Introduction
Heat induced gel formation is an important property of soy proteins for use as texture enhancing ingredient in food products.
The effects of several processing factors (heat, pH and ionic strength) on gel formation of soy proteins and the relationship between gel properties and network structure were studied by Renkema and van Vliet (2004) and Renkema et al. (2002a, b).
The rheological characteristics of model salad dressing emulsions, incorporating a dry-heated soybean protein isolate and dextran mixture (as emulsifier) were investigated by applying dynamic rheometry-tests to probe the emulsion structure and to elucidate the mechanism of their stability against creaming (Diftis et al., 2005).
Vegetable proteins are applied in a wide range of food products. Their high protein level and well-balanced amino-acid composition makes them important sources of protein, with a potential to replace meat and dairy proteins where it is appropriate. The rheological properties of vegetable protein gels as a function of heating time or temperature are discussed as well as the interfacial gelation upon adsorption of soy and wheat proteins at the air-water interface (van Vliet et al., 2002). It is well known that protein modification, i.e. denaturation, etc., improves protein functionality and application.
Batista et al. (2005) presented a comprehensive research on vegetable proteins about their macroscopic characteristics (i.e. gelling ability) by studying their molecular behavior when they were submitted to a thermal gelling process. The gels of soy, pea and lupin proteins were characterized by rheological techniques and gelation kinetics, mechanical spectra, as well as the texture of these gels were analyzed and compared. Additionally, capillary viscometry, steady-state fluorescence and fluorescence anisotropy were used to monitor the structural changes induced by the thermal denaturation, which constitutes the main condition for the formation of a gel structure.
Recent studies on gel formation and rheology of milk gels are reviewed by van Vliet et al. (2004). A distinction is made between gels formed by aggregated casein, gels of pure whey proteins and gels in which both casein and whey proteins contribute to their properties. For casein–whey protein mixtures, it has been shown that both the fraction whey protein aggregated with the casein particles and whey protein aggregates dispersed in the continuous phase have a large impact on the structural and mechanical properties of heated and acidified milk gels.
Whey protein gels prepared under acidic conditions (pH</4.6) remain largely unutilized because of their weak and brittle nature in contrast to the favorable elastic gels produced at neutral or basic conditions (Burke et al., 2002). However, such usage is important, as low pH food products are desirable due to their shelf stability and less stringent sterilization processes.
Burke et al (2002) used a two-step process involving enzyme followed by heat treatment to produce whey protein gels at low pH (4.0). Dynamic rheological measurements reveal that the gel elastic modulus and yield stress increase substantially when heat treatment is supplemented with enzyme treatment. Both the elastic modulus and yield stress increased with increasing enzyme concentration or treatment time.
The effect of microstructure on the fracture properties of whey protein isolate (WPI) gels with varying amounts of gelatin was analyzed by Brink et al. (2007) on the macro (200 µm scale) and micro (40 µm scale) levels. Eight percent WPI particulate gels with 0–6% gelatin were prepared at a pH near the isoelectric point of whey protein. The tensile stage was placed directly under the confocal laser-scanning microscope (CLSM). The structural changes of the gel during the deformation are visualized in series of micrographs with simultaneous recording of stress and strain data with the tensile stage. The pure whey protein gel exhibited uneven failure at the macro level, where the crack propagated between the whey protein clusters, whereas the crack propagated smoothly through the gelatin phase in the whey/gelatin gel system. At higher magnification the pure WPI protein gel showed porous failure behaviour and gradually ruptured.
The aim of this work was to study the rheological properties of a soy protein isolate and concentrate under varying conditions of temperature, concentration, and pH, as they related to gel strength of these materials.
Materials and Methods
Materials
A soy protein isolate SPI (90% protein min., Kjeldahl) of a medium to low viscosity, dispersible in water and/or other liquid systems can be used in various uncured poultry and red meat products as well as powdered dry mixes, which are later hydrated. This material is soluble over a wide range of pH (6-8). The SPI was completely dissolved at ambient temperatures (20-25°C) in double distilled, deionized water to the desired concentrations.
A soy protein concentrate (SPC) with very low flavour profile and high protein solubility has also been employed. A proximate analysis, indicates a protein content of approximately 70% as determined by the Kjeldahl methodology. Functionally, the product exhibits good dispersive ability, water binding and fat emulsification properties and can be used in a wide variety of foods. The greatest potential uses are where casein and non-fat dry milk are used such as meat, bakery products as well as nutritional powder drink bases. These products were obtained from ADM, Co.
Both products were dispersed in distilled water at the desired concentrations (3 and 5%) and were ready for the rheological determinations.
A preliminary three-level experimental design was undertaken to observe (macroscopically) the importance of three variables. The three variables and the levels assigned to each of them were as follows:
- pH varied from 6.0 to 8.0
- Temperatures varied from 8 to 60°C,
- Concentration varied from 3 to 10%
- Addition of 0.01 to 0.1 g of CaCl2 in 5% concentration
Rheological Measurements
A controlled strain Rheometrics Fluids Spectrometer RFS-8400 with cone and plate geometry was used to determine G΄(ω), G΄΄(ω) and n*(ω). The lower plate is driven by a servo motor which can be programmed for dynamic or steady shear experiments. The magnitude and phase of the torque response on the upper cone are measured by a 10 gm-cm/100 gm-cm alternative transducer supported by a precision air bearing. Sensitivity of the transducer is one thousandth of full scale.
Dynamic frequency scans of the storage and loss moduli were performed from 0.01 to 100 rad/s. In order to confine the measurements to approximately one minute duration, ten frequencies from 1 to 63.1 rad/s (or 1.16 to 10.04 Hz) were selected. This range represents about two decades of lower frequency similar to those monitored by Hsu (1992). Due to the sensitivity limits of the transducer on the Fluids Rheometer, the experiments had to be carefully performed at relatively appropriate concentrations (3 and 5% w/v). The sweep parameters of the instrument were settled at 65 last rate, 5 points per decade, 3.5 delay and the test geometry at 2.5 x E1 mm radius, and 0.02 (2E-2) rad angle.
For the measurements, 670 ml of SPI solutions were rapidly quenched to the measurements temperature in the rheometer using 25mm diameter cone with 0.02 rad angle. Temperature control was achieved by a circulating fluid bath for ethylene glycol and water. The temperature was controlled to within ±0.05°C due to its crucial effect on the sol-gel instant transformation process. The solutions were fresh or kept refrigerated before any measurement of viscoelastic properties was undertaken.
The instrument was set in the dynamic frequency sweep mode using 10 g.cm transducer and a strain input of 5%. For each time point, measurement of a set of ten frequencies from 1 to 63.1 rad/s takes approximately one minute. Thus, the frequency dependence of the storage and loss moduli was obtained for each of the ten frequencies, as well as the time dependence of moduli.
Results and Discussion
Effect of processing variables
A list of macroscopic observations of sol to gel formation for the SPI under various conditions included combinations of total solid concentration, pH and temperature variables, as applied to heat treatments of foods (Tables 1 and 2). The data shows the behavior of SPI in its sol to gel formation. Essentially, it is evident that all three factors including (i) concentration, (ii) pH, and (iii) temperature are involved in the degree of gelation of SPI.
The observations showed a light gelation effect at 8 and 20° C starting at pH 7, while a moderate gelation occurred when the temperature dropped from 68 to 8 and/or to 20° C.(note: at pH 6.5 no gelation was observed).
Table 1: Macroscopic observations of the effect of temperature and pH on the gelation of soy protein isolates at constant concentration of 5%.
|
pH |
|||||
|
Temperature (°C) |
6.0 |
6.5 |
7.0 |
7.5 |
8.0 |
|
8 |
– |
– |
+ |
++ |
++ |
|
20 |
– |
– |
+ |
+ |
++ |
|
68 |
– |
– |
++ |
++ |
++ |
| From68 to 8 |
– |
– |
++ |
++ |
++ |
| From 68 to20 |
– |
– |
++ |
++ |
++ |
-: no gel, +: light gelation, ++: moderate gelation
The observations showed a moderate gelation at 7% concentration and above and at temperatures 8 and 20° C, while at concentrations 6% and above and temperature 68°C very light gelation occurred.
Table 2: Macroscopic observations of temperature and soy protein concentration on the gelation of soy protein isolates
|
Soy protein concentration (w/w) |
||||||
|
Temperature (°C) |
4.0 | 5.0 | 6.0 | 7.0 | 8.0 | 10.0 |
|
8 |
– | – | + | ++ | ++ | ++ |
|
20 |
– | – | – | ++ | ++ | ++ |
|
68 |
– | – | + | + | + | + |
| from68 to 8 | – | – | ++ | |||
| from 68 to 20 | – | – | + | – | – | – |
| – | – | – | – | – | – | |
| – | – | – | – | – | – | |
-: no gel, +: very light gelation, ++: moderate gelation
Addition of 0.01 g CaCl2 in 5 % (w/w) concentration at all temperatures showed no gelation effect. Addition of 0.1 g CaCl2 in 5 % (w/w) concentration and at 8 and 20° C temperatures resulted in separation of the water, whereas at 68°C resulted in coagulation.of SPI. Addition of 0.05 g CaCl2 in 5 % (w/w) concentration and at 8.0 and 20.0° C resulted in separation of the water, whereas at 68°C resulted in coagulation of SPI. The polyelectrolyte theory should be mentioned here, taking into account the pH of the solutions, the ionic strength of the systems etc and the role of the protein under these conditions (Varzakas et al., 2011).
Table 3: The effect of temperature and concentration of Soy protein isolate on gel strength
|
Temperature (°C) |
Concentration % |
Gel Strength |
|
25 |
3 |
0.186 |
|
25 |
5 |
0.31 |
|
40 |
3 |
0.313 |
|
40 |
5 |
0.313 |
|
60 |
3 |
0.188 |
|
60 |
5 |
0.095 |
Figure 1 corresponds to SPI (60°C and 5% conc.), and is an example of the SPI rheological data. The relative gel strength (Egelandsdal et al, 1986) is calculated by to be 0.095 over the frequency range 1.59 to 63.1 radians.
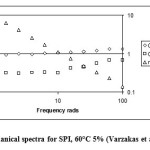 |
Figure 1: Mechanical spectra for SPI, 60°C 5% (Varzakas et al., 2011) Click here to View figure |
Table 4: The effect of temperature and concentration of Soy protein concentrate on gel strength
|
Temperature (°C) |
Concentration % |
Gel Strength |
|
25 |
3 |
0.2 |
|
25 |
5 |
0.21 |
|
40 |
3 |
0.146 |
|
40 |
5 |
0.164 |
|
60 |
3 |
0.07 |
|
60 |
5 |
0.08 |
The results for SPI can be summarised in terms of temperature, concentration and relative gel strength such that samples at 5% concentration show a decrease in relative gel strength with decreasing temperature, from 0.095 at 60°C to 0.313 at 40°C (Table 5).
Samples with 3% concentration hold relative gel strength steady with decreasing temperature from 60°C being 0.188 to 25°C being 0.186.
Figure 2 corresponds to SPC (60°C and 3% conc.). The relative gel strength over the frequency range 1.59 to 39.81 radians is 0.796. This indicates what could be referred to as a fluid gel (i.e. G’ > G’’), but the relative gel strength is tending towards fluidity.
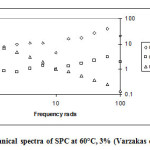 |
Figure 2: Mechanical spectra of SPC at 60°C, 3% (Varzakas et al., 2011) Click here to View figure |
The results for SPC can be summarised in terms of temperature, concentration and relative gel strength such that samples at 5% concentration show a decrease in relative gel strength with decreasing temperature, from 0.08 at 60°C to 0.146 at 40°C (Table 6).
Figures 3 and 4 show that at high temperature and high concentration we get weaker gels than at low temperature and low concentration. Lowering the concentration and keeping the temperature constant seem to have little effect on the gel strength, but points towards it becoming stronger.
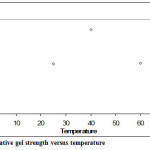 |
Figure 3: SPI relative gel strength versus temperature Click here to View figure |
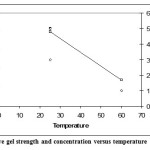 |
Figure 4: Relative gel strength and concentration versus temperature Click here to View figure |
Mechanism of sol and/or gel formation
A well known important fact of thermal effect on the SPI dispersions is that heating increases the protein-protein interactions at relatively high concentration and this resulted in an increase of solidity. These interactions between protein molecules lead to aggregation, coagulation or gelation in the protein dispersion (Catsimpoolas et al., 1970). Possible forces responsible for these interactions include hydrogen bonds, Van der Waals forces, ionic bonds, covalent bonds and hydrophobic associations. If the interactions between molecules include few cross-links and/or stretches of the molecules, the aggregates will increase in their hydration and will form fluid or viscous dispersions called “sols” or “gels”, respectively .
Since soy protein isolates are generally recognized as being soluble at low pH (<4.6), the loss of solubility and the development of solidity could be due to assessment of the extent of denaturation. However, heating at higher temperatures resulted in progressively lower soluble protein (Table 4). In other words, as heat-effect increases, hydrogen bonds are weakened. The hydrophobic effect is also changed by heating. The pH of the samples during heating affect the unfolding of soy protein isolate easily as well as the intermolecular interactions. Heating in the presence of CaCl2 to determine the role of hydrogen or ionic bonding and hydrophobic forces was attempted with a result of viscous sol or gel formation. Solidity at 10% soy protein concentration was more effective in gel formation than the lower concentrations (Table 3).
Conclusions
The concentration dependence of dynamic moduli G’ and G” of soy protein isolates (SPI) and soy protein concentrates were studied in solutions prepared under different conditions. Three-level preliminary fractional experiments were undertaken to study effects of temperature, pH, concentration, and salt (CaCl2) on the gelation of soy protein isolate and soy protein concentrate solutions. From the visual and calculated (experimental) results we can see that both of these variations may be related to the disruption of percolation clusters during the measurements. From the point of view of perculation theory it may be assumed that sol ‘monomer’ occupy the sites of a periodic lattice and bonds are formed randomly.
In summary, therefore, we conclude that all of these variables have an impact to the strength of these bio-gels and the gelation proceeds by a physical mechanism which is brought about by random interaction protein fold formation.
The relative gel strength is calculated by taking Log10 of the frequency values in Hz and the log10 of the corresponding G’ values in Pa. The relative gel strengths of samples at 60°C and 25°C show no real change in value, 0.188 to 0.186 at 60°C and 25°C respectively.
A light gelation effect was observed at 8 and 20° C starting at pH 7 and a moderate gelation as the temperature dropped from 68 to 8 and/or 20 C as well as at the higher pH of 7 and 8. On the other hand, moderate gelation was first seen at concentration 7.0 and above at temperatures 8 and 20° C, while at 68° C light gelation occurred.
Addition of 0.01 g CaCl2 in 5 % (w/w) concentration at all temperatures showed no effect Addition of 0.1 g CaCl2 in 5 % (w/w) concentration and at 8.0 and 20.0 C temperatures resulted in separation of the water, whereas at 68°C resulted in coagulation.of SPI. Addition of 0.05 g CaCl2 in 5 % (w/w) concentration and at 8.0 and 20.0° C resulted in water separation, whereas at 68°C resulted in coagulation of SPI..
Looking at SPC at 60°C 3% and taking the frequency range between 1.59 and 39.81 rads with the G’ values of 10.0 and 12.98 respectively the relative gel strength is given as 0.796. This indicates what could be referred to as a fluid gel. Looking at SPC at 40°C and 3% G’’ is greater than G’ and therefore one would expect this sample to be fluid. Taking the same frequency range as above, 1.59 and 39.81 with G’s being 14.43 and 23.14 respectively the relative gel strength is given as 0.146.
SPC at 25°C 3% and 5% shows a very similar relative gel strength, calculated to be 0.21.
Finally, it was shown that at low temperature and high concentration we get weaker gels than at high temperature and low concentration. Lowering the concentration and keeping the temperature constant seem to have little effect on the gel strength, but points towards it becoming stronger.
References
- Batista, A. P., Portugal, C. A.M., Sousa, I., Crespo, J. G., and Raymundo, A. 2005. Accessing gelling ability of vegetable proteins using rheological and fluorescence techniques. International Journal of Biological Macromolecules 36: 135–143.
CrossRef - Brink, J., Langton, M., Stading, M., and Hermansson, A-M. 2007. Simultaneous analysis of the structural and mechanical changes during large deformation of whey protein isolate/gelatin gels at the macro and micro levels. Food Hydrocolloids 21: 409–419.
CrossRef - Burke, M. D., Ha, S. Y., Pysz, M. A., and Khan, S. A. 2002. Rheology of protein gels synthesized through a combined enzymatic and heat treatment method. International Journal of Biological Macromolecules 31: 37-44.
CrossRef - Catsimpoolas, N., Funk, S.K. and Mayer, E.W. 1970. Thermal aggregation of glycinin subunits. Cereal chem. 47:331.
- Diftis, N.G., Biliaderis, C.G., and Kiosseoglou, V.D. 2005. Rheological properties and stability of model salad dressing emulsions prepared with a dry-heated soybean protein isolate-dextran mixture. Food Hydrocolloids, 19: 1025-1031.
CrossRef - Egelandsdal, B., Freitham, K., and Harbitz, O. 1986 Dynamic and rheological measurement of heat induced myosin gels: an evaluation of the methods suitability for filamentous gels. Journal of the Science of Food and Agriculture, 37: 944-954.
CrossRef - Hsu, S.H. 1992. The role of viscoelasticity and the sol-gel transition in the physiological function of biological fluids. Ph.D. Dissertation, CWRU, Cleveland, Ohio.
- Rector, D., Marsudomi, N., and Kinsella, J.E. 1991. Changes in gelling behavior of whey protein isolate and β-lactoglobulin during srorage: possible mechanism(s). J. Food Sci. 56:782.
CrossRef - Renkema, J.M.S., and van Vliet, T. 2004. Concentration dependence of dynamic moduli of heat-induced soy protein gels. Food Hydrocolloids, 18: 483-487.
CrossRef - Renkema, J.M.S., Gruppen, H., and van Vliet, T. 2002a. The influence of pH and ionic strength on heat-induced formation and rheological properties of soy protein gels in relation to denaturation and their protein composition. J. of Agric. And Food Chemistry, 50: 6064-6071.
CrossRef - Renkema, J.M.S., Knabben, J.H.M., and van Vliet, T. 2002b. Gel formation by β-conglycinin and glycinin and their mixtures. Food Hydrocolloids, 15: 407-414.
CrossRef - Rheology and structure of milk protein gels
- van Vliet, T., Lakemond, C. M.M., and Visschers, R. W. 2004. Current Opinion in Colloid & Interface Science 9: 298–304.
- van Vliet, T., Martin, A.H., and Bos, M.A. 2002. Gelation and interfacial behaviour of vegetable proteins. Current Opinion in Colloid & Interface Science 7: 462-468.
CrossRef - Varzakas, T., Labropoulos, A, and Anestis, S. 2011. Rheological Properties of a Soy Protein Isolate and Concentrate: Effect of Gel Strength. Proceedings of the 11th International Congress on Engineering and Food, 22-25 May, organised by NTUA, Athens, Greece. pp. 1103-1104.

This work is licensed under a Creative Commons Attribution 4.0 International License.




