Introduction
Tempe is a fermented food originated from Indonesia and has gained popularity in many parts of the world. This food is prepared by exposing soybeans to solid state fermentation by mixed culture of Rhizopus spp. Many studies suggested that tempe fermentation improved nutritional quality of soybeans. Isoflavones have mainly been the focus of the studies. During tempe fermentation, aglycone isoflavones, that possess higher functionality and bioavailability compared to their glycosidic counterparts,1,2 increased in quantity as the results of the fungal metabolism.3,4 Aglycone isoflavones, particularly genistein, have been known for its estrogen-like activity and cancer cell growth suppression.5 However, the effect is known to be dose-dependent.6 Besides isoflavones, other types of phenolic compounds are also present in soybean and tempe. However, many of the studies investigate the compounds that are extracted using
semi-polar solvents.3,7 While many known active phenolic compounds of tempe are semi-polar, it is of great importance to study the phenolic compound profiles derived from water extraction of tempe.
Water extraction is favored in this study because it reflects one type of tempe utilization in cuisines; it is used in the digestion/absorption process in the body; and water as the extraction solvent is safer for the environment. In addition, to the best of our knowledge, only limited number of studies utilized water for extraction while a considerable portion of phenols in soybeans are water-soluble.8 Moreover, semi-polar phenols, particularly isoflavones, may also be partially soluble in water. Besides phenols, water would potentially dissolve peptides, free amino acids, and water-soluble vitamins as well. During tempe fermentation, the cultures released proteolytic enzymes that helped hydrolyze proteins in soybeans into peptides and free amino acids.9-11 Some tempe peptides and amino acids demonstrated biofunctionality, including antihypertensive, antidiabetic, antioxidative and antitumor activity.12-13 It was also reported that water extracts of tempe could reduce the severity of enterotoxigenic Escherichia coli infection by preventing the bacteria attachment to intestinal wall cells.14
We were particularly interested in cytotoxicity against MCF-7 breast cancer cell line and 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging assay of aqueous extracts of tempe fermented at varying duration. Moreover, total phenolic compounds and total flavonoids were explored, as well as isoflavone identification and intensity comparison using LC-MS/MS to observe correlation between the abovementioned activities and phenolic compound profiles. The fermentation time was modified in order to find the optimum duration for the cytotoxicity or antioxidant activity of the extracts.
Materials and Methods
Materials
Soybean/tempe samples were prepared and fermented (for 24 hours) in Rumah Tempe Indonesia (RTI, Bogor, Indonesia) while the inocolum was obtained from PT. Aneka Fermentasi Indonesia. Ultrapurified water was used as solvent. All chemicals were of analytical grade, except for hexane (CAS: 110-54-3) for defatting that was purified technical grade solvent. DMSO (102952, CAS: 67-68-5), methanol (822283, CAS: 67-56-1), Folin Ciocalteaue (109001), sodium carbonate (106392, CAS: 497-19-8), and formic acid (822254, CAS: 64-18-6) were supplied by Merck Millipore (Burlington, Massachusetts, United States); RPMI (31800-022, LOT #1937557) and alamarBlue reagent (DAL1025, LOT #642297) were obtained from Thermo Fisher Scientific (Waltham, Massachusetts, United States); DPPH (D4313, CAS: 1898-66-4) was from Tokyo Chemical Industry (Tokyo, Japan); while alumunium chloride (254134, CAS: 12125-02-9), quercetin (CAS: 6151-25-3) and gallic acid (CAS: 149-91-7) were from Sigma-Aldrich (St. Louis, Missouri, United States).
Methods
Sample Preparation
Tempe production was performed following some established procedure that has been cited in several publications.7,12,15 Soybeans were cleaned and then boiled (100 °C, 30 min). After being kept for 22-24 hours in water containing lactic acid (pH 4.18, concentration of 1%), the soybeans were separated from its coat with a dehulling machine. The dehulled soybeans were then added with boiling water and soaked for 15 min. Later, they were strained and allowed to reach room temperature prior to subsequent processing.
One kilogram of the soybeans was collected and later defined as control. Meanwhile, the rest was mixed with 2 g inocolum/kg soybean (Raprima, PT. Aneka Fermentasi Indonesia, Bandung, Indonesia). The mixture was then transferred into perforated polyethylene bags and placed in a room (30-37°C; 70-85% humidity) for 24 hours. Some samples were harvested immediately after 24 hour fermentation, while others were placed in a room with good air circulation to allow further fermentation. Other samples were collected when fermentation time reached 48, 60, 72, and 120 hours. Samples were quickly placed in a freezer (-18°C) to stop the fermentation process.
Samples were thawed in a refrigerator (±4°C) for approximately 24 hours. Afterward, samples were weighed, sliced, and then dried in an oven (Venticell, MMM Medcenter Einrichtungen GmbH, Germany) at 50°C for 24 hours until most moisture (55-59% of the initial weight) was removed. Dried samples were weighed and later ground using Tokebi grinder (Neo Tokebi Plus, HausElec Co., Ltd., South Korea) until they turned into fine powder. The powder was then sieved and kept in an airtight container until further treatment.
Defatting and Extraction
Samples (approximately 90 g) were transferred into flasks and hexane was added (with sample to hexane ratio was 1:10). The flasks were put in a shaking incubator (DS-310C2, Dasol Scientific Co. Ltd, South Korea) for 2 hours with orbital shaking speed of 150 rpm. Afterward, samples were filtrated using filter papers under vacuum condition. The residues were then exposed to an airy condition in order to evaporate the remaining hexane from the residues. Later, they were weighed and transferred into the flasks again.
Deionized water (with water to sample ratio of 10:1) was added into the defatted samples in the flasks. The flasks were then again placed in the shaking incubator to undergo extraction for 2 hours at orbital shaking speed of 150 rpm. After the period ended, samples were filtrated using filter papers under vacuum condition. The filtrate was then placed in a rotary evaporator (Eyela NE Series, Tokyo Rikakikai Co. Ltd, Japan), with water bath temperature at 73°C and orbital speed at 15 rpm, until most of the water evaporated. The extracts were then weighed and placed in oven (50°C, overnight) to remove the remaining water.
Determination of total phenols
This assay was conducted using spectrophotometric technique.16 Samples were solubilized in deionized water to obtain concentration of 2 mg/ml. Gallic acid was diluted so that its concentrations in the reaction mixture were 0, 10, 20, 30, and 40 ppm. Samples (40 µl of sample solution with concentration of 2 mg/ml) were placed into each well. Deionized water was added to make the mixture volume reached 160 µl. We added 10 µl Folin Ciocalteau reagent and the mixture were incubated for 8 min. Finally, 30 µl sodium carbonate (10% w/v) was added and the mixture was allowed to react for 2 hours. The absorbance (λ 765 nm) was determined by using Varioskan™ Flash Multimode Reader (Thermo Scientific, Massachusetts, United States).
Determination of total flavonoids
Similar to total phenol assay, total flavonoid analysis was also based on spectrophotometry.17 Prior to analysis, sample extracts were solubilized in deionized water to obtain concentration of 2 mg/ml. Quercetin was used as the standard. The chemical was solubilized in methanol to obtain concentration of 100 ppm. Quercetin solution (0, 5, 10, 15, 20, and 25 µl) and samples (100 µl) were added into each well. Deionized water was added so that the volume reached 108 µl and followed by 6 µl of NaNO2 (5% w/v). After 5 min incubation period, 6 µl of AlCl3 (10% w/v) was added and the reaction mixture was allowed to react for 6 min. Later, 80 µl NaOH (1 M) (100 µl) was added. The absorbance was then read at wavelength of 510 nm using Varioskan™ Flash Multimode Reader (Thermo Scientific, Massachusetts, United States).
Determination of DPPH free radical scavenging activity
This antioxidant activity assay used radical 1,1-diphenyl picrylhydrazyl (DPPH).18 Water extracts were solubilized in deionized water to obtain concentration of 2 mg/ml. From each extract solution, samples were taken with a set volume (12, 30, 62, and 125 µl). Then, methanol was added until volume of methanol and extract mixture reached 200 µl. Afterwards, 50 µl of DPPH solution (from stock concentration of 40 ppm) was added. All of the samples were then placed at dark for 30 min. Later, the absorbance was measured at 515 nm using Varioskan™ Flash Multimode Reader (Thermo Scientific, Massachusetts, United States). Radical scavenging activity of extracts was expressed as inhibition concentration as follows:
![]()
with Ab as absorbance of blank while As as absorbance of samples. Later, the quantification results were used to extract concentration values at 50% inhibition (IC50) through extrapolation.
Determination of Cytotoxic Activity
Cytotoxic assay against MCF-7 cancer cells was determined using alamarBlueTM method.19 It measures viability of cancer cells by detecting molecular oxygen by addition of chromosphore resazurin that will transform to resafurin when it is reduced. 4 mg of samples were solubilized in 200 µl of dimethyl sulfoxide (DMSO). This stock solution was then diluted to obtain a series of concentration (20, 10, 5, 2.5, 1.25, 0.625, 0.3125, 0.156, 0.078, and 0.039 mg/L). Breast cancer cells MCF-7 in Roswell Park Memorial Institute (RPMI) medium (1×104 cells/ml) were plated in a 96-well plate (100 µl per well). The cells were kept for 24 hr and then the media was removed. Samples (5 µl) with abovementioned concentrations were added. After re-adding RPMI and mixing, the cells were incubated for another 24 hr. Later, 10 µl of alamarBlue reagent was added. The plates were then re-incubated for 4 hr at 37°C. Finally, the absorbance was measured at excitation wavelength of 560 nm and emission wavelength of 590 nm using Varioskan™ Flash Multimode Reader (Thermo Scientific, Massachusetts, United States). The data were then calculated as:
![]()
with CV as cell viability (%) and FI590 referred to fluorescence intensity at 590 nm emission (560 nm excitation). Negative control was prepared by adding 100 µl RPMI with 5 µl RPMI and 10 µl alamarBlue reagent. Positive control was prepared in a similar way, except for addition of 100 µl cancer cells in RPMI, instead of RPMI only. Logarithmic values of sample concentration were plotted as x-axis while percent viability was plotted as y-axis and inhibition concentration at 50% cell viability (IC50) was determined.
Isoflavone identification using LC-MS/MS
Detection of isoflavones were performed using LC-MS/MS consisting of a UPLC coupled to a quadrapole time of flight (QToF) (Waters Corporation, Milford, MA, United States). The samples were separated on an Acquity high strength silica (HSS) T3 column (1.8 µm particle size, 100 mm x 2.1 mm i.d, Waters Corporation, Milford, MA, United States), with gradient separation using combination of 0.1% formic acid in water (solvent A) and 0.1% formic acid in methanol (solvent B), as described in Table 1. The mass spectrometer was operated in negative electrospray ionization mode using MSE function. The condition for detection by the mass spectrometry was as follows: capillary voltage 1.5 kV; source temperature 120°C; cone gas flow 50 L/h; cone at 30 V; desolvation temperature 500°C; and desolvation gas flow 1000 L/h.
Table 1: Time, flow rate, and composition of solvent A and B for gradient separation of soflavones.
| Time (min) | Flow rate (ml/min) | Composition A (%) | Composition B (%) |
| 0.00 | 0.300 | 90.0 | 10.0 |
| 1.00 | 0.300 | 90.0 | 10.0 |
| 3.00 | 0.300 | 70.0 | 30.0 |
| 11.00 | 0.300 | 0.0 | 100.0 |
| 13.00 | 0.300 | 0.0 | 100.0 |
| 15.00 | 0.300 | 90.0 | 10.0 |
Statistical Analysis
All assays were performed with duplicates from two different sample preparation, except for LC-MS/MS analysis that only used one replicate. Data were analyzed using one-way analysis of variance (ANOVA) then processed further for significance of differences among treatments using Duncan’s multiple range test (p < 0.05). The statistical analysis was performed using IBM SPPS Statistics for Windows (IBM Corporation, Armonk, New York, United States).
Results
Determination of total phenols
Our data showed that concentrations of water-soluble phenols increased as soybean was fermented up to 60 hr. A very obvious increase was observed between 24 and 48 hours (Fig 1). From 48 to 120 hr, the concentrations were steady with slight decrease at 72-hr point. Meanwhile, for total phenols in the aqueous extracts of tempe, the concentrations decreased steadily as fermentation occurred between 0 and 60 hr. After 60 hr, an insignificant rise was detected but after 72 hr, the values declined again. The data also demonstrated that the maximum concentration of total phenols in the extracts was observed at control (Fig 2).
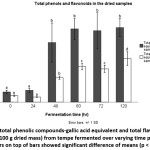 |
Figure 1: Level of total phenolic compounds-gallic acid equivalent and total flavonoids-quercetin quivalent (mg/100 g dried mass) from tempe fermented over varying time periods. Different letters on top of bars showed significant difference of means (p < 0.05) Click here to View figure |
 |
Figure 2: Level of total phenolic compounds-gallic acid equivalent and total flavonoids-quercetin equivalent (mg/g extract) of aqueous extracts of tempe fermented over varying time periods. Different letters on top of bars showed significant difference of means (p < 0.05) Click here to View figure |
Determination of total flavonoids
Similar to total phenols, total flavonoids in the dried samples also tended to increase as fermentation was prolonged but in less obvious linear correlation as data at 24 and 72 hr disrupted the expected trend. The values decreased slightly between 0 and 24 hr and between 60 and 72 hr (Fig 1). Meanwhile, data of total flavonoids present in the extracts decreased as soybean was fermented up to 48 hr. Afterward, the data slightly increased when fermentation was prolonged to 120 hr with an exception of an insignificant decrease between 60 and 72 hr (Fig 2).
Determination of DPPH free radical scavenging activity
DPPH antioxidant data did not show any straightforward relationship between fermentation time and IC50 values of the aqueous extracts. The lowest mean average for IC50 was observed at 24-hour fermentation (Fig 3), indicating that the treatment led to the highest DPPH free radical scavenging activity. A very notable increase in IC50 values was observed when fermentation time was extended from 60 to 72 hr. The maximum value was achieved at 72-hr period, showing low antioxidant activity. Further fermentation resulted in a sharp decline in IC50 values of the aqueous extracts.
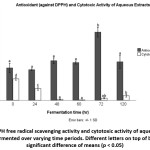 |
Figure 3: DPPH free radical scavenging activity and cytotoxic activity of aqueous extracts of tempe fermented over varying time periods. Different letters on top of bars showed significant difference of means (p < 0.05) Click here to View figure |
Determination of Cytotoxic Activity
Breast cancer, which is prevalent among women, occurred as the result of abnormal cell growth that is affected by many factors including diet. Some food components may enhance the proliferation while others suppress it. The suppression or cytotoxic activity could be performed on culture cells. The cytotoxic activity was expressed as concentration that inhibited 50% of the proliferation of the cancer cells MCF-7 (IC50). Lower IC50 values were commonly associated with higher cytotoxic activity of the extracts on the growth of the investigated cancer cell line which was MCF-7 for this study. Our data revealed a distinct relationship between fermentation time and cytotoxic activity. The IC50 values decreased as fermentation was performed from 0 (control) to 60 hr. After reaching the minimum value (8.7±4.95 µg/ml) at 60 hr, the values increased slightly as fermentation was extended from 60 to 120 hr.
Isoflavone identification using LC-MS/MS
Genistein was found at water extracts of non-fermented soybean and tempe fermented for 24 up to 120 hr. It was present at relatively high amount at non-fermented and 24-hr fermented tempe extracts. Extracts resulted from fermentation for 48, 60, 72, and 120 hr contained very minimum amount of genistein; however, there were slight increase in genistein content when fermentation time was extended from 48 to 60 hr but declined promptly when fermentation was further prolonged.
Similar to genistein, genistin, daidzein and daidzin were predominant at aqueous extracts of control (non-fermented soybean) and briefly (24 hr) fermented tempe (Fig 4). Afterward, the concentrations diminished remarkably, even to no response level for genistin and daidzin at 72 hr. Glycitein was observed at relatively low response for all treatments but somewhat higher at control and 24-hr fermented tempe. Meanwhile, glycitin was not detected in either control or tempe at all given fermentation durations.
 |
Figure 4: Comparison of LC-MS/MS response of genistein, genistin, daidzein, daidzin, glycitein and glycitin of tempe fermented at varied fermentation time Click here to View figure |
The composition and percentage of each isoflavone was given in Fig 5. Aqueous extracts of control and 24-hour fermented tempe had very similar proportion of the isoflavones. Composition of 48-hour tempe extract was also identical, but the percentages of both genistin and daidzin were larger while the proportion of genistein and daidzein declined. A notable change was observed after 48 hour fermentation. Portion of genistein and daidzein increased drastically while that of genistin and daidzin decreased. Similar finding was observed for 72-hr treatment.
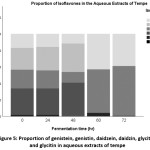 |
Figure 5: Proportion of genistein, genistin, daidzein, daidzin, glycitein and glycitin in aqueous extracts of tempe Click here to View figure |
Discussion
Our findings suggested that tempe fermentation altered total water-soluble phenolic compound and flavonoid profile of the products, as well as DPPH free radical scavenging activity, cytotoxicity and isoflavone profile of the extracts. Information regarding total phenolic compounds in food materials has been included in many studies on food functionality because this group of chemicals has been reported to show many health-promoting effects. Soybeans contained various phenolic compounds and water-soluble phenols comprised of approximately 68-81% of the total phenolic compounds.8 In general, the present study demonstrated that total water-soluble phenolic compounds of tempe increased as fermentation time was extended from 0 to 60 hr and afterward it tended to became steady. Total phenolic content in the dried unfermented soybean was much lower than that demonstrated by Kumar et al.,20 and Alu’datt et al.,8 but it was expected considering the unfermented soybean in this present study had been clean, dehulled, soaked, boiled, and defatted as the steps were also performed on other treatments as preparation for fermentation and aqueous extraction. We predicted that some phenolic compounds were lost during the process. The most significant increase of total phenol content in the dried mass was observed between 48 and 60 hr fermentation, somewhat different with the findings of Kuligowski et al.,3 that showed the largest increase was between 24 and 48 hr fermentation. However, this dissimilarity was expected as it might be a consequence of different Rhizopus strains and fermentation condition. On the other hand, the relative steadiness of total phenolic content after 48 hr observation was similar to that of Kuligowski et al.,3 particularly of the Rhizopus oligosporus strain 2710.
Unfermented full fat soybean contained 10-40% of bound phenolic compounds.8 During fermentation, the mold Rhizopus oligosporus released hydrolytic enzymes that cleaved proteins, lipids, and starch11 and consequently increased free water-soluble phenolic compounds as demonstrated in our findings and also confirmed by previous studies.3,21-22
Similar to total phenol in the dried mass of samples, our study showed that total flavonoids were inclined to increase as fermentation was prolonged up to 120 hr. Soybeans have been well known for its high flavonoid content that surpasses that of peanut, cowpea, mung bean, and lablab.23 Total flavonoids of our samples, expressed as quercetin equivalent, reached the highest points (62.59 mg/100 g dried mass) at 120-hr fermented tempe. This amount is somewhat lower than total flavonoids in soybeans (892.3-916.5 µg/g) as observed by Wang et al.,23 This inconsistency should be attributed to different sample tested (we used aqueous extracts while they used untreated soybeans and different analytical technique: they used HPLC analysis to quantify total of five flavonoids: myricetin, daidzein, quercetin, genistein and kaempferol while spectrophotometric method using quercetin as the standard was used in the present study. Besides, different soybean variety and weather condition where it was grown also might play a significant role.24
Flavonoids and other phenolic compounds are often associated with health functionality. Flavonoids in plants consists of anthoxanthins (flavones and flavonols); flavanones; flavanonols; flavans (flavan-3-ols, flavan-4-ols, and flavan-3,4-diols); anthocyanidins and isoflavonoids.25-26 While many compounds that belong to the group, for instance genistein; daidzein; quercetin; kaempferol, have been confirmed as antioxidants,27 higher total phenols or flavonoids do not necessarily mean more intense antioxidant activity.20,28 Our data also demonstrated low correlation between IC50 DPPH free radical scavenging activity with either total phenols (r=-0,432) or total flavonoids (r=0,534) (Table 2). This could be a result of antagonistic effects of some compounds29-31 or that the active compounds are not present in the amount at which they are active as many functionality of the compounds are dose-dependent.6,27
Table 2: Correlation between total phenols, total flavonoids, IC50-antioxidant activity, and IC50-cytotoxic activity.
| Parameter | IC50 – Cytotoxic Activity | IC50 – Antioxidant Activity | Total phenols | Total flavonoids |
| IC50 – Cytotoxic Activity | -0.476 | 0,926 | 0,956 | |
| IC50 – Antioxidant Activity | -0,477 | -0,432 | -0,534 | |
| Total phenols | 0,926 | -0,432 | 0,865 | |
| Total flavonoids | 0,956 | -0,539 | 0,865 |
A surprising fact was observed for relationship between cytotoxic activity and total phenols or flavonoids. Correlation coefficient of IC50 cytotoxicity and total phenols and flavonoids are very positive and indicative of strong correlation: 0.926 and 0.956 for IC50 cytotoxicity – total phenols and IC50 cytotoxicity – total flavonoids, respectively (Table 2). It signaled an inverse strong correlation between cytotoxic activity of the aqueous extracts of tempe against MCF-7 cancer cells and total phenols or flavonoids. This might be explained by negative effect of genistein when present at high concentration. LC-MS/MS responses showed that genistein was very intense at control and 24-hr fermented tempe extracts, and then drastically dropped to almost non-existent, except for a slight increase at 60-hr observation. Genistein had been reported to have both positive and negative effect in regards of cancer cell proliferation. It was believed to act as proliferation inhibitor at low concentration but induce the opposite effect as the concentration was increased.6
Our LC-MS/MS response for each isoflavone of aqueous extracts from tempe fermented at varying time demonstrated that both genistein and daidzein, two important isoflavone aglycones, were present at very high quantity at control and 24-hr tempe extracts, then the values dropped drastically to almost non-existent at 120-hr observation, with a slight increase at 60 hr. Mo et al. argued that soybean variety, not fermentation, that largely contributed to concentration of isoflavone aglycones in soybean products because they found out that the level in tempe was not higher than that in tofu.7 Another possible explanation was that because water was not the best solvent for soybean isoflavone extraction,32 it could be that aglycone level increase during fermentation, as shown as increasing ratio of aglycones to their glycosides (Fig. 5), but only a portion was present in the water extracts.
Conclusion
We confirmed that tempe fermentation enhanced the release of water-soluble phenols, particularly flavonoids, and it was likely a consequence of hydrolysis promoted by the microorganisms. At several periods of fermentation, the extracts demonstrated cytotoxic activity, as expressed by noticeable reduction of MCF-7 cancer cell growth. In contrast, all extracts did not show any appreciable DPPH free radical scavenging activity. A deeper investigation on the extracts showed that the cytotoxic activity was inversely correlated with either total phenols or flavonoids and it was supported by LC-MS/MS data, suggesting that very high concentration of the compounds, particularly genistein, might trigger cancer cell proliferation. Prolonging fermentation up to 60 hr would result in low but optimum concentration of the isoflavones to achieve the intended cancer inhibition effect. To achieve a better understanding of this subject, the study should be expanded to investigation of isoflavone release during gastrointestinal digestion after tempe consumption in order to achieve recommended intake of the product.
Acknowledgements
This study was funded by Indonesian Institute of Sciences and The World Academy of Sciences (TWAS). We would like to thank Dr. Rizna Triana Dewi, Mrs. Euis Filaila, Ms. Faiza Maryani, and Mr. Rokib for having shared valuable inputs in this study.
Conflict of Interest
We confirm that there are no known conflicts of interest associated with this study.
References
- Setchell K. D. R., Brown N. M., Zimmer-Nechemias L., Brashear W. T., Wolfe B. E., Kirschner A. S., Heubi J. E. Evidence for lack of absorption of soy isoflavone glycosides in humans, supporting the crucial role of intestinal metabolism for bioavailability. J. Clin. Nutr. 2002; 76:447-453.
CrossRef - Soni M., Rahardjo T. B. W., Soekardi R., Sulistyowati Y., Lestariningsih, Yesufu-Udechuku A., Irsan A., Hogervorst E. Phytoestrogens and cognitive function: a review. Maturitas. 2014; 77:209-220. http://dx.doi.org/10.1016/j.maturitas.2013.12.010
CrossRef - Kuligowski M., Pawłowska K., Jasińska-Kuligowska I., Nowak J. Isoflavone composition, polyphenols content and antioxidative activity of soybean seeds during tempeh fermentation. CyTA-J Food. 2016; 15:27-33. https://doi.org/10.1080/19476337.2016.1197316
CrossRef - Ahmad A., Ramasamy K., Majeed A. B. A., Mani V. Enhancement of β-secretase inhibition and antioxidant activities of tempeh, a fermented soybean cake through enrichment of bioactive aglycones. Pharm Biol. 2015; 5:758-766.
CrossRef - Li H. Q., Luo Y., Qiao C. H. The mechanisms of anticancer agents by genistein and synthetic derivatives of isoflavone. Mini-Rev Med. Chem. 2012; 12:350-362.
CrossRef - Russo M., Russo G. L., Daglia M., Kasi P. D., Ravi S., Nabavi S. F., Nabavi S. M. Understanding genistein in cancer: the “good” and the “bad” effects: a review. Food Chem. 2016; 196:589-600. https://doi.org/10.1016/j.foodchem.2015.09.085
CrossRef - Mo H., Kariluoto S., Piironen V., Zhu Y., Sanders M. G., Vincken J., Wolkers-Rooijackers, J., Nout M. J. R. Effect of soybean processing on content and bioaccessibility of folate, vitamin B12 and isoflavones in tofu and tempe. Food Chem. 2013; 141:2418-2425. https://doi.org/10.1016/j.foodchem.2013.05.017
CrossRef - Alu’datt M. H., Rababah T., Ereifej K., Alli I. Distribution, antioxidant and characterisation of phenolic compounds in soybeans, flaxseed and olives. Food Chem. 2013; 139:93-99. https://doi.org/10.1016/j.foodchem.2012.12.061
CrossRef - Baumann U., Bisping B. Proteolysis during tempe fermentation. Food Microbiol. 1995; 12:39-47. https://doi.org/10.1016/S0740-0020(95)80077-8
CrossRef - de Reu J. C., ten Wolde R. M., de Groot J., Nout M. J. R., Rombouts F. M., Gruppen H. Protein hydrolysis during soybean tempe fermentation with Rhizopus oligosporus. Agric. Food Chem. 1995; 43:2235-2239. DOI: 10.1021/jf00056a050
CrossRef - Handoyo T., Morita N. Structural and functional properties of fermented soybean (tempeh) by using Rhizopus oligosporus. Int J Food Prop. 2006; 9:347-355. https://doi.org/10.1080/10942910500224746
CrossRef - Tamam B., Syah D., Suhartono M G., Kusuma W. A., Tachibana S., Lioe H. N. Proteomic study of bioactive peptides from tempe. Biosci. Bioeng. 2019; https://doi.org/10.1016/j.jbiosc.2019.01.019
CrossRef - Koh S. P., Jamaluddin A., Alitheen N. B., Mohd-Ali N., Yusoff, H. M., Long K. Nutritional values of tempe inoculated with different strains of Rhizopus: its ɣ-aminobutyric acid content and antioxidant property. Trop. Agric. And Fd. Sc. 2012; 40:181-192.
- Roubus-van den Hil P. J., Nout M. J. R., van der Meulen J., Gruppen H. Bioactivity of tempe by inhibiting adhesion of ETEC to intestinal cells, as influenced by fermentation substrates and starter pure cultures. Food Microbiol. 2010; 27:638-644. doi:10.1016/j.fm.2010.02.008
CrossRef - Utama Z., Okazaki Y., Tomotake H., Kato N. Tempe consumption modulates fecal secondary bile acids, mucins, immunoglobulin A, enzyme activities, and cecal microflora and organic acids in rats. Plant Foods Hum. Nutr. 2013; 68:177-183. Doi: 10.1007/s11130-013-0357-x
CrossRef - Singleton V. L., Orthofer R., Lamuela-Raventos R. M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folic-ciocalteu reagent. Methods Enzymol. 1999; 299:152-178.
CrossRef - Quettier-Deleu C., Gressier B., Vasseur J., Dine T., Brunet J., Luyck M., Cazin M., Cazin J. C., Bailleul F., Trotin F. Phenolic compounds and antioxidant activities of buckwheat (Fagopyrum esculentum Moench) hulls and flour. J Ethnopharmacol. 2000; 72:35-40.
CrossRef - Singh R., Singh B., Singh S., Kumar N., Kumar S., Arora S. Investigation of ethyl acetate extract/fractions of Acacia nilotica wild Ex Del as potent antioxidant. Rec Nat Prod. 2009; 3:131-138.
- Forster G. M., Raina K., Kumar A., Kumar S., Agarwal R., Chen M. H., Bauer J. E., McClung A. M., Ryan E. P. Rice varietal differences in bioactive bran components for inhibition of colorectal cancer cell growth. Food Che 2013; 141:1545-1552. https://doi.org/10.1016/j.foodchem.2013.04.020
CrossRef - Kumar V., Rani A., Dixit A. K., Pratap D., Bhatnagar D. A comparative assessment of total phenolic content, ferric reducing-anti-oxidative power, free radical-scavenging activity, vitamin C and isoflavones content in soybean with varying seed coat colour. Food Res Int. 2010; 43:323-328. https://doi.org/10.1016/j.foodres.2009.10.019
CrossRef - Ademiluyi A. O., Oboh, G. Antioxidant properties of condiment produced from fermented bambara groundnut (Vigna subterranea Verdc). J. Food Biochem. 2011; 35:1145-1160. https://doi.org/10.1111/j.1745-4514.2010.00441.x
CrossRef - Starzyńska-Janiszewska A., Stodolak B., Duliński R., Baczkowicz M., Mickowska B., Wikiera A., Byczński Ł. Effect of solid-state fermentation tempe on antioxidant and nutritional parameters of buckwheat groats as compared with hydrothermal processing. J Food Process Pres. 2016; 40:298-305. https://doi.org/10.1111/jfpp.12607
CrossRef - Wang M. L., Gillaspie A. G., Morris J. B., Pittman R. N., Davis J., Pederson G. A. Flavonoid content in different legume germplasm seeds quantified by HPLC. Plant Genet Resour-C. 2008; 6:62-69. https://doi.org/10.1017/S1479262108923807
CrossRef - Song W., Yang R., Yang X., Sun S., Mentreddy S. R., Jiang B., Wu T., Tian S., Sapey E., Wu C., Hou W., Ren G., Han T. Spatial differences in soybean bioactive components across China and their influence by weather factors. Crop J. 2018; 6:659-668. https://doi.org/10.1016/j.cj.2018.05.001
CrossRef - Raffa D., Maggio B., Raimondi M. V., Plescia F., Daidone G. Recent discoveries of anticancer flavonoids. J. Med. Chem. 2017; 142:213-228. https://doi.org/10.1016/j.ejmech.2017.07.034
CrossRef - Ross J. A., Kasum, C. M. Dietary flavonoids: bioavailability, metabolic effects, and safety. Rev. Nutr. 2002; 22:19-34. https://doi.org/10.1146/annurev.nutr.22.111401.144957
CrossRef - Foti P., Erba D., Riso P., Spadafranca A., Criscuoli F., Testolin G. Comparison between daidzein and genistein antioxidant activity in primary and cancer lymphocytes. Arch Biochem Biophys. 2005; 433:421-427. https://doi.org/10.1016/j.abb.2004.10.008
CrossRef - Al Amri F.S., Hossain M.A. Comparison of total phenols, flavonoids and antioxidant potential of local and imported ripe bananas. J. Basic Appl. Sci. 2018; 5:245-251. https://doi.org/10.1016/j.ejbas.2018.09.002
CrossRef - Peyrat-Maillard M.N., Cuvelier M.E., Berset C. Antioxidant activity of phenolic compounds in 2,2’-azobis (2-amidinopropane) dihydrochloride (AAPH)-induced oxidation: synergistic and antagonistic effects. Am. Oil Chem.’ Soc. 2003; 80:1007-1012. https://doi.org/10.1007/s11746-003-0812-z
CrossRef - Iacopini P., Baldi M., Storchi P., Sebastiani L. Catechin, epicatechin, quercetin, rutin and resveratrol in red grape: content, in vitro antioxidant activity and interactions. Food Compost. Anal. 2008; 21:589-598. https://doi.org/10.1016/j.jfca.2008.03.011
CrossRef - Becker E. M., Ntouma G., Skibsted L. H. Synergism and antagonism between quercetin and other chain-breaking antioxidants in lipid systems of increasing structural organisation. Food Chem. 2007; 103:1288-1296. https://doi.org/10.1016/j.foodchem.2006.10.034
CrossRef - Yoshiara L. Y., Madeira T. B., Delaroza F., da Silva J. B., Ida E. I. Optimization of soy isoflavone extraction with different solvents using the simplex-centroid mixture design. J. Food Sci. Nutr. 2012; 63:978-986. https://doi.org/10.3109/09637486.2012.690026
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.







