Introduction
PCOS, also referred to as Stein-Leventhal syndrome, impairs the reproductive health of women. It is known to be the most prevalent hormonal and metabolic condition among women of reproductive age1. The primary cause of PCOS remains unknown, and disease control approaches are influenced by the estimated prevalence of PCOS in a given area. The condition has a high prevalence in women of Indian Descent. By applying Rotterdam’s criteria and the American Endocrine Society (AES criteria), the combined prevalence was found to be nearly 10%, but the National Institute of Health (NIH) criteria yielded a prevalence of 5.8% according to a study conducted in 2019. 1 Globally, one in ten women of childbearing age are affected with PCOS. Hormonal imbalances and metabolism imbalances that women with PCOS experience may have an impact on their general health and appearance. Another typical and manageable cause of infertility is PCOS which drastically affects the reproductive health of women.2 The three main attributes of PCOS are as follows:
Oligmennorhoea, indicating irregular ovulation (release of eggs from the ovaries).
Clinical or biochemical hyperandrogenism is linked to excess production of androgen leading to hirsutism, acne, alopecia, etc.
Polycystic ovaries: PCOS patients have enlarged ovaries containing numerous follicles that appear as fluid-filled sacs surrounding the eggs.
High insulin levels are frequently observed in women with PCOS. The pancreas produces an important hormone called insulin, which is helpful for the cells of the body to convert simple sugar units (glucose) into energy. If insulin is not adequately produced, it raises the glucose level in the body. This is referred to as insulin resistance (IR). Due to IR, the body starts producing abnormally high levels of insulin for the conversion of glucose which could directly elevate the level of androgen (male hormones).
A greater body mass index may also contribute to insulin resistance. People with PCOS frequently struggle with this problem because of insulin resistance, which can make weight loss more difficult. A diet rich in refined carbohydrates, such as starchy and sugary meals, might make it more difficult to control insulin resistance and, consequently, weight loss. 3
Stress may be caused by the symptoms of PCOS. Although the reason for this is unclear, depression and insulin resistance are related. According to one idea, insulin resistance alters how the body produces some hormones, which can result in chronic stress and depression. Stress is known to be induced by PCOS itself, especially when it comes to the condition’s visible signs such as excessive body and facial hair. 4 Anxiety and sadness may result from these feelings of stress. Younger women with PCOS are susceptible to anxiety and other conditions that can affect their mood. Women with PCOS may experience occasional frustration. It may be possible to elevate mood and lessen some of the PCOS-related symptoms by adopting a PCOS-friendly diet and altering lifestyle. 5
Materials and Methods
Search Strategy
A five-year time restriction from December 2018 to December 2023 was applied when searching databases such as PubMed, Scopus, and Elsevier to understand the dietary modifications and nutritional interventions carried out in PCOS subjects to improve their gut microbiome composition. This systematic review includes studies on the effects of dietary modifications and Nutritional Interventions to manage and combat the symptoms of Polycystic Ovary Syndrome (PCOS). Flow Chart 1 represents the process of systematic review.
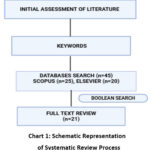 |
Chart 1: Schematic Representation of Systematic Review Process |
Selection criteria
This review includes studies on the effects of Dietary Modification and Nutrition Interventions carried out in subjects with PCOS. It has covered the research papers, and clinical trials and additionally includes certain findings from review articles.
Results
This review explores the dimensions of improving PCOS symptoms by improving gut health using dietary interventions.
The findings of the explored articles have been discussed in the discussion.
Discussion
Effects of PCOS
According to the World Health Organization (WHO) report (2023), Mental health issues and social stigma can result from PCOS’s biological and psychological repercussions, which are especially linked to obesity, body image, and infertility. 6
PCOS is an intricate condition that affects endocrine (hormonal) and metabolic functions which can cause irregular menstrual flow, infertility, weight gain, and insulin resistance (IR). Improper lifestyle or Dietary choices, environmental exposure, genetics, and alterations in the gut microbiome are some of the risk factors that can cause PCOS, in turn becoming a risk factor that could result in metabolic syndrome due to abnormal levels of insulin, higher Body Mass Index (BMI) and hormonal imbalance. 7 The pathogenic function of gut microbiota dysbiosis in PCOS development is possible. PCOS is a syndrome is also characterized by insulin resistance, which is aggravated by adipose tissue accumulation linked to hyperandrogenism, as well as dysfunction with lipotoxicity and oxidative stress. As a result, psychological, reproductive, and metabolic abnormalities are present throughout the whole clinical spectrum of the disease. The key pathophysiological factors of PCOS are hyperandrogenism and hyperinsulinemia due to decreased insulin sensitivity and hormonal dysfunction, which can also lead to type II diabetes and endometrial cancer. 8
Benefits of dietary intervention in PCOS
Treatments and dietary practices are crucial for the recovery of PCOS patients and a healthy diet and maintenance of proper nutritional status are crucial in the prevention of this illness. A variety of dietary approaches, such as the ketogenic diet (High Fat Diet), Mediterranean Diet, and Dietary Approaches to Stop Hypertension (DASH), have been shown to influence weight loss, and insulin production and enhance reproductive function. Women with PCOS benefit from diets that lower rates of obesity and insulin resistance (IR); nevertheless, to create a customized and long-lasting nutritional intervention, the condition of obesity and IR should be assessed early in the disease. 9 It has been demonstrated that the introduction of a diet, particularly a low-GI, normalizing sleep patterns, and increasing daily physical activity are modifiable factors that contribute to a considerable improvement in these parameters. Furthermore, it appears that adding antioxidants and herbs to the diet can help: Curcuma longa (turmeric) has helped reduce chronic inflammation; is also helpful in intestinal dysbiosis when incorporated with probiotics such as yogurt, buttermilk, and fermented products. 10 A certain food composition may not always be preferred, even though dietary treatments are the primary treatment in PCOS. While reaching an ideal body weight reduces PCOS symptoms, the data to determine which dietary strategy works best for this purpose is insufficient. Currently, the most popular diets for treating PCOS are the Mediterranean, diets low in calories & carbohydrates, and Dietary Approaches to Stop Hypertension (DASH) diets. 11 Therefore, the findings suggest that food sources low in glycemic index, and high in fiber along with probiotics like buttermilk can help in reducing and managing the symptoms of PCOS. Incorporating herbs such as turmeric, ginger, and cinnamon could also be helpful as they are potential antioxidants that can help reduce inflammation.
Micronutrient supplementation in PCOS
A randomized control trial divided thirty PCOS women into two groups: the group that received “multi-nutrient supplementation” i.e. unlabelled soft capsules (two) containing 200 μg folic acid each, for three months) and a “control group” (one unlabelled soft capsule containing omega-3 fatty acids and one unlabeled tablet containing nutrients such as folic acid, selenium, vitamin E, catechin, glycyrrhizin, and co-enzyme Q-10). Anti-Mullerian hormone (AMH), total testosterone, and androstenedione were the primary outcome criteria. Furthermore, luteinizing hormone (LH), follicle-
stimulating hormone (FSH) and the ratio of Luteinizing Hormone (LH) and follicle-stimulating hormone (FSH) were examined. In terms of PCOS-specific parameters (LH: FSH), women with PCOS benefit from a micronutrient supplementation that comprises omega-3 fatty acids, folic acid, selenium, vitamin E, catechin, glycyrrhizin, and co-enzyme Q10. This supplementation should be administered for a minimum of three months. 12 Women with PCOS and Metabolic Syndrome (MetS) had a significantly lower dietary intake of antioxidant micronutrients (selenium, zinc, chromium, carotenoids, and Vitamin E) than the control group (P < 0.05). 13 According to reports by the European Society of Parenteral and Enteral Nutrition (ESPEn), 2022, it is reported that modifying food habits to manage PCOS has been a major area of study over the past few decades. The impact of macronutrients and micronutrients on the management of various clinical features of PCOS is covered in this review along with recent research and clinical trial studies. Dietary therapies are important because of the potential link between PCOS and the quantity and quality of micronutrients and macronutrients, as well as the need to treat PCOS as a complex disorder. An increasing number of clinical trials examining the impact of macronutrient treatments (such as modification of fat, carbohydrate, and protein levels) and micronutrients (such as zinc, chromium, selenium, vitamin D, inositol, and vitamin E) have been conducted and found to be promising in improving the effects of PCOS reported 41 supplements. Among these, inositols (n = 86), vitamin D (n = 53), N-acetylcysteine (n = 27), and omega-3 fatty acids (n = 25) were the most frequently studied. The findings showed that additional research is needed to determine the psychological effects of micronutrient and nutraceutical supplementation in women with PCOS. Table 1 shows the effects of micronutrient supplementation on PCOS. Furthermore, it is necessary to conduct sufficiently powered primary investigations to determine the therapeutic doses required for clinical benefits. 14
Table 1: Effects of Micronutrients in PCOS
|
Symptoms |
Beneficial Nutrients | Effects |
|
Hyperandrogenism |
Inositol, omega-3 fatty acids, calcium | Helps in reducing androgen, and follicular arrests and improves oocyte maturation |
| Elevated Homocysteine | B complex vitamins
(B1, B6, B12, Folate) |
Helps in Insulin Resistance and weight management. |
|
Dyslipidaemia |
Vitamin E | Helps in dissolving fat and acts as an antioxidant thereby reducing inflammation. |
| Hormonal Imbalance | Selenium, Vitamin E, Folate, Zinc |
Improves reproductive health, regularizes menstruation, and helps in weight management. |
| Mood Disorders | Magnesium,
Vitamin D |
Helps in relieving mood swings and fatigue |
Seed cycling in PCOS
A randomized controlled trial was conducted to understand the effect of Seed cycling on PCOS. The participants of the study were divided into three groups (T0, T1, and T2) of PCOS women (20 women in each group). The first of these three groups was the control group (T0). The experimental group (T1) was comprised of the second group. In T1, METFORMIN 500 mg tab/day and a portion of the control diet were administered to 20 PCOS-afflicted women for 90 days. The third group (T2) comprised the experimental group. Within this cohort, 20 more PCOS-afflicted women received a ninety-day treatment regimen comprising seed cycling and a portion control diet. It was observed that the levels of FSH and LH significantly improved over the twelve weeks in the T1 and T2 groups respectively whereas the T0 group had no improvement. In conclusion, the seed cycling strategy works well and produces noticeable outcomes in PCOS-affected women. Women’s hormonal disturbances can be improved by seed cycling, which supports a healthy lifestyle. Irfan et al (2021) reviewed several review papers that included information about the distinct roles of flax, sesame, pumpkin, and sunflower seeds and concluded that the main component of these plant-based seeds that helps PCOS patients with hormonal imbalances and insulin resistance is n-3 fatty acids. 15 Table 2 shows the effects of seed cycling in PCOS.
Table 2: Effects of Seed Cycling in PCOS
|
Phase |
Predominant Hormone | Seeds | Role |
| Follicular Phase | Estrogen | Flax seed and Pumpkin seed |
Aids in hormone balance, helps in reducing androgen level, Pre-menstrual syndrome (PMS) symptoms and regularize menstruation as the oil seeds contain essential fatty acids such as omega 3 and omega 6 fatty acids and nutrients such as vitamin E, Vitamin B6 and zinc which will help in improving reproductive health by balancing the production of estrogen and progesterone accordingly. |
|
Luteal Phase |
Progesterone |
Sunflower Seed and Sesame seed |
Resistant starch
The component of starch known as resistant starch is that which is not broken down by microbes in the small intestine but it undergoes fermentation in the colon and produces short-chain fatty acids (SCFAs), that may have metabolic consequences. To that end, this review attempts to provide pertinent studies on the health advantages of resistant starch consumption as well as how it affects the physiological attributes to be assessed, including lipid metabolism, and intestinal. Health, glycaemic balance, and body weight. 16 shows the effect of resistant starch on health. Although preliminary, resistant starch may have a positive effect on lowering the presence of diabetes, obesity, cholesterol problems, and intestinal health. Over ten years of research, the health implications of resistant starch were conducted once it was discovered that low-glycemic index diets were made with resistant starch. Isocaloric investigations were performed by determining the metabolizable energy of the resistant starch. In rodent studies, the fermentation of resistant starch led to an apparent improvement in gut health as evidenced by elevated levels of short-chain fatty acids, a seemingly positive shift in the microbiota, and elevated expression of genes related to normal, healthy cell proliferation and apoptosis of potentially cancerous cells. Resistant starch increases insulin sensitivity in humans. Only one study out of several, though, shows a rise in serum GLP-1 in response to resistant starch added to the diet This suggests that the development of greater insulin sensitivity may entail additional processes, such as increased intestinal gluconeogenesis or higher adiponectin. 17
Effects of resistant starch in PCOS
In recent times, several dietary therapies have been widely employed as an innovative treatment for PCOS. In their work, Resistant Starch (RS) based metabolite (Soy Isoflavone) and gut microflora regulations effectively reduced the severity of reproductive characteristics resembling PCOS while upregulating gut barrier indicators and butyric acid expression. Even after letrozole treatment, PCOS in model rats was less severe when soy isoflavones (0.05%) and resistant starch (11%) were consumed. Treatment with an antibiotic cocktail inhibited the gut metabolism of soy isoflavones and had no appreciable effect on mitigating PCOS-like symptoms. Menstrual irregularity was strongly associated with bacterial taxa such as Blautia, Dorea, and Clostridium when resistant starch was consumed. Furthermore, the consumption of resistant starch increased butyric acid content. Table 3 shows the benefits of resistant starch consumption to overall health. Finally, we suggest that the severity of PCOS reproductive characteristics can be significantly reduced by utilizing both dietary therapies and gut microbiota modulations. Human Studies with RS Treatment have not yet been conducted and the data have not yet been published. 18, 19
Table 3: Benefits of Resistant Starch Consumption
|
Food Sources |
Benefits |
| Green Banana |
Regulates Insulin and improves digestive health |
|
Potatoes |
Helps in managing cholesterol and blood sugars, helps in satiety |
| Oats |
Helps in weight management, and maintains blood sugar levels. |
|
Legumes such as beans, Chickpeas, peas |
A good source of plant-based protein, improves digestive and heart health, improves blood sugar levels. |
| Nuts |
Helps in weight management, and immunity, lowers blood sugar levels, and regulates mood. |
Benefits of gut microbiome in health
According to previous studies, R.bromii, Bifidobacterium species, and E.rectale are involved in the fermentation of the large intestine as they release butyrate by the degradation of resistant starch in the human colon and help improve gut health. All these species play a vital role in cell wall breakdown in the colon, hence this study will focus on identifying the gut microbiome composition of these species, as it is focussed on resistant starch supplementation. The bacterium Ruminococcus plays a major role in the fermentation of dietary fiber to create SCFAs and butyrate, which are vital sources of energy for intestinal epithelial cells and are beneficial in regulating insulin in the body. Probiotic bacteria such as Eubacteria rectale, Lactobacillus, and Bifidobacterium are known to lower obesity. Bifidobacteria controls fat metabolism and decreased fat mass. In addition, it aids in the digestion of fiber, the prevention of infections, and the production of vital substances such as B vitamins (folate, B12 etc) and healthy fats which helps in managing blood flow and regulating the menstrual cycle. Table 4 shows the nutrients that are helpful for maintaining proper gut health. 20, 21
Table 4: Nutrients essential for gut health
|
Nutrients |
Food Sources |
|
Probiotics |
Yogurt, Buttermilk, Sauerkraut, Kimchi |
| Fibre |
Vegetables such as carrots, beans, cabbage, green leafy vegetables, Fresh Fruits such as guava, apple, pear, sweet lime, orange, Whole grains such as rice, wheat, rice flakes, millets, Legumes such as chickpea, green gram, kidney beans |
|
Omega 3 Fatty Acid |
Flax Seed, Pumpkin Seed, Sesame Seed, almonds, Walnuts, Fatty Fish, avocados |
| Herbs and Spice |
Turmeric, Cinnamon, Ginger, Cumin Seeds, Carrom Seeds, Fenugreek Seeds, Fennel Seeds |
|
Vitamin D |
Eggs, Fatty Fish, Fortified Milk, Mushrooms, additional exposure to sunlight, and supplementation are essential. |
| Calcium |
Sesame Seeds, Nuts like almonds, wholegrains like wheat, ragi, Green leafy vegetables, broccoli, dairy products specifically curd. |
|
Iron |
Green Leafy Vegetables, Legumes, seafood, organ meats |
| Zinc |
Pumpkin seeds, sunflower seeds, nuts like almonds, walnuts, legumes |
|
Magnesium |
Avocados, almonds, spinach, banana |
| Vitamin B3 |
Nuts like peanuts, mushrooms, avocado |
|
Vitamin B12 |
Dairy Products, eggs, nuts, legumes |
| Folate |
Green Leafy Vegetables, Legumes, avocado, citrus fruits |
|
Vitamin C |
Indian Goose Berry, Bell Peppers, Guava, Citrus fruits |
Effects of gut micro-biome in PCOS
The incidence and progression of PCOS have been demonstrated to be significantly influenced by the gut microbiome. Prebiotics, probiotics, and various dietary approaches are beneficial in PCOS. A multitude of molecular routes, such as the preservation of homeostasis and control of lipid and blood glucose levels, is how the gut microbiota manifests its impact in PCOS. There are limited reports of studies involving humans: – however, the impact of the gut microbiota on PCOS has been extensively documented in animal models. Prebiotics, probiotics, and polyphenols work in several ways to improve the gut microbiota, including increasing its diversity and upregulating PCOS. 22 A first meta-analysis that associated PCOS with gut microbial dysbiosis reported that compared to healthy controls, the evenness and phylogenetic abundance of the gut microbiota in PCOS patients were lower, but overall diversity indices were maintained. Changes in particular bacteria and bacterial clusters suggest a connection between gut hormone and immunity interaction. Taken together these traits in the gut microbiomes suggest that further research should be conducted to determine the cause of the disease and possible new targets for treatment. These findings also support the findings of earlier research, which indicate that pro-inflammatory rather than anti-inflammatory bacteria are the predominant microbiota in patients with PCOS with gut dysbiosis. 23 Current research on the mechanisms causing polycystic ovarian syndrome and the roles played by gut bacteria in the emergence of IR which is insulin resistance due to decreased insulin sensitivity. Reproductive failure is the main pathogenic cause of IR. Hyperinsulinemia is a disorder characterized by excessive insulin secretion. Chronic inflammation, follicular dysplasia, alterations in endometrial receptivity, infertility, and abortion are associated with IR/hyperinsulinemia. It has also been linked to anxiety, depression, and other psychiatric illnesses, and increases the risk of pregnancy difficulties. Environmental exposures and Gut Microflora, also known as the “second genome” present in the human body can boost immunity and metabolism. IR can result from dysbiosis of the gut microbiota and is strongly associated with PCOS. Therefore, a prebiotic-based dietary approach influences Gut Health thereby improving the overall health of PCOS individuals. 24
Conclusion
The review concludes that the presented studies have a direct relationship with dietary changes and gut microbiome composition in individuals with PCOS. They have been proven to be beneficial in improving the menstrual cycle and insulin resistance thereby playing a significant role in weight loss by improving gut health. Therefore, maintaining a balanced diet and incorporating micronutrients that will help reduce the symptoms of PCOS as part of the daily diet is essential. Although the data related to dietary therapies are limited, future research on the effects of dietary therapies in managing the symptoms of PCOS by improving gut health will help prove the strength of nutritional interventions in PCOS.
Strengths and limitations of the study
Strengths
One of the strengths of this study is that it focused on determining the effects of dietary therapies and micronutrient supplementation in reducing various symptoms of PCOS
Limitation
The main drawback of this study is that there are very few studies on determining the role of Gut Health in PCOS.
Acknowledgement
I would like to express my gratitude to Department of Clinical Nutrition, Faculty of Allied Health Sciences at Sri Ramachandra Institute of Higher Education and Research for the support to carry out this review as a part of my Ph.D. work.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest
The authors do not have any conflict of interest.
Data availability statement
The manuscript incorporates all datasets produced or examined throughout this research study.
Ethical approval
This research did not involve human participants, animal subjects, or any material that requires ethical approval.
Informed consent statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to Reproduce Material from other Sources
The author(s) did not reproduce material from any other sources. All the Figures and tables are original.
Clinical trial registration
This research does not involve any clinical trials
Author Contributions
- Nandita Krishnan: Conceptualization, Methodology, Writing – Original Draft.
- Palaniappan Narayanan: Visualization, Supervision.
- Deepa Parvathi Venkatachalam: Analysis, Writing – Review & Editing.
- Supriya Velraja: Project Administration.
References
- Ganie MA, Vasudevan V, Wani IA, Baba MS, Arif T, Rashid A. Epidemiology, pathogenesis, genetics & management of polycystic ovary syndrome in India. Indian J Med Res. 2019 Oct;150(4):333-344. doi: 10.4103/ijmr.IJMR_1937_17. PMID: 318239152
CrossRef - Bulsara, J., Patel, P., Soni, A. and Acharya, S. A Review: Brief Insight into Polycystic Ovarian Syndrome. Endocrine and Metabolic Science. 2021 3, https://doi.org/10.1016/j.endmts.2021.100085
CrossRef - Che X, Chen Z, Liu M, Mo Z. Dietary Interventions: A Promising Treatment for Polycystic Ovary Syndrome. Ann Nutr Metab. 2021;77(6):313-323. doi: 10.1159/000519302
CrossRef - Guo J, Tan L, Kong L. Impact of dietary intake of resistant starch on obesity and associated metabolic profiles in human: a systematic review of the literature. Crit Rev Food Sci Nutr. 2021;61(6):889-90
CrossRef - Rackerby B, Kim HJ, Dallas DC, Park SH. Understanding the effects of dietary components on the gut microbiome and human health. Food Sci Biotechnol. 2020 Dec 16;32(13):1949. doi: 10.1007/s10068-020-00844-1.
CrossRef - Singh S, Pal N, Shubham S, Sarma DK, Verma V, Marotta F, Kumar M. Polycystic Ovary Syndrome: Etiology, Current Management, and Future Therapeutics. J Clin Med. 2023 Feb 11;12(4):1454. doi: 10.3390/jcm12041454. PMID: 36835989; PMCID: PMC9964744.
CrossRef - Hoeger KM, Dokras A, Piltonen T. Update on PCOS: Consequences, Challenges, and Guiding Treatment. J Clin Endocrinol Metab. 2021 Mar 8;106(3):e1071-e1083. doi: 10.1210/clinem/dgaa839. PMID: 33211867.
CrossRef - Szczuko M, Kikut J, Szczuko U, Szydłowska I, Nawrocka-Rutkowska J, Ziętek M, Verbanac D, Saso L. Nutrition Strategy and Life Style in Polycystic Ovary Syndrome-Narrative Review. Nutrients. 2021 Jul 18;13(7):2452. doi: 10.3390/nu13072452. PMID: 34371961; PMCID: PMC8308732.
CrossRef - Juhász AE, Stubnya MP, Teutsch B, Gede N, Hegyi P, Nyirády P, Bánhidy F, Ács N, Juhász R. Ranking the dietary interventions by their effectiveness in the management of polycystic ovary syndrome: a systematic review and network meta-analysis. Reprod Health. 2024 Feb 22;21(1):28. doi: 10.1186/s12978-024-01758-5. PMID: 38388374; PMCID: PMC10885527.
CrossRef - Hager M, Nouri K, Imhof M, Egarter C, Ott J. The impact of a standardized micronutrient supplementation on PCOS-typical parameters: a randomized controlled trial. Arch Gynecol Obstet. 2019 Aug; 300(2):455-460. doi: 10.1007/s00404-019-05194-w.
CrossRef - Zaeemzadeh, N., Jahanian Sadatmahalleh, S., Ziaei, S. et al. Comparison of dietary micronutrient intake in PCOS patients with and without metabolic syndrome. J Ovarian Res. 2021; 14, 10. https://doi.org/10.1186/s13048-020-00746-0
CrossRef - Scannell N, Mantzioris E, Rao V, Pandey C, Ee C, Mousa A, Moran L, Villani A. Type and Frequency in Use of Nutraceutical and Micronutrient Supplementation for the Management of Polycystic Ovary Syndrome: A Systematic Scoping Review. Biomedicines. 2023 Dec 18;11(12):3349. doi: 10.3390/biomedicines11123349. PMID: 3813757 14.
CrossRef - Alesi S, Ee C, Moran LJ, Rao V, Mousa A. Nutritional Supplements and Complementary Therapies in Polycystic Ovary Syndrome. Adv Nutr. 2022 Aug 1;13(4):1243-1266. doi: 10.1093/advances/nmab141. PMID: 34970669; PMCID: PMC9340985.
CrossRef - Rasheed N, Ahmed A, Nosheen F, Imran A, Islam F, Noreen R, Chauhan A, Shah MA, Amer Ali Y. Effectiveness of combined seeds (pumpkin, sunflower, sesame, flaxseed): As adjacent therapy to treat polycystic ovary syndrome in females. Food Sci Nutr. 2023 Mar 25;11(6):3385-3393. doi: 10.1002/fsn3.3328. PMID: 37324929; PMCID: PMC10261760.
CrossRef - Irfan T. ., Seher K. ., Rizwan B., Fatima, S. ., & Sani, A. Role of Seed Cycling in Polycystic Ovarian Syndrome: Seed Cycling in PCOS. Pakistan BioMedical Journal. 2021; 4(2), 21–25. https://doi.org/10.54393/pbmj.v4i2.12217.
CrossRef - Zhang M, Wang O, Cai S, Zhao L, Zhao L. Composition, functional properties, health benefits and applications of oilseed proteins: A systematic review. Food Res Int. 2023 Sep;171:113061. doi: 10.1016/j.foodres.2023.113061. Epub 2023 May 29. PMID: 37330842.
CrossRef - Tomova A, Bukovsky I, Rembert E, Yonas W, Alwarith J, Barnard ND, Kahleova H. The Effects of Vegetarian and Vegan Diets on Gut Microbiota. Front Nutr. 2019 Apr 17;6:47. doi: 10.3389/fnut.2019.00047. PMID: 31058160; PMCID: PMC6478664.
CrossRef - Chen J, Chen X, Ho CL. Recent Development of Probiotic Bifidobacteria for Treating Human Diseases. Front Bioeng Biotechnol. 2021 Dec 22; 9: 770248. doi: 10.3389/fbioe.2021.770248. PMID: 35004640; PMCID: PMC8727868.20.
CrossRef - Corrie L, Awasthi A, Kaur J, Vishwas S, Gulati M, Kaur IP, Gupta G, Kommineni N, Dua K, Singh SK. Interplay of Gut Microbiota in Polycystic Ovarian Syndrome: Role of Gut Microbiota, Mechanistic Pathways and Potential Treatment Strategies. Pharmaceuticals (Basel). 2023 Jan 28;16(2):197. doi: 10.3390/ph16020197. PMID: 37259345; PMCID: PMC9967581.21.
CrossRef - Li P, Shuai P, Shen S, Perturbations in gut microbiota composition in patients with polycystic ovary syndrome: a systematic review and meta-analysis. BMC Med, 2023, 21: 302 https://doi.org/10.1186/s12916-023-02975-8
CrossRef - He, Ff, Li, Ym, Role of gut microbiota in the development of insulin resistance and the mechanism underlying polycystic ovary syndrome: a review. J Ovarian Res, 2020, 13, 73 https://doi.org/10.1186/s13048-020-00670-3
CrossRef - Adrianna Bojarczuk, Sylwia Skąpska, Amin Mousavi Khaneghah, Krystian Marszałek, Health benefits of resistant starch: A review of the literature. Journal of Functional Foods. 2022; 93: 105094, https://doi.org/10.1016/j.jff.2022.105094.
CrossRef - Keenan MJ, Zhou J, Hegsted M, Pelkman C, Durham HA, Coulon DB, Martin RJ. Role of resistant starch in improving gut health, adiposity, and insulin resistance. Adv Nutr. 2015 Mar 13; 6(2):198-205. doi: 10.3945/an.114.007419. PMID: 25770258; PMCID: PMC4352178.
CrossRef - Liyanage, G. S. G., Inoue, R., Fujitani, M., Ishijima, T., Shibutani, T., Abe, K., Kishida, T., & Okada, S, Effects of Soy Isoflavones, Resistant Starch and Antibiotics on Polycystic Ovary Syndrome (PCOS)-Like Features in Letrozole-Treated Rats. Nutrients. 2021; 13(11): 3759. https://doi.org/10.3390/nu13113759
CrossRef
Abbreviations
AES-American Endocrine Society
IR- Insulin Resistance
NIH- National Institute of Health
PCOS-Polycystic Ovarian Syndrome
RS- Resistant Starch

This work is licensed under a Creative Commons Attribution 4.0 International License.





