Introduction
Vegetables are an important factor of human nutrition. Following harvesting they undergo a series of physiological changes that affect the quality. These changes can not be stopped but can be controlled within certain limits by using the appropriate post harvest handling. Temperature control is one of the major treatments for increasing the time of their commercial life and maintain their quality. Cantwell et al (1998), reports that temperature is the only factor that determines the post-harvest quality of leafy green vegetables. It should be noted that the higher the storage temperature the faster the deterioration and the shorter the duration of the commercial life and this is because temperature affects the metabolism and respiration rate. The low temperature (in tolerance limits) and high relative humidity increase the commercial life of most fresh vegetables (Cantwell and Kasmire, 2002), delaying degradation of chlorophyll (Pogson and Morris, 1997). The degradation of chlorophyll is considered as the first visible symptom of aging, but when it appears in fact aging is already advanced (Ansaril and Chen, 2011). Degradation of chlorophyll is a complex multi-pathway as the biosynthesis of which may be summed in two basic group of reactions; the first one produce greenish derivatives while the second one, colourless compounds by an oxidative ring opening (Marquez and Sinnecker, 2007a). The degradation of chlorophyll reduces the intensity of the green color and leads to yellowing. Yellowing of leafy vegetables and broccoli is attributed to the action of the enzymes peroxidase and lipoxygenase (Murcia et al.2000). The reduction of the intensity of green vegetables is associated with aging, reduction of the nutritional value and in general their quality (Shewfelt, 2000; Cantwell and Kasmire, 2002).
The initial stage of the degradation of chlorophyll is influenced by external factors such as: the water stress, changes in temperature, light, ethylene, inadequate lighting or o combination (Heaton and Marangoni, 1996; Nooden, 1988a, 1988b; Okada et al. 1992; Taylor and Craig 1971, thomson, 1988; Aguero et al, 2011).
Consumers choose fruits and vegetables based on appearance (Abbott, 1999). An important parameter of the display is color which is affected by chemical, biochemical, microbiological and physical changes that occur during development, maturation and post harvest handling (Pankaj et al., 2011). Color is used as the maturity or aging criterion and as an indicator of physiological, mechanical or pathological lesions (Kader, 2002).
Green vegetables such as lettuce and broccoli are widely used in the Mediterranean diet. Over the last decade it has been scientifically proved and promoted that these vegetables constitute important functional food components by contributing vitamins, minerals and biologically active compounds which are associated with dietary activities (Kimura & Rodriguez-Amaya, 2003).
Leafy vegetables like lettuce are highly perishable, characterised by a short life-time and high transpiration rates due to high ratio of surface to mass, fact that contributes to accelerating senescence and colour loss. (Marquez & Sinnecker, 2007b).
The parameters for maintaining visual and nutritional quality in broccoli heads after harvest are well understood, with low temperature maintenance being of paramount importance. Recently, much attention has been focussed on the phytochemicals contained within broccoli, glucosinolates in particular, that may help prevent the onset of certain cancers and cardiovascular disease (Jones et al 2006).
The aim of this work is the study of the changes in the color and especially chlorophyll in two green vegetables widely consumed and of high commercial value, lettuce and broccoli, when stored at temperatures of 0°C, 5°C, 10°C and 20°C. According to Watada et al. (1996), the recommended storage temperature is 0°C, however vegetables are often stored at 5°C and occasionally at 10°C. The temperature of 20 ° C is the room temperature.
Materials and Methods
Sample preparation
Lettuce type Romana (Lactuca sativa, var. Paris Island), were cut at the stage of commercial maturity, (approximately 20 to 24 leaves / head) early in the morning and immediately transferred to postharvest laboratory where sorting was carried out to reduce the natural variability and remove the outer leaves.
Broccoli (Brassica oleracea, var. Marathon) were harvested when the warheads were fully developed without the florets being deployed. Immediately after cutting which was carried out early morning they were transported to the laboratory where they were screened for size and color.
Both lettuce and broccoli were grown on the farm of TEI Peloponnse in Kalamata. Following sorting they were divided into 4 groups, each of which contained 36 pieces.
The vegetables were stored in the dark to prevent potential interference of photosynthesis on chlorophyll breakdown at four different temperatures: 0, 5, 10 and 20 ° C and 90% relative humidity. At 0 , 5 and 10 ° C the lettuce storage period was 13 days whereas that of broccoli 23 and at 20 ° C the lettuce storage time was 10 days whereas that of broccoli 12 days.
The parameters studied were: the change in the total chlorophyll content and the change of color. Measurements were made on the 0, 3, 6, 10th, and 13th day in the case of lettuce and 0, 6th, 9th, 12th, 16th, 19th and 23rd in the case of broccoli.
Measurements were made at six different samples per temperature for both vegetables.
Total chlorophyll determination
The chlorophyll was extracted with the solvent dimethylsulfoxide (DMSO) in accordance with the method of (Hiscox & Isrelstam, 1979, Barnes et al, 1992). 0.1g of tissue was placed into a test tube containing 10 ml DMSO. The tube loosely capped was placed in a water bath at 60 ° C until complete depigmentation (1h). Cooling was followed at room temperature for 30 min, followed by filtration and measurement of the absorption at wavelengths 665 nm and 648 nm. Calibration of the instrument (blank) was carried out with DMSO. The concentration of total chlorophyll was expressed in mg / g fresh weight of the sample and was calculated from the formula
Chl total= 7,49 A665 + 20,34 A 648 (Hiscox & Isrelstam, 1979).
Colour Determination
The color changes of lettuces and broccoli were determined by the colorimeter Minolta (Model CR-300, Minolta Co Ltd Osaka). Before each measurement the instrument was calibrated by a white calibration plate (Y = 92.6, x = 0.3135 and y = 0,3193). The color was determined by the parameters L *, a *, b *. L * parameter gives brightness and changes between 0 (black) to 100 (white). The color parameter a * gives the green (-a *) or the red (+ a *), and the color parameter b * gives the yellow (+ b *) or blue (-b *) (Mc Guire, 1992). From the parameters L *, a * and b * the hue h ° [h ° = tan -1 (b / a)] was calculated (Lancaster et al, 1997) and the color difference E =. [(L-Lo) 2 + (a-ao) 2 + (b- bo) 2)] 1/2. Lo, ao, bo are the values at baseline. The ΔE * is important when the relationship between the visual and the numerical analyses is evaluated while the h ° are the qualitative attributes of color (Sant’Anna et al., 2013).
At heads of broccoli color was determined at 5 points (4 in the region and one in the center). In lettuce color was determined at three points of the plate (base, medium, end) in 3 leaves located immediately after the outer sheets. The change of color of both vegetables was attributed by the parameters: L *, ho and ΔE*.
Statistical analysis
Experimental data was analyzed with the statistical package Statgraphics Plus (5.1). Comparison of the means was carried out with the Fisher method of least significant differences (LSD), at a confidence level p= 0.05.
Results And Discussion
Changes in chlorophyll
Change in total chlorophyll of lettuce (α) and broccoli (b) is shown in Fig. 1.
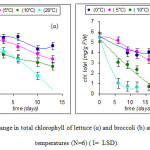 |
Figure 1: Change in total chlorophyll of lettuce (α) and broccoli (b) at 4 different temperatures (Ν=6) ( Ι= LSD).
|
Statistical analysis (ANOVA) showed that the factors affecting the degradation of chlorophyll in lettuce and broccoli are: storage temperature, storage life and their interaction (p = 0.05).
From Figure 1 it is shown that at the temperatures of 10 and 20°C a sharp degradation of chlorophyll occurs both in lettuce and broccoli. On the third day of storage at these temperatures (Figure 1a) lettuce has lost approximately 20% of the initial chlorophyll, and at the end of storage (day 13) the percentage of the loss amounted to 46% at 10°C and 64% at 20°C. In the case of broccoli (Fig.1 b) after 6 days of storage at 10 and 20°C a loss of about 44% and 80%, respectively was shown, while at the end of storage (23 days at 10° C and 12-days at 20 ° C) these rates amounted to 93 and 88%, respectively. Our results agree to those reported by (Lemoine et al 2007); Page et al 2001; Costa et al. 2006).
At 5 ° C storage chlorophyll content was constant for the first 3 days in lettuce and 6 days in broccoli. Page et al (2001) reported that broccoli presented a significant reduction in chlorophyll after 11 days at 4 ° C, while Zhuang et al (1994) reported that broccoli showed a significant reduction in chlorophyll (~ 40%) on the 6th day at 5 ° C. At the end of storage lettuce showed a loss of 30% which is consistent with the findings of Singh et al (1972), while the broccoli showed a loss of 73%. At temperatures of 5, 10 and 20 ° C the change of chlorophyll in lettuce and broccoli is practically a linear function of storage time with R2 ranging between 0.83 and 0.99 in both cases.
At the end of storage at 0°C lettuce (day 13) showed a loss of about 5%, and broccoli a loss of approx. 25%. The temperature of 0°C reduces the respiratory activity, the rate of metabolism and the degradation of chlorophyll (Pogson and Morris, 1997). The degradation of chlorophyll is a symptom of transition of chloroplasts to gerontoplasts, score characteristic of plastids of aging (Sitte et al., 1980). Degradation of chlorophyll brings to the surface colors that preexisted, in this case carotenoids (Gross, 1991).
Colour Change
Change in colour is attributed to L*, h°, ΔE* parameters, (Table 1)
Table 1: Change in color parameters L*, h°, ΔE* of lettuce and broccoli stored at 0, 5, 10 and 20°C.
|
Days |
L* |
h° |
ΔE* |
|||||||||
|
0°C |
5°C |
10°C |
20°C |
0°C |
5°C |
10°C |
20°C |
0°C |
5°C |
10°C |
20°C |
|
|
|
Lettuce |
|||||||||||
|
0 |
42.0 |
42.0 |
42.0 |
42.0 |
121.4 |
121.4 |
121.4 |
121.4 |
0 |
0 |
0 |
0 |
|
3 |
39.7a |
42.7b |
44.4bc |
45.5c |
120.7a |
120.9a |
120.8a |
117.9a |
5.0a |
4.8a |
4.3a |
5.6a |
|
6 |
42.7a |
43.4a |
43.4a |
56.8b |
122.2a |
120.9a |
119.7a |
114.6b |
6.2a |
6.3a |
8.6b |
9.6c |
|
10 |
37.8a |
42.5b |
50.8c |
57.0d |
120.4a |
118.9a |
115.8b |
109.6c |
6.3a |
7.2a |
9.0b |
16.8c |
|
13 |
39.8a |
45.4b |
47.4b |
120.0a |
114.7b |
107.8c |
6.3a |
9.5b |
11.7c |
|||
|
Broccoli |
||||||||||||
|
0 |
40.0 |
40 |
40 |
40 |
117.7 |
117.7 |
117.7 |
117.7 |
0 |
0 |
0 |
0 |
|
6 |
39.3a |
38.4a |
42.5b |
45.3c |
118.1a |
118.0a |
114.8a |
87.8b |
5.1a |
4.7a |
6.5b |
12.4c |
|
9 |
40.0a |
40.1a |
45.1b |
47.7c |
119.1a |
116.7a |
107.7b |
86.1c |
6.1a |
6.3a |
10.3b |
12.4c |
|
12 |
39.0a |
40a |
48.3b |
52c |
117.3a |
116a |
100.4b |
83.9c |
5.0a |
6.1a |
15.0b |
16.2b |
|
16 |
40.2a |
43.6b |
48.9c |
117.6a |
111.5b |
96.5c |
6.4a |
10.3b |
15.7c |
|||
|
19 |
37.8a |
44.6b |
47.9c |
118.5a |
105.3b |
99.2c |
6.4a |
12.3b |
16.6c |
|||
|
23 |
38.9a |
44.9b |
118.8a |
102.3b |
6.0a |
14.1b |
||||||
Means, lettuce= 6 samples ´ 3leaves ´ 3 measurements/leaf=54
Μeans, broccoli= 6 samples ´ 5 measurements / sample = 30
Values within small letters show statistically significant changes from other values within the same row.
Statistical analysis (ANOVA) showed that the change of color parameters L *, h ° and ΔE* in both products is affected significantly (p = 0.05) by temperature, storage time and their interaction.
The temperature of 0°C maintained brightness close to the baseline levels at both cases. Therefore, at the end of storage (day 13) lettuce lost 5% of its brightness, whereas the broccoli lost 3% at day 23. The higher the storage temperature the greater the change in brightness. Hence, at the end of storage (13th day) lettuce stored at 5°C showed an increase of about 8%, at 10°C, the increase was 13%, in particular 20 ° C, the increase recorded was of the order of 36 %.
In broccoli the increase in brightness observed at the end of storage at 5°C was 12%, at 10 ° C was 19% and at 20 ° C was 30%. In both cases the change in brightness (L *) at temperatures of 5, 10 and 20 ° C was a linear function of storage time with R2 ranging between 0.71 and 0.98.
The parameter h° (hue) at end of storage at 0°C was maintained at baseline levels in both vegetables. At temperatures of 5, 10 and 20°C in both vegetables a reduction was observed which was stronger as the storage temperature was higher. Thus at temperatures 5, 10 and 20°C the reduction of the parameter values h ° for lettuce was 5, 11 and 10% respectively, while for broccoli was 13, 16 and 28%.
An increase in the total color change ΔE* was observed from the first day of the storage. At 0°C the variation was maintained at low levels while at higher temperatures the variation was more pronounced.
At a temperature of 0° C a slight decrease in brightness (L *) was observed along with maintenance of the hue values (h°) close to baseline levels for both vegetables. The slight decrease in L* values indicates that the color became darker during storage, while the maintainenance the h° values shows evidence that there was no reduction in the intensity of green color. Our results agree with those of Chandra et al (2008) in lettuce and Gnanasekharan et al (1992) in broccoli. At higher storage temperatures an increase in the L* values was observed along with a reduction in the h° values. The increase in L * values is a sign of reduction of the intensity of green color due to appearance of yellow pigments, while low values of h° is an indication of degreening associated with aging. These results agree with those of Chandra et al (2008), Suthumchai et al. (2007) in lettuce and Lemoine et al (2008) in broccoli.
Broccoli stored at 20°C showed values of h ° less than 90 ° due to the color parameter a * taking positive values. This result agrees with Gnanasekharan et al (1992).
It should be noted that at day 3 in lettuce and day 6 in broccoli samples stored at 10 and 20 ° C there was shown a significant reduction in chlorophyll, whereas the change in h ° was not analogous, indicating that the degradation of chlorophyll starts before the color change is perceived. Measurement of chlorophyll is a more sensitive parameter for studying the aging of plant tissues. This result agrees with those of Costa et al (2006) and Finger et al (1999) in broccoli.
Comparison between lettuce and broccoli
In order to compare the degradation of chlorophyll in a leafy vegetable such as lettuce and one inflorescence like broccoli the average daily rate of chlorophyll was calculated along with the corresponding rate of total color change with temperature.
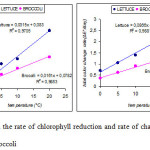 |
Figure 2: Change in the rate of chlorophyll reduction and rate of change in total colour for lettuce and broccoli Click here to View figure |
From Figure 2 it is evident that the lettuce shows a greater rate of chlorophyll reduction and change in total color. It should be noted that the color change patterns vary depending on the type of plant organ probably because of differences in the surface with the object interaction of incident light (Gnanasekharan et al 1992).
Conclusions
From the study results show that:
- In terms of colour criterion the lettuce can be stored in marketable condition at 0 ° C for 13 days, at 5 ° C for 10 days, at 10 ° C for 6 days and at 20 ° C for 3 days, a storage which agrees with literature data (Suthumchai et al. (2007), Chandra et al (2008).
- Broccoli if not stored at low temperature presents aging symptoms and degradation of chlorophyll. Therefore, at 0°C it can be stored at marketable condition for 23 days, at 5 ° C for 12 days, at 10 ° C for 6 days and at 20 ° C for 2 days, which is consistent with the observations of Cantwell and Kasmire (2002) and Finger et al (1999).
References
- Abbott J.A., 1999. Quality measurement of fruits and vegetables. Postharvest Biology and Technology, 15:207-225.
CrossRef - Agüero, M.V., Ponce, A.G., Moreira, M.R. & Roura S.I., 2011. Lettuce quality loss under condition that favours the wilting phenomenon. Postharvest Biology and Technology, 59: 124-131.
CrossRef - Ansaril, M.I. & Chen, S.G., 2011. Leaf senescence. An overview. International Journal of Recent Trends in Science and Technology, 1(3): 110-114.
- Barnes, J.D., Balaguer, L., Manrique, E., Elvira, E. and Davison, A.W., 1992. A reappraisal of the use of DMSO for the extraction and determination of chlorophylls a and b in lichens and higher plants. Environmental and Experimental Botany, 32 (2):85-95.
CrossRef - Cantwell, .M and Kasmire, F.R., 2002. Postharvest handling systems: flower, leafy and stem vegetables. In: Postharvest Technology of Horticultural crops. (ed. A. Kader), University of California Agr and Natural Resources Pub.3311,pp.423-433.
- Cantwell, M., J. Rovelo, X. Nie and V. Rubatzky, 1998. Specialty salad greens:Postharvest physiology and shelf-life. Acta Horticulturae, 467:371-377.
CrossRef - Chandra, D., Matsui, T., Suzuki, H. and Kosugi, Y., 2008. Changes in some physio-biochemical characteristics in lettuce during storage at low temperature. Journal of Biological Sciences, 8: 398-403.
CrossRef - Costa, L.M., Civello, M.P., Chaves, A.R. and Martínez, G.A., 2006. Hot air treatment decrease chlorophyll catabolism during postharvest senescence of broccoli ((Brassica oleracea L. var. Italica) heads. Journal of the Science of Food and Agriculture 86:1125-131.
CrossRef - Finger, F.L., Endres, L., Mosquim, P.R. and Puiatti, M., 1999. Physiological changes during postharvest senescence of broccoli. Pesquisa Agropecuaria Brasileira, Brasilia, 34(9) :1565-1569.
CrossRef - Gnanasekharan, V., Shewfelt, R.L. and Chinnan, M.S., 1992. Detection of color changes in green vegetables. Journal of Food Science, 57 (1):149-154.
CrossRef - Gross, J., 1991. Pigments in vegetables-Chlorophylls and carotenoids. Van Nostrand Reinhold New-York.
CrossRef - Heaton, W.J and Marangoni, G.A., 1996. Chlorophyll degradation in processed foods and senescent plant tissues. Trends in Food Science and Technology, 7: 8-15.
CrossRef - Hiscox, J.D and Israelstam, G.F., 1979. A method for the extraction of chlorophyll from leaf tissue without maceration. Canadian Journal of Botany, 57:1332-1334.
CrossRef - Jones ,B.R., Faragher,D.J., Winkler,S.,2006. A review of the influence of postharvest treatments on quality and glucosinolate content in broccoli (Brassica oleracea var. italica) heads. Postharvest Biology and Technology 41: 1–8.
CrossRef - Kader,A., 2002. Maturation and maturity index. Postharvest Technology of Horticultural crops. (ed. A. Kader), University of California Agr and Natural Resources Publication, 3311 pp. 55-62.
- Kimura, M. & Rodriguez-Amaya, D.B., 2003. Carotenoid composition of hydroponic leafy vegetables. Journal of Agricultural and Food Chemistry, 51, 2603-2607.
CrossRef - Lancaster, J.E., C.E. Lister, P.F. Reay and C.M. Triggs, 1997. Influence of pigment composition on skin color in a wide range of fruit and vegetables. Journal of the American Society for Horticultural Science, 122: 594-598.
- Lemoine, M., Civello, P.M., Chaves, R.A., Martínez, G.A., 2008. Effect of combined treatment with hot air and UV-C on senescence and quality parameters of minimally processed broccoli (Brassica oleracea L. var. Italica). Postharvest Biology and Technology, 48(1): 15-21.
CrossRef - Marquez, U.M.L. & Sinnecker, P., 2007a. Chlorophylls: Properties, biosynthesis, degradation and functions. In: Food Colorants Chemical and Functional Properties (edited by C. Socaciu). Pp. 25-49. Florida, USA: CRC Press, Taylor and Francis Group.
CrossRef - Marquez, U.M.L. & Sinnecker, P., 2007b. Chlorophylls in foods: Sources and stability. In: Food Colorants Chemical and Functional Properties (edited by C. Socaciu). Pp. 199-204. Florida, USA: CRC Press, Taylor and Francis Group.
- McGuire, R.G., 1992. Reporting of objective colour measurements. HortScience, 27: 1254-1255.
- Murcia, M.A., LóPEZ-Ayerra, B., Martinez-Tomé, M. and Garcia-Carmona, F., 2000. Effect of industrial processing on chlorophyll content of broccoli. Journal of the Science of Food and Agriculture, 80:1447-1451.
CrossRef - Nooden, L.D., 1988a. The phenomena of senescence and aging. In: Senescence and Aging in Plants (edited by L.D. Nooden & A.C. Leopold). Pp. 1-50. London, UK: Academic Press.
CrossRef - Nooden, L.D., 1988b. Postlude and prospects. In: Senescence and Aging in Plants (edited by L.D. Nooden & A.C. Leopold). Pp. 499-518. London, UK: Academic Press.
CrossRef - Okada, K., Inoue, Y., Satoh, K. & Katoh, S., 1992. Effects of light on degradation of chlorophyll and proteins during senescence of detached rice leaves. Plant and Cell Physiology, 33: 1183-1191.
- Page, T., Griffiths, G. and Buchanan-Wollaston, V., 2001. Molecular and biochemical characterization of postharvest senescence in broccoli. Plant Physiology, 125: 718-727.
CrossRef - Pankaj, B.P., Umezuruike, L.O., Fahad, Al-JAl-S., 2011. Colour measurement and analysis in fresh and processed foods: a review. Food and Bioprocess Technology, 6: 36-60.
- Pogson, B,J., and Morris, S.C., 1997. Consequences of cool storage of broccoli on physiological and biochemical changes and subsequent senescence at 20°C. Journal of the American Society for Horticultural Science, 122:553-558.
- Sant’Anna, V., Gurak, P.D., Marczak, L.D.F. & Tessaro, I.C., 2013. Tracking bioactive compounds with colour changes in foods-A review. Dyes and Pigments, 98: 601-608.
CrossRef - Shewfelt, R.L.,2000. Fruit and vegetable quality. Ιn: Fruit and Vegetable Quality: An Integrated View (edited by R.L. Shewfelt & B. Brückner). Pp. 144-157. Lancaster, UK: Technomic Press.
- Singh, R., Wang, D.J and D.K Salunke. 1972. Controlled atmosphere storage of lettuce. 2. Effects on biochemical composition of the leaves. Journal of Food Science, 37: 52-55.
CrossRef - Sitte, P., Falk, H., Liedvogel, B., 1980. Chromoplasts. In: Pigments in Plants. Ed. F.C. Czygan, pp.117-148. Stuttgart Gustav Fisher, 2nd ed.
- Suthumchai, W., Matsui, T., Kawada, K. and Kosugi,Y., 2007. Sugar metabolizing enzymes activities in lettuce head during low temperature storage. Asian Journal of Plant Sciences, 6(4):568-576.
CrossRef - Taylor, A.O. & Craig, A.S., 1971. Plants under climatic stress, Il. Low temperature, high light effects on chloroplast ultrastructure. Plant Physiology, 47(5): 719-725.
CrossRef - Thompson, J.E.,1988. The molecular basis for membrane deterioration during senescence. In: Senescence and Aging in Plants (edited by L.D. Nooden & A.C. Leopold). Pp. 52-84. London, UK: Academic Press.
CrossRef - Watada, A.E., N.P. Ko and D.A. Minott,1996. Factors affecting quality of fresh-cut horticultural products. Postharvest Biology and Technology, 9:115-125.
CrossRef - Zhuang, H., Barth, M.M and D.F Hildebrand, 1994. Packaging influence total chlorophyll, soluble protein, fatty acid composition and lipoxygenase activity in broccoli florets. Journal of Food Science, 59(6): 1
CrossRef

This work is licensed under a Creative Commons Attribution 4.0 International License.





