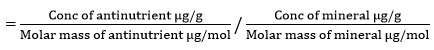Introduction
Common beans (Phaseolus vulgaris L.) are considered an affordable nutrient-rich food consisting of protein, vitamins, complex carbohydrates (slowly digestible starch and fiber), and micronutrients such as zinc and iron1. The protein in beans is high in lysine, which is limited in staple foods such as cassava, rice, and maize 2. In addition, the production of common beans is adaptable to global sustainable agriculture because of their ability to fix atmospheric nitrogen in the soil 3.
Production of beans in developing countries especially in Africa is lower than expected with an average of 893 kg/ha compared to 2.5-5 tons/ha which is achievable 4. The low yield is attributed to several field-based production constraints including poor agronomic practices, low input use, diseases, and pests among others 5. Plant breeding is one of the approaches used to overcome these challenges. Plant breeding is the art and science of modifying plant traits to produce desired characteristics 6. Breeding of beans aims at developing high-yielding bean varieties, drought tolerant, pests and diseases resistant, good tasting and cooking quality, and adaptable to mechanical harvesting and processing 7. In addition, breeding aims at improving the micronutrient quality of the beans especially iron and zinc content 8. Various international programs together with government institutions have successfully released drought-tolerant and biofortified beans rich in zinc and iron, in many countries 9 10.
Evaluating the nutritional value of the released new common bean varieties is important in combating mineral deficiency. Common beans constitute an integral part of the diet of many people in rural and poor urban communities in Africa who cannot afford red meat, chicken, and fish regularly4. Globally, an estimated two billion people are mineral deficient 11. Zinc and iron deficiencies are the main concern in developing countries12. Women of reproductive age and children under five years living in low-income countries are the most susceptible to micronutrient deficiencies1. Inadequate intake of zinc and iron is known to cause decreased resistance to infection in pregnant women, infants, and children, retard fetal growth and complications during pregnancy among many other negative health consequences13. Beans have the potential to fight against mineral deficiency and ensure food security.
The goal of this article was to evaluate the nutritional content, flatulence-causing factors, and antinutrients to mineral molar ratio of two newly developed bean varieties released in the Kenyan market. The RM 01 bean variety is drought tolerant while Faida is biofortified with high iron and zinc level.
Materials and Methods
Sample procurement and preparation
Faida and RM 01 (red mottled type) improved variety of beans, grown and harvested in the same season, were procured from Kenya Agricultural Research and Livestock Organization (KALRO), Machakos, Kenya. The bean samples were transported in water-resistant plastic bags to the laboratory. Once in the laboratory, the bean seeds were sorted to remove damaged grains and foreign materials. They were then conditioned at 25 °C and relative humidity of 60% in an incubator for 24 hours before analysis to equilibrate the moisture content. The seeds were milled using a grinder and sifted through a 50-mesh sieve size and kept in air-tight plastic bags for further analysis.
Determination of proximate composition
Crude ash, crude protein, crude fiber, crude fat, and moisture were quantified using AOAC methods 923.03, 979.09, 978.10, 920.29, and 925.09 respectively 14. The carbohydrate content was calculated by deducting the total of the dry matter percentages of ash, fiber, moisture, fat, and protein from 100 14.
Determination of oligosaccharides
The extraction of oligosaccharides was analyzed as described by Granito, Frias, Doblado Guerra, and Champ15. Two grams of bean flour were mixed with 20 ml of ethanol. The mixture was refluxed at 100 °C for 60 min with continuous shaking and passed through Whatman filter paper number 1. The solvent was dried and the sample was reconstituted with 2 ml of distilled water and acetonitrile solution (ratio of 1:1). Thereafter, the samples were passed through a 0.45 μm microfilter before injecting 20 μl into HPLC (Shimadzu CBM 20A, Japan) fitted with an APS-2 HYPERSIL (C-18) column (5 μm, 150 x 4.6 mm, Thermo Fisher) and a refractive index detector. Acetonitrile/water (63:35) was used as a mobile phase at a flow rate of 1 ml/min. Standard curves were determined using verbascose, stachyose, and raffinose standards.
Minerals determination
Zinc, calcium, and iron were quantified using AOAC, (2010)14 method.
Determination of antinutrients
Determination of tannins
Tannins content was determined using the Vanillin-HCl method of Ochanda, Onyango, and Mwasaru 16. A quantity of 250 mg of bean flour was weighed into a flask and mixed with ten ml of 4% HCI in methanol, shaken, and centrifuged at 4500 rpm for 10 min. The second extraction followed using 5 ml of 1% HCI in methanol. Using methanol, the aliquots from the two extracts were mixed to create a volume of 25 ml. Standards were prepared to range from 100 to 1000 ppm using methanol. The absorbance of sample extracts and blanks was read in a UV-VIS Spectrophotometer (Shimadzu, UV mini-1240, Japan) at 500 nm exactly 20 minutes after adding the Vanillin-HCI reagent to the samples. The quantity of tannin was conveyed in mg of catechin equivalent (CE) per gram of the sample (mg CE/g). Serial dilutions of catechin standard were made using methanol.
Determination of phytates
Phytates were extracted using HPLC as described by Ochanda, Onyango, and Mwasaru16 with some modifications. About 50 mg of samples were extracted using 10 ml of 3% H2SO4. The phytic acid was precipitated with a 3 ml FeCl3 solution. Twenty (20) μl was injected into HPLC (Shimadzu CBM 20A plus, Japan) fitted with a C-18 (μm) column at an oven temperature of 30° C. The mobile phase was a solution of 0.005N sodium acetate in distilled water with a flow rate of 1 μl/min. A standard solution was made using sodium phytate (inositol hexaphosphoric acid C6H6 (OPO3Na2)6+ H2O).
Determination of antinutrient to mineral molar ratios
Molar ratios of phytate to zinc and phytate to iron were determined to predict the bioaccessibility of these two minerals. The molar ratios were calculated using equation 117.
Antinutrient: mineral molar ratio
The molar masses for iron = 55.8 g/mol, zinc = 65.4 g/mol, phytic acid = 660.0 g/mol, 17.
Bioaccessibility determination of iron and zinc
Zinc and iron bioaccessibility was calculated using an in vitro method described by Sahuquillo, Barbera, and Farre18 with slight modification. The method employed three sequential phases: oral, gastric, and intestinal digestion. One gram of bean flour was weighed in three replications into 50 ml flasks and mixed with 1.5 ml of α amylase solution in each of the samples, then incubated for 10 minutes at 37 °C for 120 strokes. The activity of α-amylase was stopped by adjusting pH to 2.0 with 1 M HCl. Next, 2.5 ml of prepared pepsin solution was combined with the mixture above. The mixture was wrapped in parafilm and incubated for 2 hours at 37° C in a water bath shaking at 120 strokes per minute. Thereafter, the mixture was placed on ice for 10 minutes to halt pepsin digestion.
Before the intestinal digestion, the pH of the mixture was raised to 7 by adding 0.5 M NaOH. 12.5 ml of pancreatin-bile lipase solution was added to the mixture. Then incubated at 37 °C for 2 hours followed by a cooling process. Ten (10) ml of the digested sample was loaded onto the inside of the dialysis tube. The samples were put in a dialysis tube and dialyzed at room temperature for 24 hours. The solution from the dialyzing tube was transferred into separate test tubes and measured for iron and zinc content. Bioaccessibility (%) was calculated as follows:
Bioaccessibility (%) = 100 ×Y/Z………………………………………………………. Equation
Where Y is the element content of the bioaccessible fraction, and Z is the total iron or zinc beans content.
Data analysis
STATA for Windows version 12.1, 2011, was used to analyze the data. The results were presented as the mean values ± standard deviation (SD). To determine whether there were significant differences between the varieties, the data were subjected to an independent student t-test.
Results and Discussion
Proximate composition
A significant difference was observed in moisture content between Faida (7.85% ±0.10) and RM 01 (10.69% ±0.03) bean varieties as shown in Table 1. The values obtained in this study were comparable with other studies done for common bean varieties 1,19. However, the values were lower than (13.89-15.62%) those reported by Brigide, Canniatt-Brazaca, and Silva23 for biofortified bean varieties grown in Brazil. Variations in moisture content can be attributed to the difference in the bean variety and location. The low moisture content of dried beans facilitates their transportation and storability as well as prolongs their shelf life21. The final moisture content is critical in controlling how other components within the beans change. Beans with a moisture content above 13% significantly lose flavor and texture within six months 22.
Table 1: Proximate composition of improved beans grown in Kenya
|
Parameter (%) |
Faida | RM 01 |
P-value |
| Moisture | 7.85±0.10 | 10.69±0.03 | 0.0001 |
| Crude protein | 20.90±0.14 | 22.48±0.89 | 0.0392 |
| Carbohydrates | 60.04±0.16 | 54.34±1.12 | 0.0005 |
| Total ash | 4.12±0.06 | 3.98±0.04 | 0.0176 |
| Crude fat | 3.78±0.07 | 4.20±0.32 | 0.0434 |
| Crude fiber | 3.31±0.27 | 4.31±0.35 | 0.0001 |
|
Values given in the table are means of triplicate analyses (% wet basis). P ≤ 0.05. ± standard deviation |
|||
The RM 01 bean variety had a significantly higher protein content (22.48% ±0.89) than Faida (20.90% ±0.14). A study by Ojijo, Kimura, and Koaze20 reported higher values in the range of 25.3- 30.2% for Kenyan bean varieties. Brigide, Canniatt-Brazaca, and Silva23 also reported higher protein values of 23.38-31.59% for biofortified beans grown in Brazil. The protein content of these beans was comparable to those reported by Shimeli and Rakshit, (2005) 24 in Ethiopian-improved bean varieties and biofortified beans grown in Uganda25. The amino acid profile of bean protein is complementary to that of lysine-deficient cereals (maize, wheat, and rice) which are the staple food of people in developing countries 26,27. Encouraging diversification of the diet by increasing the intake of beans can be a food-based strategy for reducing the prevalence of protein malnutrition.
The crude fat content of the RM 01 variety (4.20% ± 0.32) was higher than that in Faida (3.78% ± 0.07). Crude fat observed in both varieties was higher than those reported by 19,28,29 for bean varieties. This could be attributed to differences in bean variety, cultivation practices, geographical location, and soil type.
Carbohydrates are major components of dry beans. A significant difference in carbohydrate content between the varieties was observed, with Faida having a higher content (60.04% ±0.16) than RM 01 (54.34% ±1.12). The carbohydrate content observed was within the range of 50-60% reported by other studies 1,22,31 for various bean varieties. The carbohydrate content of beans has a low glycemic index which is considered a therapeutic diet for diabetes patients1.
Total ash content differed significantly between the two varieties with RM 01 having a higher content of 4.12% ± 0.06 than Faida at 3.98% ±0.04. These results were comparable with the findings of Ojijo, Kimura, and Koaze20 who reported a total ash content range of 3.8-4.1% for Kenyan bean varieties. Higher content of 4.4 to 4.6% has been reported by Anino, Onyango, Samuel, Imathiu, Maina, and Onyangore28 for Kenyan bean varieties. Ash content is important because it corresponds to the quantity of minerals in the food.
The crude fiber content of the two varieties differed significantly. The results of this study were lower than the range of 4.6-7.4% reported for Kenyan bean varieties 28. Other studies24,32 have also reported higher values of 4.656-5.950% and 4.071-5.468% for improved beans. These differences could be explained by the bean variety and cultivation location. Beans are high-fiber food that helps in various physiological effects for human health33.
Oligosaccharides
Stachyose, raffinose, and verbascose are the predominant oligosaccharides 34. Oligosaccharides cause flatulence that deters consumers from eating beans. The human digestive tract lacks an α-galactosidase enzyme that hydrolyzes oligosaccharides, they are passed into the large intestine and fermented anaerobically by intestinal microorganisms 35, producing various gases such as hydrogen, methane, and carbon dioxide which may induce discomfort in humans 36.
Table 2: Oligosaccharides contents of Faida and RM01 bean varieties.
| Raffinose(mg/100g) | Stachyose(mg/100g) | Verbascose | |
| Faida | 414.83±8.96 | 1702.01±31.09 | ND |
| RM 01 P-value |
489.15±4.47 0.0001 |
1813.95±7.80 0.0001 |
ND |
Values in the table are means of triplicate analyses. ND-Not Detected. P≤ 0.05. ± Standard deviation
A significant difference was observed in oligosaccharides content as shown in Table 2. Stachyose was the main oligosaccharide in both bean varieties while verbascose was not detected in this study. These results are comparable to those published by 37 who reported a range of 1240 -1840 mg/100g stachyose in kidney beans grown in East Africa. Kitum, Kinyanjui, Mathara, and Sila38 observed slightly lower values of 1264 mg/100g stachyose for red haricot beans grown in Kenya.
The raffinose content was significantly different among the two varieties. The amount of raffinose observed in this study was higher compared to other studies37,38. The content of oligosaccharides in beans can be reduced by cooking, germination, soaking, and fermentation 35,39. Despite their negative effect, oligosaccharides are considered prebiotics with health benefits.
Mineral contents
Table 3 shows the mineral content of the Faida and RM 01 bean varieties. There were significant differences in iron, zinc, and calcium contents in the two bean varieties. The RM 01 bean variety had the highest calcium value of 1432.4 mg/Kg ±1.02, while Faida had the lowest value of 1280.7 mg/Kg ±1.32. These results were within the range of 850 -1900 mg/Kg reported for different common bean varieties 20,28,37.
The iron concentration of RM 01 was 49.3 mg/kg ±0.49 while Faida had 61.5 mg/kg ±0.70. The average Fe concentration of dry beans is approximately 50 mg/kg with a range of 32-92 mg/kg 40. Glahn, Wiesinger, and Lung’Aho41 reported a range of 55-94 mg/kg Fe content for 18 released biofortified East African bean varieties. While Wafula, Onduso, Saeys, Sila, and Hendrickx17 observed a range of 49.9-82.7 mg/kg for different Kenyan bean varieties. Biofortification breeding programs usually target to increase the Fe content by 50-100% from the existing baseline of 50-55 mg/Kg of most beans 42. Compared with the above-mentioned studies, RM 01 had lower iron content. It is worth mentioning that RM 01 variety is not a biofortified bean variety. The iron content can be influenced by genotype, environmental conditions, and agronomic practices 40.
Table 3: Mineral composition of the Faida and RM 01 bean varieties
| Variety | Fe(mg/Kg) | Ca(mg/Kg) | Zn(mg/Kg) |
| Faida | 61.5±0.70 | 1280.7±1.32 | 26.8±0.07 |
| RM 01 | 49.3±0.49 | 1432.4±1.02 | 21.3±0.10 |
| p-value | 0.0001 | 0.0002 | 0.0006 |
Each value is a mean of triplicate analyses. P≤0.05. ± standard deviation.
The zinc content varied significantly, with Faida having a higher concentration of 26.8±0.07 mg/Kg than RM 01 at 21.3±0.10 mg/Kg. These results are slightly lower than (22.6-50.1 mg/Kg) those observed by Wafula, Onduso, Saeys, Sila, and Hendrickx17 for Kenyan bean varieties. The baseline for zinc biofortification is 17 mg/kg, beans with 50% or 8.5 mg/kg above the goal level could be considered biofortified 8.
Anti-nutrients
Antinutrients including phytates and tannin affect mineral bioavailability. Metal ions, particularly zinc, iron, and calcium, are chelated by phytates in the gastrointestinal tract, forming insoluble complexes that are not digested or absorbed by humans 44. A significant difference was observed in phytic acid content between the two varieties. The phytic acid content for Faida and RM 01 beans was 11.70 mg/g ±0.05 and 7.60 mg/g ±0.05 respectively (Table 4). These values were lower than 16.8-23.4 mg/g cited by Wafula, Onduso, Saeys, Sila, and Hendrickx17 for Kenyan bean varieties. However, the phytate content in this study was higher than (2.23-2.74 mg/g) reported by Anino, Onyango, Samuel, Imathiu, and Maina28 for Kenyan bean varieties. The variation in phytic acid content could be attributed to variety, geographical location, soil type, and fertilizers.
Table 4: Anti-nutrients content of Faida and RM 01 bean varieties.
| Sample | Phytates (mg/g) | Tannins (mg CE /g) |
| Faida | 11.70±0.05 | 4.39±0.86 |
| RM 01 | 7.60±0.05 | 4.08±1.30 |
| P-values | 0.0011 | 0.0003 |
Each value is a mean of three replicates. P ≤0.05. ± standard deviation
There was a significant difference observed in tannin content between the two varieties (Table). The tannins content of the Faida and RM 01 were 4.39±0.86 mg CE/g and 4.08±1.30 mg CE/g respectively. These results were within the range (0.4-44.3 mg/g) reported by Wafula, Onduso, Saeys, Sila, and Hendrickx17 for Kenyan bean varieties. Tannins inhibit nutrient digestion and prevent the uptake of beneficial bioavailable components in the food. They bind to proteins forming tannin-protein complexes which inhibit digestive enzymes and lower protein digestibility due to interactions between protein substrate and ionizable iron44.
The molar ratios and bioaccessibility of iron and zinc of Faida and RM 01 bean varieties
Iron and zinc availability in the human body can be speculated by calculating the molar ratios of antinutrients to minerals 44. Table 5 shows the antinutrient-to-mineral molar ratios of the two bean varieties. Previous studies have reported that phytate to Fe molar ratio above 1 and phytate to Zn molar ratio higher than 15 have a negative effect on the bioavailability of Fe and Zn respectively45. The beans used in this study had phytate to Fe and phytate to Zn molar ratios that were above the thresholds necessary for efficient iron and zinc bioavailability (Table 5). These findings are comparable to those observed by Wafula, Onduso, Saeys, Sila, and Hendrickx17, who reported a Phytate to Fe molar range of 18.2-37.1 and a range of 39.2-83.4 for phytate to Zn molar ratios for Kenyan bean varieties. Castro-Alba, Lazarte, Bergenstahl, and Granfeldt 45 reported lower values of 7.94 and 17.4 for phytate to Fe and phytate to Zn respectively for kidney beans. The high phytic acid concentration in the beans used in this study could explain the low iron and zinc absorption.
Table 5: The antinutrient to mineral molar ratios and bioaccessibility of iron and zinc of the Faida and RM 01 bean varieties.
| Variety | Phy:Fe molar ratio | Phy:Znmolar ratio | Bioaccessibility (iron %) | Bioaccessibility (zinc %) |
| Faida | 17.08 | 42.26 | 29 | 42 |
| RM 01 | 15.19 | 35.36 | 35 | 65 |
Phy-phytates, Tan-tannins
Bioaccessibility is the percentage of a substance or nutrient in a food matrix that is released in the digestive system and subsequently made available for absorption through the intestinal mucosa45. The quality of minerals in food should be assessed by their mineral content and the percentage of the mineral that is available for use in biological processes40. Faida and RM 01 beans had bioaccessible iron percentages of 29% and 35% respectively (Table 5). Zinc bioaccessibility of RM 01 (65 %) was higher than Faida (42 %). Meaning RM 01 is a better source of iron and zinc compared to Faida which is biofortified and had higher iron and zinc content (Table 3). This might be explained by the fact that Faida contains more phytic acid as shown in Table 4. Mineral availability is affected by the presence of phytic acids, tannins, and dietary fiber 45. Direct comparison of these results with other studies18 on the bioaccessibility of minerals in beans is difficult because of differences in methods used in the determination of bioaccessibility. These methods vary on the conditions used in the simulation of gastrointestinal digestion such as temperature, pH, duration, enzymes, and agitation45.
Conclusion
Significant differences were observed in proximate composition among the two released improved bean varieties. Faida had significantly high carbohydrates and crude ash content while RM 01 was high in crude fat and crude protein. RM 01 reported higher values of flatulence-causing oligosaccharides while Faida was high in tannin and phytate content. The ratios of phytate to Zn and phytate to Fe, of the two varieties, were higher than the threshold levels for the effective bioavailability of these minerals. Bioaccessibility of iron and zinc contents was higher in RM 01 compared to Faida despite Faida having higher amounts of iron and zinc. Therefore, RM 01 is a better source of iron and zinc compared to the Faida bean variety. These findings reveal that a high mineral content does not translate to a high mineral bioaccessibility.
Acknowledgment
The authors acknowledge and thank the Department of Food Science and Technology at the Jomo Kenyatta University of Agriculture and Technology for providing materials for laboratory analysis. The authors gratefully acknowledge VLIR-UOS financial support through the Jomo Kenyatta University of Agriculture and Technology’s ‘Legume Centre of Excellence for Food and Nutrition Security (LCEFoNS),’ grant number KE2017IUC037A101.
Conflicts of Interest
There are no conflicts of interest reported by the authors. The authors are solely responsible for the manuscript’s content.
Funding Sources
This project has been funded by the VLIR-UOS through the Jomo Kenyatta University of Agriculture and Technology’s ‘Legume Centre of Excellence for Food and Nutrition Security (LCEFoNS),’ grant number KE2017IUC037A101.
References
- Hayat , Ahmad A., Masud T. Nutritional and Health Perspectives of Beans (Phaseolus vulgaris L.): An Overview. Taylor Fr. 2014;54(5):580-592. doi:10.1080/10408398.2011.596639
CrossRef - Sá G. A., Moreno Y. M. F., Carciofi B. A. M. Plant Proteins as High-Quality Nutritional Source for Human Diet. Trends Food Sci Technol. 2020;97:170-184. doi:10.1016/J.TIFS.2020.01.011
CrossRef - Roy S., Liu W., Nandety R. S., et al. Celebrating 20 Years of Genetic Discoveries in Legume Nodulation and Symbiotic Nitrogen Fixation. Plant Cell. 2020;32(1):15-41. doi:10.1105/TPC.19.00279
CrossRef - Huertas R., Karpinska B., Ngala S., et al. Biofortification of Common Bean (Phaseolus vulgaris L.) with Iron and Zinc: Achievements and Challenges. Food Energy Secur. Published online June 2022:e406. doi:10.1002/FES3.406
CrossRef - Mukankusi C., Raatz B., Nkalubo S., et al. Genomics, Genetics and Breeding of Common Bean in Africa: A review of Tropical Legume Lroject. Plant Breed. 2019;138(4):401-414. doi:10.1111/pbr.12573
CrossRef - Hallauer A. R. Evolution of Plant Breeding. Crop Breed Appl Biotechnol. 2011;11(3):197-206. doi:10.1590/S1984-70332011000300001
CrossRef - Svetleva D., Velcheva M., Bhowmik G. Biotechnology as a Useful Tool in Common Bean (Phaseolus vulgaris L.) Improvement. Euphytica. 2003;131(2):189-200. doi:10.1023/A:1023983831582
CrossRef - Beebe S. Biofortification of Common Bean for Higher Iron Concentration. Front Sustain Food Syst. 2020;4:206. doi:10.3389/FSUFS.2020.573449/BIBTEX
CrossRef - Overview L. A., Hayat I., Ahmad A., et al. Nutritional and Health Perspectives of Beans Nutritional and Health Perspectives of Beans ( Phaseolus vulgaris L .): 2014;8398. doi:10.1080/10408398.2011.596639
CrossRef - Buruchara R., Chirwa R., Sperling L., et al. Development and Delivery of Bean Varieties in Africa: The Pan-Africa Bean Research Alliance (PABRA) Model. African Crop Sci Soc. 2011;19(4):227-245.
- Mattar G., Haddarah A., Haddad J., Pujola M. New Approaches, Bioavailability and The Use of Chelates as a Promising Method for Food Fortification. Food Chem. 2022;373:13194.
CrossRef - Raes K., Knockaert D., Struijs K., & Van Camp J. Role of Processing on Bioaccessibility of Minerals: Influence of Localization of Minerals and Anti-nutritional Factors in the Plant. Trends Food Sci Technol. 2014;37:32-41.
CrossRef - Stammers A ., Lowe N ., Medina M. The Relationship Between Zinc Intake and Growth in Children Aged 1–8 years: A Systematic Review and Meta-Analysis. Eur J Clinical Nutr. Published online 2014.
CrossRef - Official Methods of Analysis of AOAC International. Agricultural Chemicals, Contaminants, drugs. edited by William Horwitz. Volume I. Published online 2010
- Granito M., Frias J., Doblado R., Guerra M., Champ M., Vidal-Valverde C. Nutritional Improvement of Beans (Phaseolus vulgaris) by Natural Fermentation. Eur Food Res Technol. 2002;214(3):226-231. doi:10.1007/s00217-001-0450-5
CrossRef - Ochanda S., Onyango C., Mwasaru M. Effects of Alkali Treatment on Tannins and Phytates in Red Sorghum, White Sorghum and Pearl Millet. J Appl. Published online 2010.
- Wafula E. N., Onduso M., Wainaina I. N., et al. Antinutrient to Mineral Molar Ratios of Raw Common Beans and Their Rapid Prediction Using Near-Infrared Spectroscopy. Food Chem. 2022;368:130773. doi:10.1016/J.FOODCHEM.2021.130773
CrossRef - Sahuquillo A., Barbera R., Farre R. Bioaccessibility of Calcium, Iron and Zinc from Three Legume Samples. Nahrung – Food. 2003;47(6):438-441. doi:10.1002/food.200390097
CrossRef - Kilonzi Sheila. Physicochemical and Functional Characterisation of Three Lablab bean (Lablab purpureus L. (Sweet) Varieties Grown in Kenya. thesis. Published online 2020:1-137.
- Ojijo K ., Kimura T., Koaze H., Composition, Soaking and Softening Characteristics of Some Kenyan Beans (Phaseolus vulgaris L.). Food Sci Technol Researh. Published online 2000:12-18.
CrossRef - Kyomugasho C., Kamau P., Aravindakshan Evaluation of Storage Stability of Low Moisture Whole Common Beans and Their Fractions Through the Use of State Diagrams. Food Res Int. 2021;140:109794.
CrossRef - Reyes‐Moreno C. O., Paredes‐López. Hard‐to‐Cook Phenomenon in Common Beans. A Review. Crit Rev Food Sci Nutr. 1993;33(3):227-286.
CrossRef - Brigide P., Canniatt-Brazaca S. G., Silva M. O. Nutritional Characteristics of Biofortified Common Beans. Food Sci Technol. 2014;34(3):493-500. doi:10.1590/1678-457x.6245
CrossRef - Shimeli E., Rakshit S. Proximate Composition and Physico-Chemical Properties of Improved Dry Bean (Phaseolus vulgaris L.) Varieties Grown in Ethiopia. LWT. 2005;38(2005):331-338
CrossRef - Nkundabombi M., Nakimbugwe D. Effect of Processing Methods on Nutritional, Sensory, and Physicochemical Characteristics of Biofortified Bean Flour. Food Sci Nutr. 2016;4(3):384-397. doi:10.1002/fsn3.301
CrossRef - Boye J., Canada A. F., Profile S., Zare F., Pletch A. Pulse Proteins: Processing, Characterization, Functional Properties and Applications in Food and Feed. Food Res Int. 2010;43(2):414-431. doi:10.1016/j.foodres.2009.09.003
CrossRef - Hall C., Hillen C. Composition, Nutritional Value, and Health Benefits of Pulses. Cereal Chem. 2017;94(1):11-31. doi:10.1094/CCHEM-03-16-0069-FI
CrossRef - Anino C., Onyango A. N., Imathiu S., Maina J., Onyangore F. Chemical Composition of The Seed and Milk of Three Common Bean (Phaseolus vulgaris L) Varieties. J Food Meas Charact. 2019;13(2):1242-1249. doi:10.1007/s11694-019-00039-1
CrossRef - Barros M., Prudencio S. Physical and Chemical Characteristics of Common Bean Varieties. org. 2016;37(2):751-762. doi:10.5433/1679-0359.2016v37n2p751
CrossRef - Maphosa Y. The Role of Legumes in Human Nutrition. Functional Food – Improved Heal through Adequate Food. Published online 2017. doi:10.5772/intechopen.69127
CrossRef - Los F. G. B., Zielinski A. A. F., Wojeicchowski J. P., Nogueira A. Demiate I.M. Beans (Phaseolus vulgaris L.): Whole Seeds with Complex Chemical Composition. Curr Opin Food Sci. 2018;19:63-71. doi:10.1016/J.COFS.2018.01.010
CrossRef - Natabirwa , Muyonga J. H., Nakimbugwe D., Lungaho M. Physico‐Chemical Properties and Extrusion Behaviour of Selected Common Bean Varieties. Wiley Online Libr. 2018;98(4):1492-1501. doi:10.1002/jsfa.8618
CrossRef - Tosh S., Yada S. Dietary Fibres in Pulse Seeds and Fractions: Characterization, Functional Attributes, and Applications. Food Res Int. 2010;43:450-460. doi:10.1016/j.foodres.2009.09.005
CrossRef - Singh B., Singh J. P., Shevkani K., Singh N., Kaur A. Bioactive Constituents in Pulses and Their Health Benefits. J Food Sci Technol. 2017;54(4):858-870. doi:10.1007/S13197-016-2391-9
CrossRef - Han I. H., Baik B. K. Oligosaccharide Content and Composition of Legumes and Their Reduction by Soaking, Cooking, Ultrasound, and High Hydrostatic Pressure. Cereal Chem. 2006;83(4):428-433. doi:10.1094/CC-83-0428
CrossRef - Granito M., Champ M., David A., Bonnet C., Guerra M. Identification of Gas-Producing Components in Different Varieties of Phaseolus Vulgaris by In Vitro Fermentation. J Sci Food Agric. 2001;81(6):543-550. doi:10.1002/JSFA.839
CrossRef - Shimelis E. A., Rakshit S. K. Effect of Processing on Antinutrients and In Vitro Protein Digestibility of Kidney Bean (Phaseolus vulgaris L.) Varieties Grown in East Africa. Food Chem. 2007;103(1):161-172. doi:10.1016/j.foodchem.2006.08.005
CrossRef - Kitum V., Kinyanjui P., Julius M. Effect of Lb. Plantarum BFE 5092 Fermentation on Antinutrient and Oligosaccharide Composition of Whole Red Haricot Bean (Phaseolus vulgaris L). Int J Food Sci. Published online 2020
CrossRef - Fernandes A. C., Nishida W., Da Costa Proença R. P. Influence of Soaking on The Nutritional Quality of Common Beans (Phaseolus vulgaris L.) Cooked With or Without The Soaking Water: A Review. Int J Food Sci Technol. 2010;45(11):2209-2218. doi:10.1111/j.1365-2621.2010.02395.x
CrossRef - Glahn R. P., Noh H. Redefining Bean Iron Biofortification: A Review of the Evidence for Moving to a High Fe Bioavailability Approach. Front Sustain Food Syst. 2021;5:215. doi:10.3389/FSUFS.2021.682130/BIBTEX
CrossRef - Glahn R. P., Wiesinger J. A., Lung’Aho M. G. Iron Concentrations in Biofortified Beans and Nonbiofortified Marketplace Varieties in East Africa are Similar. J Nutr. 2020;150(11):3013-3023. doi:10.1093/jn/nxaa193
CrossRef - Joshi-Saha A ., Sethy S., Misra G. Biofortified Legumes: Present Scenario, Possibilities and challenges. F Crop Res. 2022;279:108467
CrossRef - Read S. A., Obeid S., Ahlenstiel C., Ahlenstiel G. The Role of Zinc in Antiviral Immunity. Adv Nutr. 2019;10(4):696-710. doi:10.1093/ADVANCES/NMZ013
CrossRef - Luo Y., Gu Z., Han Y. The Impact of Processing on Phytic Acid, In vitro Soluble Iron and Phy/Fe Molar Ratio of Faba Bean (Vicia faba L.). J Sci food Agric. 2009;89(5):861-866. doi:10.1002/jsfa.3525
CrossRef - Castro‐Alba V., Claudia Phytate, Iron, Zinc, and Calcium Content of Common Bolivian Foods and Their Estimated Mineral Bioavailability. Food Sci Nutr. 2019;7(9):2854-2865. doi:10.1002/fsn3.1127
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.