test preparation) or without Fenuflakes (control preparation).
The intervention – Fenuflakes
Two batches of samples of Fenuflakes (debittered fenugreek seed flakes) were provided by Indus Biotech, Ltd., Pune, India and used to prepare the food preparations. Both samples have same the specifications and contained more than 25% of each protein, soluble fibres, and insoluble fibres with negligible carbohydrate and fat content by AOAC official methods, as described earlier.23 Sample 1 (used for oats porridge, cooked raw rice, rice idly, semolina upma) contained 60.0 % total dietary fibres, 34.9 % soluble fibre, 25.1% insoluble fibre, 34.07 % protein content, 0.4 % net carbohydrate, and 1.72 % total fat content. Sample 2 (used for potato sandwich and potato paratha) contained 57.53 % total dietary fibres, 25.28 % soluble fibres, 32.25 % insoluble fibres, 40.07 % protein content, 0.21 % total carbohydrate and 0.52 % total fat content.
The intervention, Fenuflakes, was debittered and did not have a bitter taste. However, the open-label design was selected to mask the distinct flavor of fenugreek containing food.
Food Preparations
This study evaluated the glycemic index of six commonly consumed Indian food preparations: oats porridge, pressure-cooked plain raw rice, rice idly, semolina upma, potato sandwich, and potato paratha. Each food preparation was prepared in two versions: without fenugreek seed flakes (Control) and with the incorporation of 10 g of Fenuflakes per serving (Test), each providing 50 g (or 25 g for oat porridge) of available carbohydrate-containing portions.
The details of the food preparations are listed in Table 1.
Table 1: Food preparations with and without Fenuflakes
|
Preparations |
Method |
|
Oats Porridge
|
1. Rolled oats + salt (± Fenuflakes flakes) + potable water à mix. 2. Cook on medium flame (4 minutes) à stir à soft and glossy oats. 3. Cool (8 minutes) à RT à serve |
|
Cooked Raw Rice
|
1. Rice + water à rinse x 3 2. Strain rice à pressure cook rice (± Fenuflakes flakes) + water. 3. Cook on medium flame (10 minutes) à10 whistles. 4. Cool (10 minutes) à RT à serve |
|
Rice Idly
|
1. Idly rice + water, urad dal + water à rinse x 3 times à soak in water separately (5 hours). 2. Rice (± Fenuflakes powder) (soak for 10 minutes) + urad dal (soak for 5 minutes) à grind à make to batter à add salt and mix. 3. Batter (rest for 5 hours) à refrigerate (10 hours) à rest (1 hours) at RT 4. 35 g batter à idly mold à steam on medium flame (13 minutes) à cool à RT à serve |
|
Semolina Upma
|
1. Boiling water (± Fenuflakes powder) à Mix à + Semolina à mix à heat (5 minutes) à cool. 2. Groundnut oil + mustard seeds + whole black gram + split Bengal gram à roast (1 minutes). 3. Onion + ginger + salt + curry leaves à sauté à add water. 4. Add cooled semolina à stir à cover with lid (1 minutes) à cool (10 minutes) à serve |
|
Potato Sandwich
|
1. Potatoes à pressure cook à cool à mash. 2. Mint + coriander leaves + green chillies + water à blend. 3. Mashed Potatoes (± Fenuflakes flakes) à make paste + chopped onion + salt + spices à mix. 4. Dough into white bread à toast (golden-brown) à cut (4 squares) à serve |
|
Potato Paratha
|
1. Wheat flour + water (± Fenuflakes flakes) + salt à mix à knead à dough à grease with groundnut oil à rest. 2. Potatoes à pressure cook à cool à peel à mash (± Fenuflakes flakes) + chopped onion + coriander + green chilies + spices. 3. Wheat dough à roll into circle à 30 g potato stuffing à dumpling. 4. Dumpling à flatten à cook both sides on medium flame à cool à RT à serve |
± Fenuflakes – (+) Fenuflakes (Test) or (-) without Fenuflakes (Control), RT- Room Temperature
Nutritional analyses were conducted on three batches of each food preparation to determine the content of available carbohydrates, proteins, fats, fibres, moisture, and ash, following validated AAOC methods.30 The total available carbohydrate content was quantified by an enzymatic method using a Megazyme assay kit (K-ACHDF; Megazyme International, Ireland). The amount of available carbohydrates was standardized across all food preparations, ensuring that participants consumed servings containing either 50 or 25 g of available carbohydrates, depending on the specific preparation.
Study procedure
The study procedure has been briefly depicted in Figure 1. All participants visited the GI testing centre on each test day in the morning after an overnight fast of 10-12 h. Upon arrival, participants completed a brief questionnaire regarding their diet and physical activity habits over the previous 24 h to ensure a consistent diet and level of physical activity throughout the study period.
Participants’ anthropometric characteristics, such as height, weight and body fat (%) (using a digital body composition machine, HBF -224, Omron Health Care Co., Ltd. Kyoto, Japan), waist circumference (using a non-stretchable measuring tape) were recorded. Blood pressure [using digital apparatus, HEM 8712, Omron Healthcare Manufacturing Vietnam Co., Ltd, Vietnam)] and fasting blood glucose (using a glucose analyser, Hemocue 201+, HemoCue AB, Ängelholm, Sweden) were also recorded.
Baseline fasting blood glucose levels were measured on collected blood immediately before and 5 minutes after consumption of food preparations using an automatic lancet device (Hemocue 201+, HemoCue AB, Ängelholm, Sweden). Before the finger prick, the participants were instructed to gently massage and warm their hands to increase blood flow and reduce plasma dilution. The mean of the two readings was used for further analysis.
Food Consumption and Blood Glucose Monitoring
Participants consumed a reference food (55 g of glucose dissolved in 250 ml of water) on three separate occasions. For the preparation of oat porridge, a modified reference food of 27.5 g of glucose in 125 ml of water was used. The participants consumed the reference food, control food preparations, and test food preparations for breakfast at the same time window in a randomized crossover order. A two-day washout period was implemented between each measurement to minimize carry-over effects.
Participants consumed the assigned food preparation within 15 minutes, with the first bite-taking time marked as the baseline time (0). Blood samples were collected 5 minutes before, immediately after, and 15, 30, 45, 60, 90, and 120 minutes after food consumption. Participants were provided with 250 ml of water along with food preparation and an additional 250 ml of water during the subsequent two hours. Throughout the study period, the participants were instructed not to engage in any strenuous physical activity.
To determine the GI value of each food preparation, a plot of blood glucose levels versus time was plotted, and the incremental area under the curve (IAUC) of the food preparations (Reference, Test or Control) was calculated using the trapezoid rule, ignoring the area below the baseline. The IAUC of the reference food was assigned a GI value of 100 and the GI values of the Test or Control food preparations were expressed as a percentage of the reference food as recommended by the FAO/WHO method26 using the formula: GI value (%) = (IAUC of food preparation/IAUC of reference food) × 100. Each food was assigned to the GI category as low (GI < 55), medium (GI > 55 and ≤ 69), or high (GI ≥ 70), as recommended in the past.28
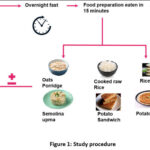 |
Figure 1: Study procedure. |
Statistical Analysis
All 15 participants were included in the statistical analysis (intent-to-treat population). Values with a coefficient of variation > 30 % and > 2 standard deviations (SDs) were considered outliers and excluded from the analysis. The data of nutrient composition, IAUC and GI of the foods are represented as the mean ± standard error of means (SEM) and analysed by independent ‘t’; test for difference between the groups. All analyses were performed using SAS (version 9.4, SAS Institute, Inc., Cary, NC, USA), with statistical significance set at P < 0.05.
Results
Demographic and clinical characteristics
Eight male (53%) and seven female (47%) participants were recruited for this study. The values of all demographic and clinical characteristics (Table 2) were within the normal physiological limits.
Table 2: Demographic and clinical characteristics of the study participants
|
Characteristics |
Mean ± SD |
|
Age (years) |
25.2 ± 5.1 |
|
Weight (kg) |
57.7 ± 5.8 |
|
Height (m) |
165.0 ± 8.8 |
|
Body mass index (kg/m2) |
21.1 ± 1.3 |
|
Waist circumference (cm) |
75.3 ± 5.2 |
|
Body fat (%) |
23.7 ± 7.3 |
|
Pulse (beats per minutes) |
76.0 ± 10.0 |
|
Systolic blood pressure (mmHg) |
111.6 ± 11.5 |
|
Diastolic blood pressure (mmHg) |
75.8 ± 8.0 |
|
Fasting blood glucose (mg/dl) |
85.2 ± 7.4 |
Number of participants = 15, SD – standard deviation, mmHg- millimetre of mercury
Nutrient composition
The nutrient composition of each food preparation is presented in Table 3. The available carbohydrate content in (Test) preparations was not significantly different (vs. Control).
Table 3: Nutrient composition of the food preparations with (Test) and without (Control) Fenuflakes addition.
|
Food Preparations |
Content in g (Mean ± SD) |
||||||
|
AC# |
Protein |
Fat |
TDF |
SDF |
IDF |
Ash |
|
|
Cooked raw rice |
|||||||
|
Control (Without Fenuflakes) |
50.1 ± |
6.5 ± |
0.5 ± |
2.4 ± |
0.8 ± |
1.6 ± |
0.2 ± |
|
Test (With Fenuflakes) |
49.7 ± |
9.7 ± |
2.0 ± |
6.2 ± |
2.0 ± |
4.2 ± |
0.7 ± |
|
Rice Idly |
|||||||
|
Control (Without Fenuflakes) |
50.2 ± |
8.3 ± |
0.4 ± |
4.4 ± |
0.6 ± |
3.9 ± |
2.0 ± |
|
Test (With Fenuflakes) |
49.9 ± |
11.3 ± |
0.5 ± |
12.1 ± |
3.0 ± |
9.1 ± |
3.5 ± |
|
Semolina upma |
|||||||
|
Control (Without Fenuflakes) |
50.1 ± |
8.7 ± |
7.7 ± |
7.9 ± |
0.9 ± |
6.3 ± |
4.1 ± |
|
Test (With Fenuflakes) |
50.0 ± |
13.1 ± |
8.9 ± |
20.5 ± |
4.1 ± |
16.4 ± |
6.2 ± |
|
Potato paratha |
|||||||
|
Control (Without Fenuflakes) |
50.1 ± |
11.7 ± |
4.2 ± |
15.3 ± |
2.2 ± |
13.1 ± |
2.9 ± |
|
Test (With Fenuflakes) |
50.0 ± |
13.1 ± |
4.5 ± |
22.4 ± |
5.0 ± |
17.6 ± |
1.9 ± |
|
Potato sandwich |
|||||||
|
Control (Without Fenuflakes) |
50.1 ± |
11.4 ± |
1.7 ± |
8.3 ± |
1.4 ± |
6.8 ± |
4.0 ± |
|
Test (With Fenuflakes) |
50.0 ± |
15.4 ± |
2.0 ± |
16.1 ± |
3.9 ± |
12.3 ± |
5.4 ± |
|
Oats porridge |
|||||||
|
Control (Without Fenuflakes) |
25.2 |
6.5 ± |
5.0 ± |
8.6 ± |
2.2 ± |
6.4 ± |
2.6 ± |
|
Test (With Fenuflakes) |
24.9 ± |
9.0 ± |
3.6 ± |
11.1 ± |
3.6 ± |
7.5 ± |
3.3 ± |
n = 3, # – Portion size of food sample in g, equivalent to 50 g (or 25 g for oat porridge) of AC (available carbohydrates) * P < 0.05 (v/s Control)
Effects on GI
The glucose levels and corresponding GI details are presented in Table 4. The average blood glucose response for each recipe has been presented in Figure 2.The differences in IAUC were statistically significant for cooked raw rice and potato paratha. Upon addition of Fenuflakes, the IAUC reduced for oats porridge by 8%, cooked raw rice by 35 % (P < 0.05), rice idly by 21 %, semolina upma, 5 %, potato sandwich and 8% and that of potato paratha by 27% (P < 0.05).
Furthermore, the addition of 10 g Fenuflakes to 25 or 50 g of available carbohydrate-containing portions of food significantly (P < 0.001) reduced the GI values of cooked raw rice (P < 0.001), idly (P < 0.001), and potato paratha (P < 0.01). The GI values of the Fenuflakes containing semolina upma, potato sandwich, and oat porridge were less, but the differences were not significant as compared to corresponding control food preparations.
The incorporation of Fenuflakes into various food preparations resulted in a significant modification of their GI values. The GI of Fenuflakes containing cooked raw rice shifted from a high GI category to a Low GI category. The GI of rice idly, semolina upma, and potato paratha showed a transition from the high GI category to the medium GI category with Fenuflakes addition. However, the addition of Fenuflakes to Potato sandwiches or oat porridge did not show any change in the GI category.
Table 4: Glycemic index (GI) of food preparations with (test) and without (control) Fenuflakes addition
|
Food Preparations |
Portion Size (g)$ |
IAUC (mg/minutes/dL) Mean ± SEM |
Glycemic Index |
|
|
Mean ± SEM |
Category# |
|||
|
Cooked raw rice |
||||
|
Control (Without fenuflakes) |
188 |
3928 ± 483 |
73.4 ± 5.9 |
High |
|
Test (With Fenuflakes) |
231 |
2573 ± 238* |
47.2 ± 2.3*** |
Low |
|
Rice Idly |
||||
|
Control (Without Fenuflakes) |
192 |
4183 ± 410 |
82.0 ± 3.8 |
High |
|
Test (With Fenuflakes) |
252 |
3295 ± 416 |
58.1 ± 3.6*** |
Medium |
|
Semolina upma |
||||
|
Control (Without Fenuflakes) |
273 |
3856 ± 426 |
76.3 ± 4.7 |
High |
|
Test (With Fenuflakes) |
284 |
3673 ± 317 |
67.4 ± 3.7 |
Medium |
|
Potato paratha |
||||
|
Control (Without Fenuflakes) |
182 |
4264 ± 457 |
78.8 ± 3.6 |
High |
|
Test (With Fenuflakes) |
206 |
3107 ± 323* |
58.9 ± 5.2** |
Medium |
|
Potato sandwich |
||||
|
Control (Without Fenuflakes) |
272 |
4224 ± 427 |
83.1 ± 6.4 |
High |
|
Test (With Fenuflakes) |
273 |
3905 ± 357 |
81.5 ± 8.2 |
High |
|
Oats porridge |
||||
|
Control (Without Fenuflakes) |
329 |
2421 ± 286 |
74.5 ± 4.5 |
High |
|
Test (With Fenuflakes) |
406 |
2219 ± 336 |
70.0 ± 7.5 |
High |
Number of participants = 15, SEM – Standard Error of Mean, CI – Confidence interval. *P < 0.05, **P < 0.01, ***P < 0.001 (Test v/s Control) by independent t-test.
$ – Portion size of food sample in g, equivalent to 50 g (or 25 g for oats porridge) available carbohydrates.
# GI category: low (GI < 55), medium (GI > 55 and ≤ 69), or high (GI ≥ 70).
 |
Figure 2: Average blood glucose response for each recipe. |
Discussion
The present study evaluated the impact of adding 10 g of Fenuflakes to Indian food breakfast items on their glycemic index (GI). The findings demonstrated that the incorporation of Fenuflakes led to a consistent decrease in IAUC and GI value across all food preparations when compared to their respective controls. Specifically, the addition of Fenuflakes to rice- and wheat-based dishes such as cooked rice, idly, upma, and paratha resulted in a reduction in postprandial glucose levels and GI, whereas bread- and oat-based preparations did not show similar beneficial effects on glucose levels or GI classification.
The GI values obtained from white rice-based preparations without Fenuflakes (controls), were consistent with reported values of cooked rice31 and steamed “idly” (a rice cake).32 The significant decrease in the GI value of Fenuflakes containing cooked rice from high to low in the current study can be attributed to the increased dietary fiber content. Additionally, upon the addition of Fenuflakes, the GI of idly was significantly reduced from high to medium, perhaps due to the combined effect of fibers present in Fenuflakes and urad dal. In addition, pulses and lentils are known as rich sources of protein, dietary fibers31, and nonabsorbable carbohydrates11, all of which might have contributed to their low GI values.
Upon the addition of water to roasted semolina, the resulting gelatinized product was found to have a high GI value in the present study. Semolina is a rich source of starch, which is responsible for blood glucose levels elevation33. However, upon the addition of Fenuflakes, the GI category was lowered to medium, perhaps because of higher amount of dietary fibres, which can reduce starch digestibility and postprandial blood glucose spikes.34
Boiled and mashed potatoes have a higher glycemic index (GI) than fried, microwaved, or baked forms of potatoes, mainly due to the degree of gelatinization and physical changes in the microstructure.35 In the present study, potato-based food preparations (potato paratha and sandwich) contained boiled and mashed potatoes and showed higher GI values. However, Fenuflakes addition to potato paratha (wheat dough and filling), significantly reduced the GI. At On the other hand, GI lowering effects was not observed for potato sandwich, where Fenuflakes are not added to bread dough. Our results are supported by previous studies where the addition of fenugreek seed powder to wheat flour (dough) increases the viscosity of foods36 and has a positive correlation with a reduction in postprandial glucose response.11
In case of oat porridge, addition of Fenuflakes did not change the IAUC or GI category. These results may be attributable to the similarity in physical properties, especially the particle size of both flakes (Fenuflakes and rolled oats/oat flakes). The smaller particle size and thinner profile of flakes (v/s grains) enables quicker hydration, promotes starch gelatinization led to enormous increase in GI values especially pre-heating conditions during porridge preparation.37
Taken together, GI lowering effects of Fenuflakes can be attributed to a higher content of dietary fibers. The dietary fibers are non-digestible carbohydrates that are not digested in the upper GIT. However, adding soluble dietary fibers delays the absorption of dietary carbohydrates38 and gastric emptying by forming a gel matrix 34.This gel matrix can reduce the rate of glucose diffusion to the absorptive surfaces of intestinal villi, leading to a reduction in blood glucose levels.39 Fenuflakes is rich in a water-soluble dietary fibres (such as galactomannans) might have played important role in the observed GI reduction properties and post-prandial glucose reduction observed in the current study, perhaps through delayed gastric emptying34 and modulation of gastrointestinal transit due to its physicochemical properties of viscosity, fermentability and water solubility.40. The inter-preparation variations of GI lowering effects of Fenuflakes may be due to procedural factors and differences in the physical characteristics of the tested food preparations. These differences need to be taken into consideration while extrapolating the results.
To the best of our knowledge, our study is first clinical evidence of GI lowering potential of fenugreek seed or constituent addition to commonly used breakfast food preparations using appropriate controls with robust procedures. The study was conducted as per validated protocols and ethical standards, practices and procedures as suggested by international organizations for GI measurements, food categorization and clinical study conduct. Moreover, the lack of bitter taste with improved palatability of fenuflakes makes it more useful ingredient for food preparations.. However, double-blind clinical study design could not be adopted due to characteristic smell of fenuflakes (although significantly less than fenugreek seeds) . Nevertheless, the open-label nature of the study does not compromise the validity of results as the outcome (plasma glucose levels) did not involve psychological or behavioral perceptions.
Conclusions
The present study showed the GI lowering potential of Fenuflakes when added to commonly used rice- wheat-based Indian breakfast food preparations. Additional studies with wide range of international food preparations can support Fenuflakes as a broader GI lowering option. .
Acknowledgements
We acknowledge the Contract Research Services of the Madras Diabetes Research Foundation, Chennai, India. India.
Funding Sources
This study was funded by Indus Biotech Limited, Pune, India (No. IBS455).
Conflict of interest
Indus Biotech Limited, Pune, India, funded the study but did not have a role in data collection, acquisition, analysis, or interpretation.
Data Availability Statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethics committee statement
The study protocol was approved by the Ethics Committee of the “Madras Diabetes Research Foundation (MDRF) (No: ECR/194/Inst/TN/2013/RR-19).
References
- Seal C. J., Courtin C. M., Venema K., de Vries J.(s). Health benefits of whole grain: effects on dietary carbohydrate quality, the gut microbiome, and consequences of processing. Comprehensive Reviews in Food Science and Food Safety. 2021; 20(3):2742-2768.
CrossRef - Magliano D. J., Boyko E. J., eds. IDF Diabetes Atlas. 10th ed. International Diabetes Federation; 2021.
CrossRef - Khan M. A. B., Hashim M. J., King J. K., Govender R. D., Mustafa H., Al Kaabi J.(s). Epidemiology of Type 2 Diabetes – Global Burden of Disease and Forecasted Trends. J Epidemiol Glob Health. 2019; 10(1):107.
CrossRef - Sachdev M., Misra A.(s). Heterogeneity of Dietary practices in India: current status and implications for the prevention and control of type 2 diabetes. European Journal of Clinical Nutrition. 2022; 77(2):145-155.
CrossRef - Tan D., Drewnowski A., Lê K. A.(s). New metrics of dietary carbohydrate quality. Curr Opin Clin Nutr Metab Care. 2023; 26(4):358-363.
CrossRef - Jenkins D. J., Wolever T. M., Taylor R. H., et al(s). Glycemic index of foods: a physiological basis for carbohydrate exchange. Am J Clin Nutr. 1981; 34(3):362-6.
CrossRef - Barclay A. W., Petocz P., McMillan-Price J., et al(s). Glycemic index, glycemic load, and chronic disease risk—a meta-analysis of observational studies. Am J Clin Nutr. 2008; 87(3):627-637.
CrossRef - Bhupathiraju S. N., Tobias D. K., Malik V. S., et al(s). Glycemic index, glycemic load, and risk of type 2 diabetes: results from 3 large US cohorts and an updated meta-analysis. Am J Clin Nutr. 2014; 100(1):218-232.
CrossRef - Vlachos D., Malisova S., Lindberg F. A., Karaniki G.(s). Glycemic Index GI) or Glycemic Load (GL) and Dietary Interventions for Optimizing Postprandial Hyperglycemia in Patients with T2 Diabetes: A Review. Nutrients. 2020; 12(6):1561.
CrossRef - Zhang K., Dong R., Hu X., Ren C., Li Y.(s). Oat-Based Foods: Chemical Constituents, Glycemic Index, and the Effect of Processing. Foods. 2021; 10(6)
CrossRef - Giuntini E. B., Sardá F. A., de Menezes E. W.(s). The Effects of Soluble Dietary Fibers on Glycemic Response: An Overview and Futures Perspectives. Foods. 2022; 11(23):3934.
CrossRef - Ngo T. V., Kunyanee K., Luangsakul N.(s). Insights into Recent Updates on Factors and Technologies That Modulate the Glycemic Index of Rice and Its Products. Foods. 2023; 12(19):3659.
CrossRef - Romão B., Falcomer A. L., Palos G., et al(s). Glycemic Index of Gluten-Free Bread and Their Main Ingredients: A Systematic Review and Meta-Analysis. Foods. 2021; 10(3):506.
CrossRef - Pasmans K., Meex R. C. R., van Loon L. J. C., Blaak E. E.(s). Nutritional strategies to attenuate postprandial glycemic response. Obesity reviews : an official journal of the International Association for the Study of Obesity. 2022; 23(9):e13486.
CrossRef - Murillo S., Mallol A., Adot A., et al(s). Culinary strategies to manage glycemic response in people with type 2 diabetes: A narrative review. Front Nutr. 2022; 9:1025993.
CrossRef - Kaur J., Kaur K., Singh B., Singh A., Sharma S.(s). Insights into the latest advances in low glycemic foods, their mechanism of action and health benefits. J Food Meas Charact. 2022; 16(1):533-546.
CrossRef - Augustin L. S. A., Kendall C. W. C., Jenkins D. J. A., et al(s). Glycemic index, glycemic load and glycemic response: An International Scientific Consensus Summit from the International Carbohydrate Quality Consortium (ICQC). Nutr Metab Cardiovasc Dis. 2015; 25(9):795-815.
CrossRef - Gong J., Fang K., Dong H., Wang D., Hu M., Lu F.(s). Effect of fenugreek on hyperglycaemia and hyperlipidemia in diabetes and prediabetes: A meta-analysis. Journal of ethnopharmacology. 2016; 194:260-268.
CrossRef - Pathak P., Srivastava S., Grover S.(s). Development of food products based on millets, legumes and fenugreek seeds and their suitability in the diabetic diet. Int J Food Sci Nutr. 2000; 51(5):409-14.
CrossRef - Kumar S. V., Rao R. J., Devi A. K., Mohanty S.(s). Comparative Study of Fenugreek Seeds on Glycemic Index In High And Medium Dietary Fiber Containing Diets In NIDDM Patients.: Comparative Study of Fenugreek Seeds on Glycemic Index. Nat J Integr Res Med. 2011; 2(3):29-37.
CrossRef - Robert S. D., Ismail A. A., Wan Rosli W. I.(s). Trigonella foenum-graecum seeds lowers postprandial blood glucose in overweight and obese individuals. J Nutr Metab. 2014; 2014:964873.
- Singh S., Chaurasia P. K., Bharati S. L.(s). Hypoglycemic and hypocholesterolemic properties of Fenugreek: A comprehensive assessment. Applied Food Research. 2023; 3(2):100311.
CrossRef - Thakurdesai P. A., Deshpande P. O., Pujari R. R., Gumaste S. A., Pore M. P.(s). Prenatal developmental oral toxicity evaluation of defatted fenugreek seed flakes rich in fibre and protein (FenuflakesTM) in laboratory rats. Curr Res Nutr Food Sci. 2023; 11(1)
CrossRef - Nevara G. A., Muhammad S. K. S., Zawawi N., Mustapha N. A., Karim R.(s). Dietary fiber: Fractionation, characterization and potential sources from defatted oilseeds. Foods. 2021; 10(4):754.
CrossRef - Singh R. B. Functional foods and nutraceuticals in metabolic and non-communicable diseases. Academic Press; 2021.
- FAO/WHO. Carbohydrates in human nutrition. Report of a Joint FAO/WHO Expert Consultation. 1998:1-140. FAO food and nutrition paper No 66. 14-18 April 1997.
- Mann J., Cummings J. H., Englyst H. N., et al(s). FAO/WHO Scientific Update on carbohydrates in human nutrition: conclusions. Eur J Clin Nutr. 2007; 61(S1):S132-S137.
CrossRef - Brouns F., Bjorck I., Frayn K. N., et al(s). Glycaemic index methodology. Nutr Res Rev. 2005; 18(1):145-171.
CrossRef - ISO. International Standard 26642: 2010—Food products–Determination of the glycaemic index (GI) and recommendation for food classification. 1st ed. Geneva, Switzerland: International Organization for Standardization; 2010. p. 11.
- Latimer G. W., ed. Official Methods of Analysis of AOAC INTERNATIONAL. 22nd ed. Oxford University Press Inc; 2023.
CrossRef - Kaur B., Ranawana V., Henry J.(s). The Glycemic Index of Rice and Rice Products: A Review, and Table of GI Values. Crit Rev Food Sci Nutr. 2016; 56(2):215-236.
CrossRef - Foster-Powell K., Holt S. H., Brand-Miller J. C.(s). International table of glycemic index and glycemic load values: 2002. Am J Clin Nutr. 2002; 76(1):5-56.
CrossRef - Biswas P., Jayaseelan P., Das M., Sikder A., Chaudhury K., Banerjee R.(s). Processing of semolina, a wonder resource for resistant starch production: In vitro digestibility and biochemical evaluation. Int J Biol Macromol. 2022; 222(Pt B):1918-1924.
CrossRef - Zhang H., Sun S., Ai L.(s). Physical barrier effects of dietary fibers on lowering starch digestibility. Curr Opin Food Sci. 2022; 48:100940.
CrossRef - Tahvonen R., Hietanen R. M., Sihvonen J., Salminen E.(s). Influence of different processing methods on the glycemic index of potato (Nicola). J Food Compost Anal. 2006; 19(4):372-278.
CrossRef - Sakhare S. D., Prabhasankar P.(s). Effect of Fenugreek fiber on Rheological and chapati making quality of whole wheat flour. Journal of Food Science and Technology. 2022:1-10.
- Tosh S. M., Chu Y.(s). Systematic review of the effect of processing of whole-grain oat cereals on glycaemic response. Br J Nutr. 2015; 114(8):1256-1262.
CrossRef - Weickert M. O., Pfeiffer A. F. H.(s). Impact of Dietary Fiber Consumption on Insulin Resistance and the Prevention of Type 2 Diabetes. J Nutr. 2018; 148(1):7-12.
CrossRef - Robert S. D., Ismail A. A., Rosli W. I.(s). Reduction of postprandial blood glucose in healthy subjects by buns and flatbreads incorporated with fenugreek seed powder. Eur J Nutr. 2016; 55(7):2275-80.
CrossRef - Müller M., Canfora E. E., Blaak E. E.(s). Gastrointestinal transit time, glucose homeostasis and metabolic health: modulation by dietary fibers. Nutrients. 2018; 10(3):275.
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.






