Introduction
Many vegetables are categorized within the family Cruciferae. The most common cruciferous vegetables are watercress, Brussels sprouts, broccoli, cabbage, kai choi, kale, horseradish, radish and turnip. Recent scientific research has focused on health benefit effect of cruciferous vegetables due to their high content of beneficial substances, glucosinolates and their metabolites such as isothiocyanates (ITCs).1 About 120 naturally occurring glucosinolates have been identified so far but their type and content varies among vegetables; the amount being highest in Brussel sprouts > swede > savoy cabbage > broccoli.2 Isothiocyanates are a group of secondary metabolites which are naturally formed prior to hydrolysis of glucosinolates by the plant enzyme, myrosinase. One such important ITC is phenethyl isothiocyanate (PEITC), which has been shown to be a dominant phytochemical in cruciferous vegetables.3 It is an extensive studied ITC because of its high protective capability against a wide variety of cancers, such as lung,4,5 breast, oesophagus, forestomach, pancreas, prostate and colon as well as leukemia.6-9
Bioavailability of bioactive compounds from food following oral intake depends largely on their stability in human digestive system.10 Most of substances are changed due to enzymes and the acid-base in human gastrointestinal digestion. Study of the digestive fate of bioactive food compounds can help to improve performance and, therefore, promote health benefits. The in vitro digestion stimulates gastrointestinal digestion in human body and can be used to monitor any changes after oral intake. The result from in vitro digestion can be used to extrapolate in actual in vivo.
The consumption of cruciferous vegetables varies among populations. In Thailand and some Asian countries, Raphanus sativus (L.) var. caudatus Alef (Thai rat-tailed radish), Brassica juncea (L.) Czern. (leaf mustard) and Brassica juncea (L.) Coss. var. sareptana Sinskaja (mustard green) are majorly consumed. Being a member of cruciferous family, only a report has been made so far for Thai rat-tailed radish. The extract of which inhibited proliferation of HCT116 colon cancer cell line where cytotoxicity and cancer cell death induction via apoptosis are believed to be the mechanisms of action.11 However, scientific supporting data for health benefits potential of these vegetables especially after consumption is still inadequate. Therefore, the current research was aimed to determine bioactive compounds and antioxidant capacity of cruciferous vegetables. The effects of digestion system on these characteristics were also evaluated employing in vitro study.
Materials and Methods
Chemicals and reagents
Methanol, hexane, sodium acetate, acetic acid, hydrochloric acid, iron (III) sulfate and 2,4,6 tripyridyl-s-triazine (TPTZ) were purchased from RCI LABSCAN. Ferric chloride, sodium carbonate, pepsin, dipotassium phosphate, butyrated hydroxytoluene, sodium bicarbonate, benzene-1,2-dithiol, trolox, sodium chloride, potassium chloride, phenethyl isothiocyanate, 2,2-diphenyl 1 picrylhydrazyl and pancreatin were obtained from Sigma-Aldrich.
Samples and extract preparation
Mustard green, leaf mustard and Thai rat-tailed radish were purchased from market in Chiang Rai during August-September, 2016. The plants were washed well and subjected to extraction according to method described previously 12, with slight modification. The edible parts of vegetables were ground for 3 min, mastication time in normal oral consumption, to allow natural conversion of glucosinolates to isothiocyanates by myrosinase activity. The ground samples were then soaked in solvent (water, ethanol or hexane) at the ratio of plants to solvent 1:20 (wt:vol) prior to leaving the mixture in the dark at room temperature for 24 hours. The solvents were filtered by Whatman filter paper No.1 under vacuum condition. The filtrates were evaporated under vacuum at 50 oC to dryness and freeze-dried to obtain sample powder. The samples were stored at -20 oC prior to analysis.
Total phenolic content
Total phenolic content (TPC) determined by using Folin–Ciocalteu assay.13 Samples (30 µL) were added into 96-well plate prior to 120 µL of Folin–Ciocalteau’s reagent (10-time dilution) and 150 µL ml of sodium carbonate (7.5% w/v) were included. Samples were incubated for 30 min at room temperature and then the absorbance at 765 nm was recorded. The results were expressed as g Gallic acid equivalent (GAE)/ 100 g sample.
Phenethyl isothiocyanate content
Determination of phenethyl isocyanates (PEITC) content was carried out using UV-spectrophotometric method14. To a 96-well, 90 µL of K2PO4 (pH 8), 90 µL of methanol, 10 µL of the sample and 10 µL of benzene-1,2-dithiol (80 mM in methanol) were added and mixed well. Samples were then incubated at 60˚C for 90 minutes and cooled down to room temperature. Absorbance was measured at 365 nm and quantification was achieved by using a calibration curve of phenethyl isothiocyanate (PEITC).
DPPH radical-scavenging activity
The antioxidant activity was measured using a 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay.15 To 195 µL of 0.1M DPPH solution, 5 µL of dissolved samples were added. The absorbance at 515 nm was read after 30 min of incubation at room temperature in the dark. Trolox solutions (in 95% ethanol) ranged from 0–1000 µM were used to establish calibration curve. The antioxidant capacity of the samples was calculated and expressed as µM Trolox equivalent (TE)/100 g sample.
Ferric reducing antioxidant power (FRAP) assay
Ferric reducing antioxidant power (FRAP) assay uses antioxidants as reductants in a redox-linked colorimetric method. The antioxidant measured by using the Ferric reducing ability described previously.16 Sample powder was dissolved in water and then 10 µL of samples was added into 190 µL of FRAP solution in the 96-well microplate. The mixture was allowed to incubate at room temperature for 30 min. The absorbance was determined at 593 nm and ferric reducing ability of plant extract was expressed as µM FeSO4/100 g sample.
In vitro gastrointestinal digestion
In vitro gastrointestinal digestion was slightly modified according to the method reported before.17 Briefly, 1 mL of each extract was mixed with saline (140 mM NaCl and 5 mM KCl containing 150 μM BHT) to create a final volume of 4.5 mL. The mixtures were mixed well and acidified with 0.1 M/1 M HCl until it reached pH 2. Then, gastric digestion was performed with the addition of 125 μL of pepsin solution (40 mg/mL of 0.1 M HCl) and the mixtures were placed in a shaker at 37 °C for 1 hr. Thereafter, the pH of the solution was adjusted to 6.9 with the addition of 0.1 M/1 M NaHCO3. Further intestinal digestion was performed with the addition of 625 μL of pancreatin solution (2 mg/mL of 0.1 M NaHCO3) and incubated in a shaker at 37 °C for 2 hrs. The digesta volume is adjusted to 9 mL with saline and stored at −80 °C for further experiments.
Statistical analysis
Data were expressed as mean values ± standard deviation. The data were subjected to analysis using SPSS 17.0 for Windows. Analysis of variance (ANOVA) was conducted and Duncan’s multiple range test was used to identify mean separation.
Results and discussion
Total phenolic content (TPC) values before and after in vitro digestion
Total phenolic content (TPC) of vegetable extracts is shown in Table 1. Among three types of vegetable, mustard green showed the highest TPC values 19.78 ± 0.01 g GAE/100 g dry weight whereas leaf mustard had the lowest TPC. Previous studies reported, in a fresh weight basis, that TPC in different types of plant was diverse. Kale contained 773 ± 46.88 mg GAE /100 g of fresh weight which is close to TPC in mustard green in the current study (544.35 ± 0.24 mg GAE /100 g of fresh weight, data not shown) and significantly higher than broccoli and Brussels sprouts at 289 ± 6.12 and 331 ± 33.22 mg GAE /100 of fresh weight, respectively.18 In addition, the ranges of total polyphenol in mustard green and Thai rat-tailed radish were similar to that reported in green tea and black tea which varied from 21.02 ± 1.54 to 14.32 ± 0.45 and 17.62 ± 0.42 to 8.42 ± 0.55 g GAE/100 g, respectively.19
Phytochemicals in plant extracts are aimed to be utilized as supplements or nutraceuticals. However, extraction with water was employed in the current study to represent a common cooking method for vegetable consumption compared to solvent extract. Since most phenolic compounds are water soluble, it is not surprising that water was the most suitable solvent for TPC extraction from cruciferous vegetables. In plants, phenolics occur in soluble forms as well as in combination with cell wall components. To 100% total phenolics, only 20 and 32% bound phenols were accounted in broccoli and cabbage, respectively.20 It has been reported that water solubility of polyphenol depends on types of substances and their hydroxyl group. Those that are soluble in water are susceptible to water treatment. After boiling, phenolic compounds was reduced for they were leached out to the water unlike steaming where an increase in polyphenols and glucosinolates were observed compared with fresh broccoli.21 Moreover, plant leaf usually contain high amount of flavonols, the most ubiquitous flavonoids in foods which is water soluble polyphenol.22 These flavonols accumulate in the outer and aerial tissues (skin and leaves) because their biosynthesis is stimulated by light. This might explain the higher TPC content found in mustard green, a leafy vegetable, compared with other vegetables when extracted with water. Water extract of Thai rat-tailed radish also contained significant higher TPC than methanol and hexane extracts implying that the type of phenolic compounds in Thai rat-tailed radish are likely to be water soluble compounds. The lowest TPC content was noted in leaf mustard.
In vitro gastrointestinal digestion method was employed to simulate physiological conditions of the stomach and small intestine i.e. pH, temperature, and enzyme conditions.23 It was observed that TPC values decreased (27.9-41.6%) in all samples after in vitro digestion as shown in Figure 1A. Previous research showed that during gastric phase most of phenolics were not affected but marked reduction occurred during intestinal phase.24 Dietary polyphenols has been reported for their sensitivity to alkaline condition in intestine where structural changes lead to chemical property alteration.25 There are many types of phenolic compounds and each of which has different stability in acid-base conditions. With regards to phenolic acids, previous study showed that it decreased after the gastric digestion while flavonoids and non-anthocyanic polyphenols showed greater stability.24,26 As a strong antioxidant, ascorbic acid inhibited phenolic acids degradation during alkaline hydrolysis.27 This suggests that the co-consupmtion of these vegetables with vitamin C rich food can prevent TPC loss. Similar results were demonstrated in pomegranate juice, marc and peel extract where the decrease of TPC was observed only after the duodenal phase of in vitro digestion (p < 0.05).28 In human gastrointestinal tract, the other reason for reduction of TPC only in intestinal phase is probably due to most of the glycosides in TPC resist acid hydrolysis in the stomach whereas small intestines are rich in the activity of glycosidases which are responsible for hydrolysis of glycoside bonds and formation of aglycones.29 In addition to enzymatic degradation, the phenolics can also be hydrolyzed by the colonic microflora in intestinal.30
Recently, reduction of availability of total phenolic compounds in strawberry, raspberry, blackberry, and blueberry has been reported. Prior to in vitro digestion, their availability was ranged 33-73% and when antioxidant activity was concerned, 90% reduction was observed in all berries as measured by ORAC method.31 Moreover, it was reported that the effect of gastrointestinal digestion on total phenolic content of solid and liquid food was different. Phenolic contents and antioxidant activities of cereals, legumes, vegetables, tuberous vegetables, chocolates and fruits were greater than those obtained from aqueous extraction such as red wine, coffee and yerba mate after digestion. It was suggested that the phenolics that bound with solid and complex matrix are protected from digestion system such as enzymes and pH.32
Table 1: Total phenolic (TPC) and phenethyl isothiocyante (PEITC) content before and after in vitro digestion of vegetables extracts
|
Treatment |
Before digestion |
After digestion | |||
| TPC | PEITC | TPC |
PEITC |
||
| Vegetable | Solvent | g GAE/100g | µmol/100g | g GAE/100g | µmol/100g |
| Mustard green | Hexane | 14.12 ± 0.24c | 5.41 ± 0.62b | 13.33 ± 0.13c | 3.41 ± 0.16b |
| Methanol | 17.73 ± 0.24b | 5.56 ± 0.50b | 15.73 ± 0.72b | 3.22 ± 0.24c | |
| Water | 19.78 ± 0.01a | 9.65 ± 1.08a | 18.05 ± 0.41a | 4.21 ± 0.10a | |
| Leaf mustard | Hexane | 3.85 ± 0.21g | 3.00 ± 0.41cd | 2.14 ± 0.15i | 1.30 ± 0.07e |
| Methanol | 3.01 ± 0.07fg | 2.17 ± 0.16d | 2.93 ± 0.06h | 1.26 ± 0.17e | |
| Water | 4.85 ±0.78f | 3.06 ± 0.29c | 4.44 ± 0.13g | 1.60 ± 0.05d | |
| Thai rat-tailed radish | Hexane | 7.76 ± 0.26e | 3.01 ± 0.16cd | 5.92 ± 0.04f | 2.19 ± 0.09c |
| Methanol | 11.02 ± 1.88d | 3.27 ± 0.02c | 8.17 ± 0.08e | 2.16 ± 0.17c | |
| Water | 13.89 ± 0.09c | 3.78 ± 0.15cd | 11.83 ± 0.17d | 1.82 ± 0.19d | |
Values are expressed as mean ± standard deviation of three replicate measurements. a,b,c,d Values in the same column with different superscripts were significantly different at the level of p < 0.05. Where TPC = total phenolic content and PEITC = phenethyl isothiocyanate
Phenethyl isothiocyanate content before and after in vitro digestion
Phenethyl isothiocyanate (PEITC) content was evaluated since it is one of isothiocyanates that is well defined for chemopreventive action.33 PEITC content in all vegetables was highest when water was used as a solvent (Table 1). In addition, mustard green that extracted by water containned the highest PEITC concentration being 9.65±1.08 µmol/100 g. The higher PEITC in water extract than other solvents even though PEITC is a low water soluble compound34 is probably due to myrosinase enzyme activity that is crucial for conversion of glucosinolate to PEITC was decreased in organic solvents including ethanol, hexane or methanol.35 The solvents cause changes of enzyme conformation and reduce the enzyme dynamics.36 Moreover, PEITC values in this study were considerably higher than previous report studied in watercress where the values were 0.233-0.688 µmol/100 g sample.37 Different type of plant and also the variations in ITCs content within individual cruciferous vegetables might be attributed to environmental and genetic factors, which determined the content of glucosinolates and the ratio of ITCs producing glucosinolates to the other glucosinolates in cruciferous vegetables.12
In vitro digestion showed substantial effects on the content of PEITC in the samples. The amount of PEITC was reduced by half as shown in Figure 1D. The highest reduction observed was 56.7% and it is more likely that the effects were less pronounced in hexane and methanol extracts compared with water extract. Previous study showed that PEITC is unstable in aqueous media and rapidly degraded to phenethylamine at low pH.34 At acidic condition of stomach, PEITC in water extract can be degraded to phenethylamine. It is the electrophilic character of the carbon atom of isothiocyanate group that makes ITCs very susceptible to chemical reactions. In which case the isothiocyanate group (S=C=N) is hydrolyzed by enzymes prior to phenethylamine and S=C=O are formed under acidic condition. Moreover, this reactive moiety probably react with other nucleophilic compounds in plant extract. Furthermore, after they are absorbed across the intestinal membrane, one of many factors that limits bioavailbility of bioactive compounds is the metabolism by biotransformation enzymes. Even though PEITC is the potent anticancer agent that could modulate phase I and phase II detoxcification enzymes at very low concentration38, only those that pass the liver and reach the target site intact are accounted for bioactivity in the body.
DPPH and FRAP values before and after in vitro digestion
DPPH and FRAP values are illustrated in Table 2. In agreement with TPC values, DPPH and FRAP values were highest in mustard green extracted by water being 8.18±0.01µM FeSO4/100 g and 7.75±0.31 µM TE/ 100g, respectively. However, after underwent in vitro digestion, DPPH and FRAP values in all samples significantly decreased to 4.93 ± 0.04 g TE/100g (39.73%) and 2.15 ± 0.08 g FeSO4/100g (72.38%) and the % reductions are shown in Figures 1B and 1C. The most reduction was found in hexane extract of mustard green and water extract of Thai rat-tailed radish for DPPH (53.5%) and FRAP (88.0%), respectively. Substantial reduction of antioxidant capacity after digestion while only modest changes of TPC content was observed suggesting that phenolic compounds in these vegetables might not be only compounds that provide antioxidant activity in the vegetables and those that responsible for reduction of antioxidant activities are susceptible to digestion condition. It can be hypothesized that the extract contains non-phenolic substances such as vitamin C and B, amino acids and peptides, that were not analyzed in this study and they are involved in antioxidant activity.39 Manach and colleagues29 found that hydrophilic antioxidants such as vitamin C and vitamin B in Brassica vegetables made up 89% of the total antioxidant capacity.23 However, it is noteworthy that, study in orange juice, vitamin C content was not accessible after in vitro digestion.40 Furthermore, the antioxidant capacity of these vegetables were rather not associated with PEITC content. Table 3 shows that PEITC possessed very low FRAP and DPPH values. This is in agreement with Plumb and co-workers41 that glucosinolates and their derivatives had low antioxidant activity even though isothiocyanates can protect against cancer but the mechanism of action is likely not in relation to direct scavenging with free radicals.
Table 2: Antioxidant activity of vegetables extracted by different solvents before and after in vitro digestion
|
Treatment |
Before digestion |
After digestion | |||
| DPPH | FRAP | DPPH |
FRAP |
||
| Vegetable | Solvent | g TE/100g | g FeSO4/100g | g TE/100g | g FeSO4/100g |
| Mustard green | Hexane | 6.86± 0.18d | 3.96 ± 0.45c | 3.19 ± 0.31e | 2.02 ± 0.13a |
| Methanol | 7.70± 0.10b | 3.23 ± 0.22d | 4.66 ± 0.04b | 0.87 ± 0.02d | |
| Water | 8.18 ± 0.10a | 7.75 ± 0.31a | 4.93 ± 0.04a | 2.15 ± 0.08a | |
| Leaf mustard | Hexane | 4.30 ± 0.02g | 2.25 ± 0.05e | 2.16 ± 0.04h | 1.13 ± 0.10bc |
| Methanol | 4.23 ± 0.02g | 3.12 ± 0.18d | 2.40 ± 0.13g | 1.25 ± 0.05b | |
| Water | 6.41 ± 0.08e | 3.09 ± 0.17d | 4.45 ± 0.15bc | 0.37 ± 0.04f | |
| Thai rat-tailed radish | Hexane | 4.57 ± 0.19f | 3.06 ± 0.27d | 2.81 ± 0.14f | 1.01 ± 0.07c |
| Methanol | 6.47 ± 0.20e | 3.00 ± 0.15d | 3.48 ± 0.07d | 1.23 ± 0.09b | |
| Water | 7.44 ± 0.06c | 4.38 ± 0.17b | 4.39 ± 0.17c | 0.68 ± 0.08e | |
Values are expressed as mean ± standard deviation of three replicate measurements. a,b,c,d Values in the same column with different superscripts were significantly different at the level of p < 0.05. Where DPPH = 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay and FRAP = Ferric reducing antioxidant power assay
Table 3: Antioxidant activity of PEITC at different concentrations
|
PEITC (µg/mL) |
DPPH |
FRAP |
| g TE/100g | g FeSO4/100g | |
| 0 | 0.23 ± 0.03b | 0.009 ± 0.01ns |
| 100 | 0.24 ± 0.01ab | 0.014 ± 0.01 ns |
| 200 | 0.24 ± 0.02ab | 0.010 ± 0.01 ns |
| 300 | 0.25 ± 0.01ab | 0.010 ± 0.01 ns |
| 400 | 0.25 ± 0.01ab | 0.011 ± 0.01 ns |
| 500 | 0.26 ± 0.01a | 0.009 ± 0.01 ns |
Values are expressed as mean ± standard deviation of three replicate measurements.
a,b Values in the same column with different superscripts were significantly different at the level of p < 0.05.
ns Values in the same column were not significantly different.
Where DPPH = 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay and FRAP = Ferric reducing antioxidant power assay.
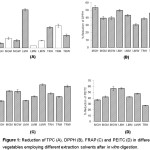 |
Figure 1: Reduction of TPC (A), DPPH (B), FRAP (C) and PEITC (D) in different vegetables employing different extraction solvents after in vitro digestion. Click here to View figure |
Values are mean ± standard deviation of triplicate analysis.
Diagonal striped bars were significantly different between before and after in vitro digestion by the LSD test at the level of p<0.05.
Note: MGH=mustard green extracted by hexane, MGM=mustard green extracted by methanol, MGW=mustard green extracted by water, LMH=leaf mustard extracted by hexane, LMM=leaf mustard extracted by methanol, LMW=leaf mustard extracted by water, TRH=Thai rat-tailed radish extracted by hexane, TRM= Thai rat-tailed radish extracted by methanol and TRW= Thai rat-tailed radish extracted by water
Conclusion
The results of this study revealed that cruciferous vegetables that are consumed in Thailand and many Asian countries can be good sources of human health protection. Among various varieties, mustard green had the highest TPC and antioxidant capacity. Water extraction could liberate bioactive compounds that afford extensive health benefit effects of the vegetable. However, when chemopreventive compound in term of PEITC was considered, mustard green was the promising cancer prevention source available in daily meal. In vitro digestion demonstrated significant reduction of antioxidants and their activities. Therefore, prevention of degradation during human digestion should be further studied.
Acknowledgements
The authors would like to thank Mae Fah Luang University for a support of the services and facilities including the research grant.
References
- Verhoeven D.T, Verhagen H, Goldbohm R.A, Van den Brandt P.A, van Poppel G. A review of mechanisms underlying anticarcinogenicity by brassica vegetables. Chemico-Biological Interactions; 103: 79-129: (1997).
CrossRef - Steinbrecher A, Nimptsch K, Husing A, Rohrmann S, Linseisen J. Dietary glucosinolate intake and risk of prostate cancer in the EPIC-Heidelberg cohort study. International Journal of Cancer; 125: 2179-2186: (2009).
CrossRef - Canistro D, Croce C.D, Iori R, Barillari J, Bronzetti G, Poi G, Cini M, Caltavuturo L, Perocco P, Paolini M. Genetic and metabolic effects of gluconasturtiin, a glucosinolate derived from cruciferae. Mutatation Research; 545: 23-35: (2004).
CrossRef - Hecht S.S. Chemoprevention by isothiocyanates. Journal of Cellular Biochemistry; 22: 195-209: (1995).
CrossRef - Mi L, Wang X, Govind S, Hood B.L, Veenstra T.D, Conrads T.P, Saha D.T, Goldman R, Chung F.L. The role of protein binding in induction of apoptosis by phenethyl isothiocyanate and sulforaphane in human non-small lung cancer cells. Cancer Research; 67: 6409-6416: (2007).
CrossRef - Hu R, Kim B.R, Chen C, Hebbar V, Kong A.N. The roles of JNK and apoptotic signaling pathways in PEITC-mediated responses in human HT-29 colon adenocarcinoma cells. Carcinogenesis; 24: 1361-1367: (2003).
CrossRef - Johnson C.R, Chun J, Bittman R, Jarvis W.D. Intrinsic cytotoxicity and chemomodulatory actions of novel phenethyl isothiocyanate sphingoid base derivatives in HL-60 human promyelocytic leukemia cells. Journal of Pharmacology and Experimental Therapeutics; 309: 452-461: (2004).
CrossRef - Ogawa K, Futakuchi M, Hirose M, Boonyaphiphat P, Mizoguchi Y, Miki T, Shirai T. Stage and organ dependent effects of 1-O-hexyl-2,3,5-trimethylhydroquinone, ascorbic acid derivatives, n-heptadecane-8-10-dione and phenylethyl isothiocyanate in a rat multiorgan carcinogenesis model. International Journal of Cancer; 76: 851-856: (1998).
CrossRef - Telang U, Brazeau D.A, Morris M.E. Comparison of the effects of phenethyl isothiocyanate and sulforaphane on gene expression in breast cancer and normal mammary epithelial cells. Experimental Biology and Medicine (Maywood); 234: 287-295: (2009).
CrossRef - Rein M. J, Renouf M, Cruz‐Hernandez C, Actis‐Goretta L, Thakkar S.K, da Silva Pinto M. Bioavailability of bioactive food compounds: a challenging journey to bioefficacy. British Journal of Clinical Pharmacology; 75(3): 588–602: (2013).
CrossRef - Pocasap P, Weerapreeyakulb N, Barusrux S. Cancer preventive effect of Thai rat-tailed radish (Raphanus sativus L. var. caudatus Alef). Journal of Functional Foods; 5(3):1372–1381: (2013).
CrossRef - Tang L, Paonessa J.D, Zhang Y, Ambrosone C.B, McCann S.E. Total isothiocyanate yield from raw cruciferous vegetables commonly consumed in the United States. J ournal of Functional Foods; 5(4): 1996-2001: (2014).
CrossRef - Deetae P, Parichanon P, Trakunleewatthana P, Chanseetis C, Lertsiri S. Antioxidant and anti-glycation properties of Thai herbal teas in comparison with conventional teas. Food Chemistry; 133(3): 953–959: (2012).
CrossRef - Lola-luz H, Hennequart F, Gaffney M. Effect on yield, total phenolic, total flavonoid and total isothiocyanate content of two broccoli cultivars (Brassica oleraceae var italica) following the application of a commercial brown seaweed extract (Ascophyllum nodosum). Agricultural and Food Science; 23: 28-37: (2014).
- Wang J, Lu X, Al-Qadiri H.M, Ross C.F, Powers J.R, Tang J, Rasco B. Determination of total phenolic content and antioxidant capacity of onion (Allium cepa) and shallot (Allium oschaninii) using infrared spectroscopy. Food Chemistry; 29(2): 637-644: (2011).
CrossRef - Porter V. Antioxidant properties of green broccoli and purple-sprouting broccoli under different cooking conditions. Bioscience Horizons; 5: hzs004: (2012).
- Benziea I, Strainb J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Analytical Biochemistry; 239(1): 70–76: (1996).
CrossRef - Sikora E, Cieslik E, Leszczynska T, Filipiak-Florkiewicz A, Pisulewski P.M. The antioxidant activity of selected cruciferous vegetables subjected to aquathermal processing. Food Chemistry; 107(1): 55-59: (2008).
CrossRef - Anesini C, Ferraro E.G, Filip R. Total polyphenol content and antioxidant capacity of commercially available tea (Camellia sinensis) in Argentina. Journal of Agriculture and Food Chemistry; 56 (19): 9225-229: (2008).
CrossRef - Chu Y.F, Sun J, Wu X, Liu R.H. Antioxidant and antiproliferative activities of common vegetables. Journal of Agriculture and Food Chemistry; 50: 6910-6916: (2002).
CrossRef - Gliszczyńska-Swigło A, Ciska E, Pawlak-Lemanska K, Chmielewski J, Borkowski T, Tyrakowska B. Changes in the content of health-promoting compounds and antioxidant activity of broccoli after domestic processing. Food Additives & Contaminants; 23: 1088-1098: (2006).
CrossRef - Mithen R, Faulkner K, Magrath R, Rose P, Williamso G, Marquez J. Development of isothiocyanateenriched broccoli, and its enhanced ability to induce phase 2 detoxification enzymes in mammalian cells. Theoretical Applied Genetics; 106(4): 727–734: (2003).
CrossRef - Vallejo F, Gil-izquierdo A, Pearez-vicente A, Garci AA-viguera C. In vitro gastrointestinal digesttion study of broccoli inflorescence phenolic compounds. Journal of Agriculture and Food Chemistry; 52(1):135-138: (2004).
CrossRef - Jafri L, Saleem S, Ihsan-ul-Haq, Ullah N, Mirza B. In vitro assessment of antioxidant potential and determination of polyphenolic compounds of Hedera nepalensis K. Koch. Arabian Journal of Chemistry; Article in press: (2014).
- Céspedes L.C, Sampietro A.D, Seigler S.D, Rai M. Natural antioxidants and biocides from wild medicinal plants. London:CAB International: (2013).
- Soriano Sancho R.A, Pavan V, Pastore G. M. Effect of in vitro digestion on bioactive compounds and antioxidant activity of common bean seed coats. Food Research International; 76(1): 74-78: (2015).
CrossRef - Nardini M, Cirillo E, Natella F, Mencarelli,D, Comisso A, Scaccini C. Detection of bound phenolic acids: prevention by ascorbic acid and ethylenediaminetetraacetic acid of degradation of phenolic acids during alkaline hydrolysis. Food Chemistry; 79(1): 119–124: (2002).
CrossRef - Fawole O. A, Opara U.L, Stability of total phenolic concentration and antioxidant capacity of extracts from pomegranate co-products subjected to in vitro digestion.BMC Complementary and Alternative Medicine; 16(1):358: (2016).
CrossRef - Gumienna M, Lasik M, Czarnecki Z. Bioconversion of grape and chokeberry wine polyphenols during simulated gastrointestinal in vitro digestion. Journal of Food Sciences and Nutrition; 62(3): 226-233: (2011).
CrossRef - Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L. Polyphenols: food sources and bioavailability. The American journal of clinical nutrition; 79(5): 727-747: (2004).
- Marhuenda J, Alemán M. D, Gironés-Vilaplana A, Pérez A, Caravaca G, Figueroa F, Mulero J, Zafrilla P. Phenolic composition, antioxidant activity, and in vitro availability of four different berries. Journal of Chemistry; 2016: 7 pages: (2016), Article ID 5194901.
- Koehnlein E. A, Koehnlein É. M, Corrêa R. C.G, Nishida V. S, Correa V. G, Bracht A, Peralta R.M. Analysis of a whole diet in terms of phenolic content and antioxidant capacity: effects of a simulated gastrointestinal digestion; 67(6):614-23: (2016).
- Ioannides C, Konsue N. A principal mechanism for the cancer chemopreventive activity of phenethyl isothiocyanate is modulation of carcinogen metabolism. Drug Metabolism Reviews; 47(3): 356-373: (2015).
CrossRef - Negrusz A, Moore C.M, McDonagh N.S, Woods E.F, Crowell J.A., Levine B.S. Determination of phenethylamine, a phenethyl isothiocyanate marker, in dog plasma using solid-phase extraction and gas chromatography-mass spectrometry with chemical ionization. Journal of Chromatography B: Biomedical Sciences and Applications; 718: 193-198: (1998).
CrossRef - Muller H. Determination of the carotenoid content in selected vegetables and fruit by HPLC and photodiode array detection. Zeitschrift fur Lebensmittel- Untersuchung und—Forschung A; 204: 88–94: (1997).
- Klibanov A.M. Improving enzymes by using them in organic solvents. Nature; 409(6817): 241- 246: (2001).
CrossRef - Mcavoy R.J, Palanuswamy U.R, Bible B.B, Stuart J.D. Ontogenic variations of ascorbic acid and phenethyl isothiocyanate concentrations in watercress (Nasturtium officinale R.Br.) leaves. Journal of Agricutural and Food Chemistry; 51(18): 5504−5509 (2003)ใ
CrossRef - Konsue N, Ioannides C. Tissue differences in the modulation of rat cytochromes P450 and phase II conjugation systems by dietary doses of phenethyl isothiocyanate. Food and Chemical Toxicology; 46: 3677-3683: (2008).
CrossRef - Castro R.J, Sato H.H. Antioxidant activities and functional properties of soy protein isolate hydrolysates obtained using microbial proteases. International Journal of Food Science & Technology; 49: 317–328: (2014).
CrossRef - Aschoff J.K, Kaufmann S, Kalkan O, Neidhart S, Carle R, Schweiggert R.M. In Vitro bioaccessibility of carotenoids, flavonoids, and vitamin C from differently processed oranges and orange juices [Citrus sinensis (L.) Osbeck]. Journal of Agricultural and Food Chemistry; 63(2): 578-587: (2015).
CrossRef - 41. Plumb G.W, Lambert N, Chambers S.J, Wanigatunga S, Heaney R.K, Plumb J.A, Aruoma O.I, Halliwell B, Miller N.J, Williamson G. Are whole extracts and purified glucosinolates from cruciferous vegetables antioxidants? Free Radical Research.; 25(1): 75-86: (1996).
CrossRef.

This work is licensed under a Creative Commons Attribution 4.0 International License.





