Introduction
Prostatic cancer is among the frequent cancers in the world after the skin cancer in terms new cancer cases and annual causalities.1 About 0.3 million new cases has been registered in 2024,. The data for the onset of prostrate cancer (PC) varies in the developed and under developing countries.2 Key risk factors of the PC are gender and aging associated as indicated by the statistics are reported in men and during mid 60s.3 Family history, race which is AA males and brca1/brca2 mutations all are the related primary risk factors but these remained a syptomatic during early years.4 Thus, PC and its progression is a complex phenomenon that comprised of genotypes, environment and hormonal dysregulation. This gradually progress from prostatic intraepithelial neoplasia (PIN) and localized adenocarcinoma that metastasize to other organs. Underlying mechanism for carcinogenesis is based on an alteration in signal transduction of androgen receptors, loss of PTEN tumor suppressor gene and even gain of multiple copies of MYC and related genes. Several epigenetic modes imply DNA methylation and histones in carcinogenesis and malignant transformation have been proposed.5
Treatment of PC depends on the stage of cancer and resources such as active surveillance or surgery followed by radiation and hormonal agents such as ADT, chemotherapy, immunotherapy or enzyme inhibitors PARP.6 PARP inhibitors and immunotherapies are thought as novel and still demand investigations to improve treatment efficacy and outcomes. Understanding epidemiology, risk factors, pathogenesis, and molecular mechanisms of PC, is crucial for developing effective prevention strategies and superior treatment options. Ongoing research of personalized medicine and precision oncology holds promise for improving outcomes and quality of life for PC patients.7
Chemotherapies are effective methods of cancer control especially in the high-risk populations. Chemopreventive molecules prevent cancer in early stages of carcinogenesis, later on complications, and mortalities. Mechanistically, these drug molecules follow diverse cellular pathways, modulate carcinogenesis and upregulate detoxification enzymes at translational level. Moreover, these drug molecules act as strong antioxidants that neutralize DNA damaging reactive oxygen species (ROS), alters fundamental cell signaling involving nuclear factor kappa B (NF-κB) and PI3K/Akt pathway essential for cell proliferation and survival, and prevent COX-2 expression.8 In addition, these natural bioactive drug molecules promote apoptosis in precancerous or cancerous cells without affecting normal cells.9 Recent research highlights the effectiveness of specific substances in maintaining the malaise from PC.9 For instance, finasteride (25% reduction) and dutasteride (23% reduction) have shown notable decrease in PC development in the patients as indicated by prostrate cancer prevention trial (PCPT) studies.10
Tea plant (Camellia sinensis) native to East-Asia and Southeast-Asia much valued for its leaves that are processed into different types of tea.11 Green tea, made from unoxidized leaves, which keeps its light flavor and contains bioactive molecules i.e., catechins, which are beneficial for health by posing antioxidant and anti-inflammatory effects. Black tea is also brewed from C. Sinensis leaves is fully oxidized boasting of rich flavors comprised of theaflavins and thearubigins which have metabolic and cardiovascular health benefits. Oolong tea is partially oxidized, exhibits complex flavors and is noted for its potential in weight management and regulating metabolic activities.12 White tea is one of the least processed form of young leaves and possess low flavor profile bearing potentially more antioxidants. Moreover, tea EGCG probably has the potential to influence carcinogenic activity. Tea polyphenols including EGCG show antioxidant activities and altered signal transduction pathways having potential roles in carcinogenesis.13
Tea holds significant historical and cultural importance, evolving from its origins as a medicinal beverage in ancient times to becoming a globally beloved drink.14 Tea antioxidants have various benefits including promotion of cardiovascular health, enhancement of attention span and memory, prevention of chronic ailments, reducing inflammation, and overall body immunity. More and more studies are exploring the diverse health benefits of tea and its potential use in modern day life to prevent life style oriented ailments, significance in global health and wellness.15
BACs in C. sinensis
Studies confirmed the presence of different polyphenols in tea plant which have string pharmacological potential. C. sinensis is rich in antioxidant catechins which further classified into epigallocatechingallate (EGCG), epigallocatechin (EGC), epicatechingallate (ECG), and epicatechin (EC). EGCG, in particular, has garnered attention for its potential health benefits, including anti-cancer, anti-inflammatory, and cardioprotective effects.12 Regarding flavonoids, C. sinensis contains quercetin, kaempferol, and myricetin, which contribute to its antioxidant activity and potential health benefits.16 There exists numerous differences in purine alkaloids and are thought as quality components in tea. C. sinensis naturally contains caffeine, a stimulant that can enhance alertness and concentration. Caffeine contents varies among different tea types, with higher concentrations in black tea compared to green and white teas.17 Another alkaloid theobromine, found in cocoa and chocolate may contributes to stimulating effects.18 Presence of theanine moderate the stimulating effects of caffeine, contributing to reputation of tea as a calming beverage worldwide.19
C. sinensis is rich in vitamins and minerals, and their concentration depends on tea type and brewing methods. Commonly vitamin C, E, and B-complex, various minerals like Mn, K, and F are also present.19 Tea leaves also contain certain volatile oils including terpenes, sesquiterpenes, and other aromatic compounds, which contribute to their aroma and flavor profiles (Figure 1).20
Mechanism of action of C. sinensis BACs
BACs in C. sinensis represent a broad category of compounds that contribute to its health-promoting properties.21 Of these compounds, catechins are notable, and flavonoids are known for their higher antioxidant potential. Catechins like EGCG is important for its beneficial effects on the human body including anticancer, anti-inflammatory, and cardiovascular disease prevention effects.22 Tea flavonoids viz. quercetin, kaempferol and myricetin add strength to the antioxidant properties and potentially bring other benefits to the consumer.23 Alkaloids on the other hand, include caffeine and theobromine which are well-known stimulant to boost central nervous system are present in higher quantity in black tea.24
Tea theanine could also decrease the alertness caused by caffeine to help promote the relaxing image of tea consumption. Tea type and brewing process may determine the quantity of different vitamins and minerals.24 Minerals additives through volatile oils present in tea leaves such as terpenes and sesquiterpenes found to further enhance aroma and taste.25 By these diverse BACs, in addition to the pleasant taste of the drink, tea can provide beneficial health effects that researchers continue investigating since decades.26 Some important tea BACs along with their mechanism of action tabulated in table 1.
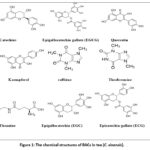 |
Figure 1: The chemical structures of BACs in tea (C. sinensis). |
Table 1: BACs in C. sinensis and their mechanism of action.
| Sr. No. | BACs | Mechanism of action, in vivo and in vitro studies | References |
| 1 | EGC | ↑Apoptosis,
↓Cell proliferation, ↑Cell signaling activity |
27-28 |
| 2 | EC | ↑Antioxidant activity,
↓Angiogenesis, ↑Immune responses |
29 |
| 3 | EGC | ↓Tumor growth,
↑Cell cycle arrest, ↑DNA repair |
30-31 |
| 4 | EGC | ↓Inflammations,
↓ROS, ↑Enzymatic activity |
27, 32 |
| 5 | Catechin | ↑Antioxidant activity,
↓Tumor growth, ↑Metabolic activity |
33-34 |
| 6 | Gallocatechin (GC) | ↓ROS,
↓DNA damage, ↓Cancer cell growth |
33, 35 |
| 7 | Theaflavin (TF) | ↓Tumor cell invasion,
↑Apoptosis |
36-37 |
| 8 | Thearubigin (TR) | ↑Immune responses,
↑Antioxidant activity |
38 |
| 9 | Quercetin | ↓Cancer cell proliferation,
↑Cell signaling cascades, ↑Antioxidant activity |
39-40 |
| 10 | Kaempferol | ↑Apoptosis,
↓Angiogenesis, ↑Antioxidant activity |
41 |
| 11 | L-theanine | ↑Immune responses,
↓Stress-induced tumor growth |
42 |
| 12 | Caffeine | ↓DNA repair,
↑Cell cycle arrest |
43 |
| 13 | Myricetin | ↑Antioxidant activity,
↑Apoptosis, ↓Angiogenesis |
44-45 |
| 14 | Theobromine | ↑Immune responses,
↑Antioxidant activity |
46-47 |
| 15 | Chlorogenic Acid | ↑Antioxidant activity,
↓Tumor growth, ↑AMPK |
48 |
| 16 | Rutin | ↑Antioxidant activity,
↓Inflammations, ↑Cell signaling |
49 |
| 17 | Gallic Acid | ↑Antioxidant activity,
↑Apoptosis, ↓Cell proliferation |
35, 50 |
| 18 | Caffeic Acid | ↑Antioxidant activity,
↓Inflammations, ↓Tumor growth |
51 |
Mechanisms of Action in Chemoprevention
Antioxidant Activity and Redox Homeostasis
Free radicals are usually very unstable and always tries for stability by complexing with DNA, proteins or lipids.52 ROS produced during cellular metabolism, radiations, pollution and chemicals may cause damage to biomolecules specifically shear DNA to induce genetic mutations that lead carcinogenesis.53 Polyphenolic chemopreventive agents in C. sinensis such as antioxidant EGCG donate electrons to stabilize free radicals and help avoid cellular damages. It plays a significant role in protection from oxidative damage, maintain genomic stability against mutations and caners.54 Polyphenols can up regulate the endogenous antioxidant enzymes such as superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase by quickly reducing ROS to less toxic or stable compounds to lower oxidative stress and its associated pathologies.55 Some antioxidants quickly help activate generation of ROS such by NADPH oxidase and xanthine oxidase enzymes. Many polyphenols are capable to interact with such prooxidant metal ions such as free cellular iron and copper which are used as catalysts in the Fenton reaction and amplify formation of ROS.
Inhibition of NF-κB Pathway
C. sinensis chemopreventive polyphenols potently inhibit inflammatory NF-κB pathway to modulate immune responses through various mechanisms.56 Normally, NF-κB remain inactive in the cytoplasm by binding to its inhibitor IκB,57 and pro-inflammatory cytokines, pathogens or certain stresses activates phosphorylation of the later to release and translocate former into the nucleus.57 Once it gets inside the nucleus, NF-κB triggers transcriptional activity for the upregulation of inflammatory cytokines, cell proliferation, supports tumor development, enlargement and metastasizing.57 Tea polyphenolic compounds modulate NF-κB pathway through multiple signaling pathways. EGCG inhibit phosphorylation and subsequent degradation of IκB, preventing the release and nuclear translocation of NF-κB.58 Certain phytochemicals can directly inhibit the DNA-binding activity of NF-κB, preventing it from activating its target genes.59 By inhibiting the NF-κB pathway, these BACs could potentially reduce chronic inflammations, inhibit the proliferation and survival of cancer cells.
Activation of AMP-activated Protein Kinase (AMPK)
AMPK is prescribed as one of the molecular targets that have vital implementation for cellular energy regulation via glucose uptake, fatty acid oxidation, and mitochondrial biogenesis.21 Concerning cancer, it is revealed that there are more functions of AMPK associated with the cell metabolism, high glycolysis, lipogenesis, and cell growth.60 One another signaling pathway involved in the growth of cells and synthesis of proteins is mammalian target of rapamycinm (mTOR) which is counteracted by AMPK activation.60 The modulation of AMPK regulated metabolism is necessary for the provisions of energy rich ATPs essential for viable and enhanced growth of cancer cells. C. sinensis BACs up-regulates the level of autophagy that is a process of selective degradation of damaged cellular organelles and proteins.61 EGCG in the tea have been illustrated to regulate activation of AMPK pathway that may inhibit tumorigenesis, induce cell apoptosis, and thus considered as anticancer target and prevention strategy.62 Necrosis, a form of apoptosis, is essential in helping to control and shape the life of organisms since it removes damaged or abnormal cells. Mitochondrial cytochrome c happens to interact with Apaf-1 and pro-caspase-9 to form the apoptosome complex which in turns activates caspase-9, which further activates chain caspases to activate cell apoptosis through cleavage of various cellular substrates. Thus, chemopreventive agents including polyphenols from tea can activate caspases that directly leads to the selective killing of precancerous and cancerous cells to halt the further development of cancer.
Mitochondrial Pathways
The mitochondrial (intrinsic) pathway of apoptosis is regulated by Bcl-2 protein family that consists of the pro-apoptotic proteins (Bax, Bak) and anti-apoptotic proteins (Bcl-2, Bcl-xL etc.). Overall, the equilibrium between these proteins defines the cell’s destiny. Chemopreventive agents can influence this balance in such a way that favoring apoptosis in cancer cells is the outcome. Among these, chemopreventive agents can induce reduction of mitochondrial membrane potential, which is an essential process initiated in intrinsic apoptosis. This disruption results to the release of other apoptogenic factors such as cytochrome c, Smac/DIABLO and AIF into the cytosol from the mitochondria.63 These chemopreventive agents can suppress anti-apoptotic proteins (Bcl-2, Bcl-xL) and upregulate levels of pro-apoptotic proteins (Bax, Bak) that lead to MOMP and apoptosis. For example, it has been found that EGCG lowers the expression of Bcl-2 proteins and at the same time raises Bax proteins resulting into apoptosis. For further activation, cytochrome c moving into the cytoplasm interacts with Apaf-1 which results in the formation of apoptosome and activation of caspase-9. Some other proteins localized to the mitochondria include Smac/DIABLO, which can inactivate IAPs (inhibitor of apoptosis proteins) that, in turn, down regulates caspase activity as well as encouraging apoptosis.64
Therefore, many chemopreventive agents by modulating the rate of mitochondrial function and alteration of the ratio of the pro- apoptotic proteins and anti-apoptotic one desires directed apoptosis in cancer cells leaving normal cells intact. In fact, these sorts of selective induction of apoptosis are vital for the cancer prevention as well as cancer treatment since they destroy mutated cells without affecting the normal cells.65
Inhibition of Angiogenesis and Metastasis
Angiogenesis is an essential event during tumor growth and metastasis to receive an adequate blood circulation concerning oxygen and nutrients needed for the growth and proliferating cells. Vascular endothelial growth factor (VEGF) is a key regulator of angiogenesis that stimulate the endothelial cell proliferation and enhance the migration and formation of blood vessels.66 C. sinensis BACs inhibit angiogenesis by suppressing VEGF production or activity.67 Tea BACs inhibit the expression of VEGF at the transcriptional level in multiple cancer cell lines by down regulating Protumorigenic hypoxia-inducible factor-1α (HIF-1α), a common condition in most solid tumors.68 VEGF works through interacting with VEGF receptors (VEGFRs) on the surface of endothelial cells, and tea BACs block the interaction between the both to inhibit angiogenesis.68 Tea BACs can also interact within other pathways (PI3K/Akt pathway), activate VEGF, which are useful for regulating angiogenesis.69 This inhibition can slow down tumor growth and reduce the potential for metastasis (Figure 2).
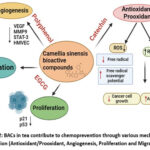 |
Figure 2: BACs in tea contribute to chemoprevention through various mechanisms of action (Antioxidant/Prooxidant, Angiogenesis, Proliferation and Migration). |
MMP Inhibition
Matrix metalloproteinases (MMPs) are enzymes capable of breaking down large amounts of the extracellular matrix (ECM). This process is very significant in the invasion of the cancer cells and formation of new tissue or metastases because it enables the cancer cell to cross the ECM front that had been formed and to proliferate into the neighboring tissues. MMPs, particularly MMP-2 and MMP-9 that are often overexpressed on the tumors and can be detected when metastatic potential and poor prognosis are increased.70 Various chemopreventive agents reported in tea plant can affect the expression of MMPs,71 and its activity and expression through multiple mechanisms. Among the above-discussed chemopreventive additives some influence the MMPs enzymatic activity right at the molecular level. For instance, EGCG has been shown to bind and fix the MMPs active site and block any further degradation of ECM.71 This direct inhibition may assist in prevention of cancer cells to invade and migrating to the rest of the body.
Tea bioactive molecules also suppress MMP at the transcript levels in cancer cells also.72 It can also inhibit other transcription factors for instance AP-1 and NF-κB that are activators of MMP genes.72 They prevent the production of MMP, reducing the overall proteolytic activity in the area surrounding the neoplastic cell proliferation and compromising the cancer cells invasion capabilities. C. sinensis chemoprevention compounds target signaling pathways which regulate expression of MMPs.72 For example, they can inhibit the MAPK and PI3K/Akt pathways by which MMPs are produced in reaction to signals.73 Such agents assist in altering these pathways that subsequently reduces MMP generation and activity besides the capacity of the cancer cells to invade the surrounding ECM. By inhibiting MMP activity and expression, chemopreventive agents can significantly reduce cancer cell invasion and metastasis. This inhibition helps to contain the tumor within its original site, preventing the spread of cancer to distant organs and improving overall prognosis.74
Preclinical Studies and in vivo/in vitro evidences
PC xenograft models involve implanting human PC cells into immunocompromised mice, allowing researchers to study tumor growth and the effects of potential chemopreventive agents in a living organism. These models offer worthy information concerning the efficiency of diverse compounds as anti-tumor agents.75 Several of these studies have shown that TP and especially EGCG effectively suppress the proliferation of PC xenografts. For instance, one investigation revealed that the effects of green tea polyphenols significantly inhibited the growth of human prostate carcinoma xenografts in nude mice. The treated group had smaller tumor size and had diminished angiogenesis related-proteins like VEGF to the untreated group (Figure 3). Another study investigated the effects of green tea polyphenols on the growth of androgen-dependent as well as androgen-independent PC xenografts. The outcomes revealed that green tea polyphenols were able to have an effect on both type of tumors and therefore, showed that green tea polyphenols could be more effective in acting as a chemopreventive agent.
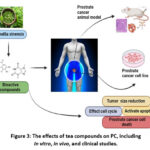 |
Figure 3: The effects of tea compounds on PC, including in vitro, in vivo, and clinical studies. |
Dose-response studies are critical for understanding the effective concentrations of chemo preventive agents and their potential toxicity. These studies aid in the identification of the greatest amount for drug effectiveness while at the same time excluding any unfavorable side effects. Research on the dose-response relationships of tea polyphenols has shown that higher doses generally result in greater tumor inhibition. However, there is a threshold beyond which no additional benefits are observed, and higher doses may even lead to toxicity. For example, a study evaluated different doses of green tea polyphenols in mice with PC xenografts. However, the research established that moderate doses exhibit considerable suppression tumor growth, very high doses did not provide additional benefits and were associated with liver toxicity.
Specifically, the in vitro experiments using the PC cell lines allow the investigation of the cellular basis of chemopreventive agents. These studies allow researchers to dissect the specific pathways and targets affected by these compounds. Numerous studies investigated the consequence of tea polyphenols on different PC cells including LNCaP, PC-3 and DU145. For example, it has been discovered that cellular studies indicate that EGCG reduces the proliferation effect on these cell lines and that it triggers apoptosis. EGCG has been found to decrease the viability of PC cells in a dose-dependent manner by modulating cell cycle regulators, such as p21 and p27, and inducing cell cycle arrest at the G1 phase.76-77 Additionally, EGCG has been demonstrated to cause apoptosis in the prostate-cancer cells by both the intrinsic and extrinsic systems. The compound has effect on the caspase-3 and caspase-9 which in turn causes DNA fragmentation and apoptotic cell death. In addition to this, EGCG has been observed to suppress Bcl-2, which is an anti-apoptotic protein and at the same increase Bax, which is a pro apoptotic protein thus enhancing the mitochondrial mediated apoptosis.78
Molecular Targets and Pathways
Chemopreventive agents work through changing the events within selected molecular targets that regulate the process of carcinogenesis.79 Tea polyphenols modulate several molecular targets in PC including NF-κB, AMPK, and PI3K/Akt pathways are noteworthy.80-81 NF-κB regulates genes involved in inflammation, cell proliferation, and survival. EGCG has been shown to inhibit the activation of NF-κB in PC cells, thereby reducing the expression of pro-inflammatory cytokines and anti-apoptotic proteins.82 Energy sensor AMPK regulates cellular metabolism and growth, while tea polyphenols activated AMPK inhibit mTOR signaling, downregulate translational activity, and cell proliferation in PC cells.83 Moreover, EGCG has been shown to inhibit PI3K activity and reduce Akt phosphorylation, leading to decreased cell survival by inducing apoptosis in PC cells.69 Tea polyphenols can disrupt the molecular mechanisms driving PC progression, highlighting their potential as effective chemopreventive agents.
Clinical studies and human evidence
Population-based epidemiological studies explored the link between drinking tea and the occurrence of PC and offers valuable insights into how tea might protect against PC by examining the dietary habits, lifestyles, and cancer rates. It is found that men who regularly drink tea tend to have a lower risk of developing PC.84 This study highlighted the potential protective effects of the green tea for advanced PC. Certain meta-analysis comprising of 24 observational studies covering 155,000 participants indicated that increased consumption of green tea helps lower the risk of developing PC especially among Asian men. No significant effects of tea were observed on dyslipidemia and there were no consistent findings concerning black tea and other kinds of tea, evidencing that further research is required to establish the effects of other tea sorts. Correlation studies find a relation between frequent tea intake with amount consumed, and the occurrence of PC. Some of these control factors include age, family history, smoking and other dietary measures to isolate the effects of tea consumption. These studies often use randomized controlled trials (RCTs) which are applied to compare the effects in the groups consuming tea interventional agents and the control groups which may be given only placebo or standard treatment.84 The clinical trial of different teas and their outcomes are discussed in table 2.
A double-blind placebo-controlled clinical trial study assessed green tea catechins exposed to PC patients showed a lower PSA than placebo group.85 Decrease in PSA is linked with progression of PC, may indicate possible use of green tea catechins in controlling PC. Clinical trials have reported multifarious outcomes regarding the effects of tea on PC biomarkers and progression.86 The results can vary due to different type of teas studied, the duration of intervention, and the specific endpoints assessed (PSA levels, tumor progression). In a systematic review and meta-analysis, which included 17 studies on over a million participants, green tea consumption was associated with a reduced risk of PC.87 Clinical trials on tea and PC can encounter some limitations, another concern is the studies heterogeneity involving study design and methodology that may influence the findings comparability and assessment across the trials. Variations in tea composition, dose, and bioavailability also contribute to inconsistencies in finding. Adherence to tea interventions and the accurate assessment of tea consumption through self-reported methods can also introduce bias into study outcomes. Variations in genetic susceptibility, dietary patterns, and lifestyle factors among populations can influence the observed effects of tea on PC outcomes.88
Table 2: Summary of Clinical Trials Investigating the Effects of Tea Consumption on PC Risk.
| Clinical Trials/Studies | Population | Tea Type | Duration | Outcome Measures | Results | Conclusion | Reference |
| 1 | 1000 men, age 50-70 | Green Tea | 5 yrs | Incidence of prostate cancer, PSA levels | 20% reduction in PC incidence, significant decrease in PSA levels |
Green tea may reduce PC risk |
89 |
| 2 | 500 men, age 45-65 | Black Tea | 3 yrs | PC progression, PSA levels | No significant change in PC progression, slight decrease in PSA levels |
Black tea has limited impact on PC progression |
90 |
| 3 | 800 men, age 55-75 | Green Tea | 4 yrs | PC incidence, biomarkers of inflammation | 15% reduction in PC incidence, reduced inflammation biomarkers |
Green tea shows potential in reducing PC risk |
91-92 |
| 4 | 600 men, age 50-70 | Green Tea Extract | 2 yrs | PC risk, antioxidant levels | Increased antioxidant, 10% reduction in PC risk |
Green tea extract may be beneficial for PC prevention |
93 |
| 5 | 700 men, age 50-75 | Green and Black Tea | 3 yrs | PSA levels, PC biomarkers | Moderate decrease in PSA levels, improved biomarker profiles |
Combination (green and black tea) may have synergistic effects |
94 |
| 6 | 900 men, age 60-80 | Green Tea | 6 yrs | PC incidence, quality of life measures | 25% PC reduction, improved quality of life |
Long-term green tea consumption beneficial for PC prevention |
90, 92 |
| 7 | 450 men, age 55-70 | Oolong Tea | 3 yrs | PC progression, PSA levels | No change in PC progression, stable PSA levels |
Oolong tea has neutral effect on PC progression |
95 |
| 8 | 1000 men, age 50-75 | Green Tea | 5 yrs | Incidence of high-grade PC, PSA levels | 18% PC reduction, significant decrease in PSA levels |
Green tea may specifically reduce risk of aggressive PC | 89, 92 |
| 9 | 650 men, age 55-75 | White Tea | 2 yrs | PC incidence, oxidative stress markers | Significant reduction in oxidative stress markers, 12% reduction in PC incidence |
White tea protects against PC |
14 |
| 10 | 800 men, age 50-70 | Green Tea | 4 yrs | PC incidence, DNA methylation markers |
20% reduction in PC incidence, favorable changes in DNA methylation markers |
Green tea induce epigenetic modifications stop carcinogenesis |
96-97 |
Bioavailability and Metabolism of Tea Compounds
The level of tea polyphenols which is available in the body depends on the kind of tea used, how it was prepared and the digestive process of an individual. Tea polyphenols deep tissue distribution is also affected by their metabolism that occurs after they undergo intense gastrointestinal tract and liver metabolism once ingested.98
Epidemiological studies demonstrated that antioxidant present in tea leaves, tea polyphenols are conjugated in the liver and are then excreted.99 These compounds can enhance the bioavailability when taken in presence of food matrix, the type of gut microbiota and genetic differences in enzyme-metabolizing systems. The approaches to increase bioavailability of the tea compounds are increasing the solubility by applying formulation approaches, concomitant use of bioavailability enhancers such as Piperine, and by using nano-particular formulations for cellular uptake.100
Conclusion
BACs obtained from tea (C. sinensis) are a potential source for the chemoprevention of PC. Existence of compounds as catechins (EGCG), flavonoids (quercetin, kaempferol), alkaloids (caffeine, theobromine), theanine, vitamins, minerals, and volatile oils have antioxidant, anti-inflammatory, and possibly anticancer effects. These BACs have been proved to exhibit antiproliferative, antiaromatase, and antioxidant potentials in altering the various signaling pathways associated with PC. Furthermore, the synergistic interactions among these BACs enhance their overall therapeutic potential, offering a holistic approach to reducing the risk of PC. While challenges remain in translating these findings into clinical applications, ongoing research and clinical trials continue to support the promising role of tea-derived BACs in PC chemoprevention. With the advancement in understanding molecular mechanisms of action and optimized targeted delivery and efficacy, tea BACs stand poised to contribute significantly to strategies aimed at preventing and managing PC.
Acknowledgement
The author would like to thank China Tea Science Society.
Funding Sources
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Conflict of Interest
The authors do not have any conflict of interest.
Data Availability Statement
Even though adequate data has been given in the form of tables and figures, however, all authors declare that if more data required then the data will be provided on request basis.
Ethical Statement
This research did not involve human participants, animal subjects, or any material that requires ethical approval
Informed Consent Statement
This study did not involve human participants, and therefore, informed consent was not required.
Permission to Reproduce Material from Other Sources
Not applicable
Clinical Trial Registration
This research does not involve any clinical trials.
Author Contributions
- Mingxi Li – Proposed this idea and drafted initial manuscript, Helped in preparing figures and tables and overall quality of the manuscript.
- Muhammad Farrukh Nisar – Proposed this idea and drafted initial manuscript, Helped in preparing figures and tables and overall quality of the manuscript.
- Yudi Gan – Helped in preparing figures and tables and overall quality of the manuscript.
- Chunpeng Wan – Proposed this idea and drafted initial manuscript, Helped in preparing figures and tables and overall quality of the manuscript.
References
- Hayter C. Cancer Confidential: Backstage Dramas in the Radiation Clinic: University of Toronto Press; 2022.
CrossRef - Karthaus W.R., Hofree M., Choi D., Linton E.L., Turkekul M., Bejnood A., Carver B., Gopalan A., Abida W., Laudone V. Regenerative potential of prostate luminal cells revealed by single-cell analysis. 2020;368(6490):497-505. https://doi.org/410.1126/ science. aay0267.
CrossRef - Li K., Li T., Feng Z., Huang M., Wei L., Yan Z., Long M., Hu Q., Wang J., Liu S. CD8+ T cell immunity blocks the metastasis of carcinogen-exposed breast cancer. Science Advances. 2021;7(25): eabd8936. https://doi.org/8910.1126/sciadv.abd8936.
CrossRef - Jansson F. Family History and Prognosis of Prostate Cancer, Karolinska Institutet (Sweden); 2020.
- Acharjee S., Chauhan S., Pal R., Tomar R.S. Mechanisms of DNA methylation and histone modifications. Progress in molecular biology and translational science. 2023;197:51-92. https://doi.org/10.1016/bs.pmbts.2023.1001.1001.
CrossRef - Alqahtani S., Welton K., Gius J.P., Elmegerhi S., Kato T.A. The Effect of Green and Black Tea Polyphenols on BRCA2 Deficient Chinese Hamster Cells by Synthetic Lethality through PARP Inhibition. Int J Mol Sci. Mar 14 2019;20(6):1274. https://doi.org/1210.3390 %1272Fijms20061274.
CrossRef - Bernal A., Bechler A.J., Mohan K., Rizzino A., Mathew G. The current therapeutic landscape for metastatic prostate cancer. 2024;17(3):351. https://doi.org/310.3390%3392Fph17030351.
CrossRef - De Lazzari G., Opattova A., Arena S. Novel frontiers in urogenital cancers: from molecular bases to preclinical models to tailor personalized treatments in ovarian and prostate cancer patients. Journal of Experimental & Clinical Cancer Research. 2024;43(1):146. https://doi.org/110.1186/s13046-13024-03065-13040.
CrossRef - Shahbaz M., Imran M., Hussain M., Alsagaby S.A., Momal U., Naeem H., Abdelgawad M.A., El-Ghorab A.H., Al Abdulmonem W., Waqar A.B. Curcumin: a bioactive compound with molecular targets for human malignancies. Food and Agricultural Immunology. 2023;34(1):2280524. https://doi.org/2280510.2281080/09540105.09542023.02280524.
CrossRef - Ko Y.H., Kim S.W., Kim H., Bae Y.-J. The use of 5-alpha reductase inhibitors improves the detection of prostate cancer by increasing opportunities for repeated prostate-specific antigen testing: a decade-long (2007–2016) nationwide observational study in Korea. Journal of Urologic Oncology. 2023;21(1):53-58. https://doi.org/10.1200/jco.2008.1216.9599.
CrossRef - Pan S.-Y., Nie Q., Tai H.-C., Song X.-L., Tong Y.-F., Zhang L.-J.-F., Wu X.-W., Lin Z.-H., Zhang Y.-Y., Ye D.-Y. Tea and tea drinking: China’s outstanding contributions to the mankind. Chinese Medicine. 2022;17(1):27. https://doi.org/10.1186/s13020-13022-00571-13021.
CrossRef - Samanta S. Potential bioactive components and health promotional benefits of tea (Camellia sinensis). Journal of the American Nutrition Association. 2022;41(1):65-93. https://doi.org/10.1080/07315724.07312020.01827082.
CrossRef - Björnebo L., Nordström T., Discacciati A., Palsdottir T., Aly M., Grönberg H., Eklund M., Lantz A. Association of 5α-reductase inhibitors with prostate cancer mortality. JAMA oncology. 2022;8(7):1019-1026. https://doi.org/1010.1001/jamaoncol.2022.1501.
CrossRef - Hinojosa-Nogueira D., Pérez-Burillo S., de la Cueva S.P., Rufián-Henares J.Á. Green and white teas as health-promoting foods. Food & function. 2021;12(9):3799-3819. https://doi.org/3710.1039/d3791fo00261a.
CrossRef - Zhou S., Zhang J., Ma S., Ou C., Feng X., Pan Y., Gong S., Fan F., Chen P., Chu Q. Recent advances on white tea: Manufacturing, compositions, aging characteristics and bioactivities. Trends in Food Science & Technology. 2023;134:41-55. https://doi.org/ 10.1016/j.tifs.2023.1002.1016.
CrossRef - Samynathan R., Shanmugam K., Nagarajan C., Murugasamy H., Ilango R.V.J., Shanmugam A., Venkidasamy B., Thiruvengadam M. The effect of abiotic and biotic stresses on the production of bioactive compounds in tea (Camellia sinensis (L.) O. Kuntze). Plant Gene. 2021;27:100316. https://doi.org/100310.101016/j.plgene.102021.100316.
CrossRef - Pradhan S., Dubey R. GC–MS analysis and molecular docking of bioactive compounds of Camellia sinensis and Camellia assamica. Archives of microbiology. 2021;203(5):2501-2510. https://doi.org/2510.1007/s00203-00021-02209-00206.
CrossRef - Armstrong L., do Carmo M.A.V., Wu Y., Esmerino L.A., Azevedo L., Zhang L., Granato D. Optimizing the extraction of bioactive compounds from pu-erh tea (Camellia sinensis var. assamica) and evaluation of antioxidant, cytotoxic, antimicrobial, antihemolytic, and inhibition of α-amylase and α-glucosidase activities. Food Research International. 2020;137:109430. https://doi.org/109410. 101016/j.foodres.102020.109430.
CrossRef - Cheng Y., Sun J., Zhao H., Guo H., Li J. Functional mechanism on stem cells by tea (Camellia sinensis) bioactive compounds. Food Science and Human Wellness. 2022;11(3):579-586. https://doi.org/510.1016/j.fshw.2021.1012.1014.
CrossRef - Myo H., Yaowiwat N., Pongkorpsakol P., Aonbangkhen C., Khat-Udomkiri N. Butylene glycol used as a sustainable solvent for extracting bioactive compounds from Camellia sinensis flowers with ultrasound-assisted extraction. ACS omega. 2023;8(5):4976-4987. https://doi.org/4910.1021/acsomega.4972c07481.
CrossRef - Zhao T., Li C., Wang S., Song X. Green tea (Camellia sinensis): A review of its phytochemistry, pharmacology, and toxicology. 2022;27(12):3909. https://doi.org/3910.3390/molecules27123909.
CrossRef - Wang C., Han J., Pu Y., Wang X. Tea (Camellia sinensis): a review of nutritional composition, potential applications, and Omics Research. Applied Sciences. 2022;12(12):5874. https://mdpi.com/2076-3417/5812/5812/5874#.
CrossRef - Bortolini D.G., Haminiuk C.W.I., Pedro A.C., Fernandes I.d.A.A., Maciel G.M. Processing, chemical signature and food industry applications of Camellia sinensis teas: An overview. Food Chemistry: X. 2021;12:100160. https://doi.org/100110.101016/j.fochx. 102021.100160.
CrossRef - Długaszek M., Kaszczuk M. Assessment of the nutritional value of various teas infusions in terms of the macro-and trace elements content. Journal of Trace Elements in Medicine and Biology. 2020;59:126428. https://doi.org/126410.121016/j.jtemb.122019.126428.
CrossRef - Maleš I., Pedisić S., Zorić Z., Elez-Garofulić I., Repajić M., You L., Vladimir-Knežević S., Butorac D., Dragović-Uzelac V. The medicinal and aromatic plants as ingredients in functional beverage production. Journal of Functional Foods. 2022;96:105210. https://doi.org/105210.101016/j.jff.102022.105210.
CrossRef - Sadgrove N.J., Padilla-González G.F., Leuner O., Melnikovova I., Fernandez-Cusimamani E. Pharmacology of natural volatiles and essential oils in food, therapy, and disease prophylaxis. Frontiers in Pharmacology. 2021;12:740302. https://doi.org/740310. 743389/fphar.742021.740302.
CrossRef - Kciuk M., Alam M., Ali N., Rashid S., Głowacka P., Sundaraj R., Celik I., Yahya E.B., Dubey A., Zerroug E. Epigallocatechin-3-gallate therapeutic potential in cancer: mechanism of action and clinical implications. 2023;28(13):5246. https://doi.org/5210.3390/ molecules28135246.
CrossRef - Song X., Du J., Zhao W., Guo Z. Epigallocatechin-3-gallate (EGCG): mechanisms and the combined applications. Combinatorial Chemistry & High Throughput Screening. 2017;20(10):872-885. https://doi.org/810.2174/1386207321666171218115850.
CrossRef - Hid E.J., Mosele J.I., Prince P.D., Fraga C.G., Galleano M. (−)-Epicatechin and cardiometabolic risk factors: A focus on potential mechanisms of action. Pflügers Archiv-European Journal of Physiology. 2022;474(1):99-115. https://doi.org/110.1007/s00424-00021-02640-00420.
CrossRef - Li L., He J., Zhang G., Chen H., Luo Z., Deng B., Zhou Y., Kang X. Role of caspase family in intervertebral disc degeneration and its therapeutic prospects. 2022;12(8):1074. https://doi.org/1010.3390/biom12081074.
CrossRef - Zhang X., Li J., Li Y., Liu Z., Lin Y., Huang J.-a. Anti-melanogenic effects of epigallocatechin-3-gallate (EGCG), epicatechin-3-gallate (ECG) and gallocatechin-3-gallate (GCG) via down-regulation of cAMP/CREB/MITF signaling pathway in B16F10 melanoma cells. 2020;145:104634. https://doi.org/104610.101016/j.fitote.102020.104634.
CrossRef - Renzetti A., Betts J.W., Fukumoto K., Rutherford R.N. Antibacterial green tea catechins from a molecular perspective: Mechanisms of action and structure–activity relationships. Food & function. 2020;11(11):9370-9396. https://doi.org/9310.1039/d9370fo02054k.
CrossRef - Baranwal A., Aggarwal P., Rai A., Kumar N. Pharmacological actions and underlying mechanisms of catechin: A review. Mini Reviews in Medicinal Chemistry. 2022;22(5):821-833. https://doi.org/810.2174/1389557521666210902162120.
CrossRef - Wu Y., Wang T., Xin Y., Wang G., Xu L.-A. Overexpression of GbF3′ 5′ H1 provides a potential to improve the content of epicatechin and gallocatechin. 2020;25(20):4836. https://www.mdpi.com/1420-3049/4825/4820/4836#.
CrossRef - Yang K., Zhang L., Liao P., Xiao Z., Zhang F., Sindaye D., Xin Z., Tan C., Deng J., Yin Y. Impact of gallic acid on gut health: focus on the gut microbiome, immune response, and mechanisms of action. Frontiers in immunology. 2020;11:580208. https://doi.org/580210.583389/fimmu.582020.580208.
CrossRef - Shan Z., Nisar M.F., Li M., Zhang C., Wan C. [Retracted] Theaflavin Chemistry and Its Health Benefits. Oxidative medicine and cellular longevity. 2021;2021(1):6256618. https://doi.org/6256610.6251155/6252021/6256618.
CrossRef - Zhuan W., Wujun W., Zhenzhen R., Zhilong H., Yuchun F., Jie Z., Guiyi G., Lihe J. Inhibitory Effect and Mechanism of Theaflavin (TF 1) on Hepatoma Carcinoma Bel-7402 Cells. Journal of Tea Science. 2023;43(2):287-296. https://doi.org/210.13305/ j.cnki.jts. 20230418. 20230001.
- Tanaka T., Matsuo Y. Production mechanisms of black tea polyphenols. Chemical and Pharmaceutical Bulletin. 2020;68(12):1131-1142. https://doi.org/1110.1248/cpb.c1120-00295.
CrossRef - Alizadeh S.R., Ebrahimzadeh M.A. O‐Glycoside quercetin derivatives: Biological activities, mechanisms of action, and structure–activity relationship for drug design, a review. Phytotherapy Research. 2022;36(2):778-807. https://doi.org/710.1002/ptr.7352.
CrossRef - Karimi A., Naeini F., Azar V.A., Hasanzadeh M., Ostadrahimi A., Niazkar H.R., Mobasseri M., Tutunchi H. A comprehensive systematic review of the therapeutic effects and mechanisms of action of quercetin in sepsis. 2021;86:153567. https://doi.org/ 153510.151016/ j.phymed.152021.153567.
CrossRef - Alkandahri M.Y., Pamungkas B.T., Oktoba Z., Shafirany M.Z., Sulastri L., Arfania M., Anggraeny E.N., Pratiwi A., Astuti F.D., Indriyani. Hepatoprotective effect of kaempferol: a review of the dietary sources, bioavailability, mechanisms of action, and safety. Advances in pharmacological and pharmaceutical sciences. 2023;2023(1):1387665. https://doi.org/1387610.1381155/ 1382023/1387665.
CrossRef - Sohail A.A., Ortiz F., Varghese T., Fabara S.P., Batth A.S., Sandesara D.P., Sabir A., Khurana M., Datta S., Patel U.K. The cognitive-enhancing outcomes of caffeine and L-theanine: a systematic review. 2021;13(12):e20828. https://doi.org/20810.27759/cureus.20828.
- Ren X., Chen J.-F. Caffeine and Parkinson’s disease: multiple benefits and emerging mechanisms. Frontiers in Neuroscience. 2020;14:602697. https://doi.org/602610.603389/fnins.602020.602697.
CrossRef - Imran M., Saeed F., Hussain G., Imran A., Mehmood Z., Gondal T.A., El‐Ghorab A., Ahmad I., Pezzani R., Arshad M.U. Myricetin: A comprehensive review on its biological potentials. Food science & nutrition. 2021;9(10):5854-5868. https://doi.org/5810.1002/ fsn5853.2513.
CrossRef - Xie Y., Wang Y., Xiang W., Wang Q., Cao Y. Molecular mechanisms of the action of myricetin in cancer. Mini Reviews in Medicinal Chemistry. 2020;20(2):123-133. https://doi.org/110.2174/1389557519666191018112756.
CrossRef - Gu R., Shi Y., Huang W., Lao C., Zou Z., Pan S., Huang Z. Theobromine mitigates IL-1β-induced oxidative stress, inflammatory response, and degradation of type II collagen in human chondrocytes. International Immunopharmacology. 2020;82:106226. https://doi.org/106210.101016/j.intimp.102020.106226.
CrossRef - Zhang M., Zhang H., Jia L., Zhang Y., Qin R., Xu S., Mei Y. Health benefits and mechanisms of theobromine. Journal of Functional Foods. 2024;115:106126. https://doi.org/106110.101016/j.jff.102024.106126.
CrossRef - Lu H., Tian Z., Cui Y., Liu Z., Ma X. Chlorogenic acid: A comprehensive review of the dietary sources, processing effects, bioavailability, beneficial properties, mechanisms of action, and future directions. Comprehensive reviews in food science and food safety. 2020;19(6):3130-3158. https://doi.org/3110.1111/1541-4337.12620.
CrossRef - Imani A., Maleki N., Bohlouli S., Kouhsoltani M., Sharifi S., Maleki Dizaj S. Molecular mechanisms of anticancer effect of rutin. Phytotherapy Research. 2021;35(5):2500-2513. https://doi.org/2510.1002/ptr.6977.
CrossRef - Bai J., Zhang Y., Tang C., Hou Y., Ai X., Chen X., Zhang Y., Wang X., Meng X. Gallic acid: Pharmacological activities and molecular mechanisms involved in inflammation-related diseases. Biomedicine & pharmacotherapy. 2021;133:110985. https://doi.org/ 110910.111016/j.biopha.112020.110985.
CrossRef - Pavlíková N. Caffeic acid and diseases—Mechanisms of action. International Journal of Molecular Sciences. 2022;24(1):588. https://doi.org/510.3390/ijms24010588.
CrossRef - Zaric B.L., Macvanin M.T., Isenovic E.R. Free radicals: Relationship to Human Diseases and Potential Therapeutic applications. The International Journal of Biochemistry & Cell Biology. 2023/01/01/ 2023;154:106346. https://doi.org/106310.101016/ j.biocel.102022.106346.
CrossRef - Swetha M., Keerthana C., Rayginia T.P., Anto R.J. Cancer chemoprevention: A strategic approach using phytochemicals. Frontiers in Pharmacology. 2022;12:809308. https://doi.org/809310.803389/fphar.802021.809308.
CrossRef - George B.P., Chandran R., Abrahamse H. Role of phytochemicals in cancer chemoprevention: Insights. 2021;10(9):1455. https://doi.org/1410.3390/antiox10091455.
CrossRef - Yan Z., Zhong Y., Duan Y., Chen Q., Li F. Antioxidant mechanism of tea polyphenols and its impact on health benefits. Animal Nutrition. 2020/06/01/ 2020;6(2):115-123. https://doi.org/110.1016/j.aninu.2020.1001.1001.
CrossRef - Barnabei L., Laplantine E., Mbongo W., Rieux-Laucat F., Weil R. NF-κB: At the Borders of Autoimmunity and Inflammation. Front Immunol. 2021;12:716469. https://doi.org/716410.713389/fimmu.712021.716469.
CrossRef - Guo Q., Jin Y., Chen X., Ye X., Shen X., Lin M., Zeng C., Zhou T., Zhang J. NF-κB in biology and targeted therapy: new insights and translational implications. Signal transduction and targeted therapy. 2024/03/04 2024;9(1):53. https://doi.org/10.1038/s41392-41024-01757-41399.
CrossRef - Li Z.D., Liu F., Zeng Y., Liu Y., Luo W., Yuan F., Li S., Li Q., Chen J., Fujita M., Zhang G., Li Y. EGCG suppresses PD-1 expression of T cells via inhibiting NF-κB phosphorylation and nuclear translocation. Int Immunopharmacol. May 30 2024;133:112069. https://doi.org/112010.111016/j.intimp.112024.112069.
CrossRef - Luo Q., Luo L., Zhao J., Wang Y., Luo H. Biological potential and mechanisms of Tea’s bioactive compounds: An Updated review. Journal of Advanced Research. 2023/12/05/ 2023;S2090-1232 (23):378-388. https://doi.org/310.1016/j.jare.2023.1012.1004.
- Chaudhary P., Mitra D., Das Mohapatra P.K., Oana Docea A., Mon Myo E., Janmeda P., Martorell M., Iriti M., Ibrayeva M., Sharifi-Rad J., Santini A., Romano R., Calina D., Cho W.C. Camellia sinensis: Insights on its molecular mechanisms of action towards nutraceutical, anticancer potential and other therapeutic applications. Arabian Journal of Chemistry. 2023/05/01/ 2023;16(5):104680. https://doi.org/104610.101016/j.arabjc.102023.104680.
CrossRef - Brimson J.M., Prasanth M.I., Malar D.S., Sharika R., Sivamaruthi B.S., Kesika P., Chaiyasut C., Tencomnao T., Prasansuklab A. Role of Herbal Teas in Regulating Cellular Homeostasis and Autophagy and Their Implications in Regulating Overall Health. Jun 23 2021;13(7):2162. https://doi.org/2110.3390/nu13072162.
CrossRef - Chen B.-H., Hsieh C.-H., Tsai S.-Y., Wang C.-Y., Wang C.-C. Anticancer effects of epigallocatechin-3-gallate nanoemulsion on lung cancer cells through the activation of AMP-activated protein kinase signaling pathway. Scientific Reports. 2020/03/20 2020;10(1):5163. doi: 5110.1038/s41598-41020-62136-41592.
CrossRef - Mandal R., Barrón J.C., Kostova I., Becker S., Strebhardt K. Caspase-8: The double-edged sword. Biochimica et Biophysica Acta (BBA)-Reviews on Cancer. 2020;1873(2):188357. https://doi.org/188310.181016/j.bbcan.182020.188357.
CrossRef - Shoshan-Barmatz V., Arif T., Shteinfer-Kuzmine A. Apoptotic proteins with non-apoptotic activity: expression and function in cancer. 2023;28(5):730-753. https://doi.org/710.1007/s10495-10023-01835-10493.
CrossRef - Michie J., Kearney C.J., Hawkins E.D., Silke J., Oliaro J. The immuno-modulatory effects of inhibitor of apoptosis protein antagonists in cancer immunotherapy. 2020;9(1):207. https://doi.org/210.3390/cells9010207.
CrossRef - Zhao X.-Y., Wang X.-Y., Wei Q.-Y., Xu Y.-M., Lau A.T. Potency and selectivity of SMAC/DIABLO mimetics in solid tumor therapy. 2020;9(4):1012. https://doi.org/1010.3390/cells9041012.
CrossRef - Chen D., Milacic V., Chen M.S., Wan S.B., Lam W.H., Huo C., Landis-Piwowar K.R., Cui Q.C., Wali A., Chan T.H., Dou Q.P. Tea polyphenols, their biological effects and potential molecular targets. Histol Histopathol. Apr 2008;23(4):487-496. https://doi.org/410.14670/hh-14623.14487.
- Rashid M., Zadeh L.R., Baradaran B., Molavi O., Ghesmati Z., Sabzichi M., Ramezani F. Up-down regulation of HIF-1α in cancer progression. 2021;798:145796. https://doi.org/145710.141016/j.gene.142021.145796.
CrossRef - Liu S., Xu Z.L., Sun L., Liu Y., Li C.C., Li H.M., Zhang W., Li C.J., Qin W. (‑)‑Epigallocatechin‑3‑gallate induces apoptosis in human pancreatic cancer cells via PTEN. Mol Med Rep. Jul 2016;14(1):599-605. https://doi.org/510.3892/mmr.2016.5277.
CrossRef - Kanlayavattanakul M., Khongkow M., Klinngam W., Chaikul P., Lourith N., Chueamchaitrakun P. Recent insights into catechins-rich Assam tea extract for photoaging and senescent ageing. Scientific Reports. 2024;14(1):2253. https://doi.org/2210.1038/s41598-41024-52781-41592.
CrossRef - Kim H.S., Kim M.H., Jeong M., Hwang Y.S., Lim S.H., Shin B.A., Ahn B.W., Jung Y.D. EGCG blocks tumor promoter-induced MMP-9 expression via suppression of MAPK and AP-1 activation in human gastric AGS cells. Anticancer Res. Mar-Apr 2004;24(2B):747-753. https://europepmc.org/article/MED/15161022.
- Koh Y.W., Choi E.C., Kang S.U., Hwang H.S., Lee M.H., Pyun J., Park R., Lee Y., Kim C.H. Green tea (-)-epigallocatechin-3-gallate inhibits HGF-induced progression in oral cavity cancer through suppression of HGF/c-Met. J Nutr Biochem. Nov 2011;22(11):1074-1083. https://doi.org/1010.1016/j.jnutbio.2010.1009.1005.
CrossRef - Hung S.W., Li Y., Chen X., Chu K.O., Zhao Y., Liu Y., Guo X., Man G.C., Wang C.C. Green Tea Epigallocatechin-3-Gallate Regulates Autophagy in Male and Female Reproductive Cancer. Front Pharmacol. 2022;13:906746. https://doi.org/906710.903389/ fphar.902022.906746.
CrossRef - Latronico T., Petraglia T., Sileo C., Bilancia D., Rossano R., Liuzzi G.M. Inhibition of MMP-2 and MMP-9 by Dietary Antioxidants in THP-1 Macrophages and Sera from Patients with Breast Cancer. 2024;29(8):1718. https://doi.org/1710.3390/molecules29081718.
CrossRef - Hao Q., Henning S.M., Magyar C.E., Said J., Zhong J., Rettig M.B., Vadgama J.V., Wang P. Enhanced Chemoprevention of Prostate Cancer by Combining Arctigenin with Green Tea and Quercetin in Prostate-Specific Phosphatase and Tensin Homolog Knockout Mice. 2024;14(1):105. https://doi.org/110.3390/biom14010105.
CrossRef - Khan M., Hearn K., Parry C., Rashid M., Brim H., Ashktorab H., Kwabi-Addo B. Mechanism of antitumor effects of saffron in human prostate cancer cells. 2023;16(1):114. https://doi.org/110.3390/nu16010114.
CrossRef - Marzioni D., Mazzucchelli R., Fantone S., Tossetta G. NRF2 modulation in TRAMP mice: An in vivo model of prostate cancer. Molecular Biology Reports. 2023;50(1):873-881. https://doi.org/810.1007/s11033-11022-08052-11032.
CrossRef - Mia M.A.R., Dey D., Sakib M.R., Biswas M.Y., Prottay A.A.S., Paul N., Rimti F.H., Abdullah Y., Biswas P., Iftehimul M. The efficacy of natural bioactive compounds against prostate cancer: Molecular targets and synergistic activities. Phytotherapy Research. 2023;37(12):5724-5754. https://doi.org/5710.1002/ptr.8017.
CrossRef - Xie X., Li Y., Lian S., Lu Y., Jia L. Cancer metastasis chemoprevention prevents circulating tumour cells from germination. Signal transduction and targeted therapy. 2022/10/02 2022;7(1):341. https://doi.org/310.1038/s41392-41022-01174-w.
CrossRef - Penfold L., Woods A., Pollard A.E., Arizanova J., Pascual-Navarro E., Muckett P.J., Dore M.H., Montoya A., Whilding C., Fets L., Mokochinski J., Constantin T.A., Varela-Carver A., Leach D.A., Bevan C.L., Nikitin A.Y., Hall Z., Carling D. AMPK activation protects against prostate cancer by inducing a catabolic cellular state. Cell Rep. Apr 25 2023;42(4):112396. https://doi.org/112310.111016/j.celrep.112023.112396.
CrossRef - Siddiqui I.A., Adhami V.M., Afaq F., Ahmad N., Mukhtar H. Modulation of phosphatidylinositol-3-kinase/protein kinase B- and mitogen-activated protein kinase-pathways by tea polyphenols in human prostate cancer cells. J Cell Biochem. Feb 1 2004;91(2):232-242. https://doi.org/210.1002/jcb.10737.
CrossRef - Lee Y.-H., Kwak J., Choi H.-K., Choi K.-C., Kim S., Lee J., Jun W., Park H.-J., Yoon H.-G. EGCG suppresses prostate cancer cell growth modulating acetylation of androgen receptor by anti-histone acetyltransferase activity. Int J Mol Med. 2012/07/01 2012;30(1):69-74. https://doi.org/10.3892/ijmm.2012.3966.
- Penfold L., Woods A., Pollard A.E., Arizanova J., Pascual-Navarro E., Muckett P.J., Dore M.H., Montoya A., Whilding C., Fets L., Mokochinski J., Constantin T.A., Varela-Carver A., Leach D.A., Bevan C.L., Nikitin A.Y., Hall Z., Carling D. AMPK activation protects against prostate cancer by inducing a catabolic cellular state. Cell Reports. 2023/04/25/ 2023;42(4):112396. https://doi.org/112310.111016/j.celrep.112023.112396.
CrossRef - Huang Y., Chen Q., Liu Y., Tian R., Yin X., Hao Y., Yang Y., Yang J., Li Z., Yu S. Association between tea consumption and colorectal cancer: a systematic review and meta-analysis of a population-based study. BMC gastroenterology. 2023;23(1):294. https://doi.org/210.1186/s12876-12023-02928-12878.
CrossRef - Micali S., Territo A., Pirola G.M., Ferrari N., Sighinolfi M.C., Martorana E., Navarra M., Bianchi G. Effect of green tea catechins in patients with high-grade prostatic intraepithelial neoplasia: Results of a short-term double-blind placebo controlled phase II clinical trial. ARCHIVIO ITALIANO DI UROLOGIA ANDROLOGIA. 2017;89(3):197-202. https://doi.org/110.4081/aiua.2017.4083.4197.
CrossRef - Kumar N.B., Dickinson S.I., Schell M.J., Manley B.J., Poch M.A., Pow-Sang J. Green tea extract for prevention of prostate cancer progression in patients on active surveillance. 2018;9(102):37798. https://doi.org/37710.18632/oncotarget.26519.
CrossRef - Habs M., Binder K., Krauss S., Müller K., Ernst B., Valentini L., Koller M. A balanced risk–benefit analysis to determine human risks associated with pyrrolizidine alkaloids (PA)—The case of tea and herbal infusions. 2017;9(7):717. https://doi.org/710.3390/nu9070717.
CrossRef - Ho E., Beaver L.M., Williams D.E., Dashwood R.H. Dietary factors and epigenetic regulation for prostate cancer prevention. Advances in nutrition. 2011;2(6):497-510. https://doi.org/410.3945/an.3111.001032.
CrossRef - Bosland M.C., Horton L., Condon M.S. Effects of green tea on prostate carcinogenesis in rat models and a human prostate cancer xenograft model. The Prostate. 2022;82(11):1117-1124. https://doi.org/1110.1002/pros.24364.
CrossRef - Thomas R., Greef B., McConnachie A., Stanley B., Williams M. Dietary consumption of tea and the risk of prostate cancer in the Prostate, Lung, Colorectal and Ovarian Cancer Screening Trial. British Journal of Nutrition. 2022;128(4):653-658. https://doi.org/610.1017/s0007114521003664.
CrossRef - Henning S.M., Wang P., Lee R.-P., Trang A., Husari G., Yang J., Grojean E.M., Ly A., Hsu M., Heber D. Prospective randomized trial evaluating blood and prostate tissue concentrations of green tea polyphenols and quercetin in men with prostate cancer. Food & function. 2020;11(5):4114-4122. https://doi.org/4110.1039/d4110fo00565g.
CrossRef - Kumar N.B., Hogue S., Pow-Sang J., Poch M., Manley B.J., Li R., Dhillon J., Yu A., Byrd D.A. Effects of green tea catechins on prostate cancer chemoprevention: the role of the gut microbiome. 2022;14(16):3988. https://doi.org/3910.3390/cancers14163988.
CrossRef - Sharifi-Zahabi E., Hajizadeh-Sharafabad F., Abdollahzad H., Dehnad A., Shidfar F. The effect of green tea on prostate specific antigen (PSA): A systematic review and meta-analysis of randomized controlled trials. Complementary therapies in medicine. 2021;57:102659. https://doi.org/102610.101016/j.ctim.102020.102659.
CrossRef - Uduwana S., Abeynayake N., Wickramasinghe I. Synergistic, antagonistic, and additive effects on the resultant antioxidant activity in infusions of green tea with bee honey and Citrus limonum extract as additives. Journal of Agriculture and Food Research. 2023;12:100571. https://doi.org/100510.101016/j.jafr.102023.100571.
CrossRef - Lin Y.-H., Wang C.-C., Lin Y.-H., Chen B.-H. Preparation of catechin nanoemulsion from oolong tea leaf waste and its inhibition of prostate cancer cells DU-145 and tumors in mice. 2021;26(11):3260. https://doi.org/3210.3390/molecules26113260.
CrossRef - Bouyahya A., Mechchate H., Oumeslakht L., Zeouk I., Aboulaghras S., Balahbib A., Zengin G., Kamal M.A., Gallo M., Montesano D. The role of epigenetic modifications in human cancers and the use of natural compounds as epidrugs: Mechanistic pathways and pharmacodynamic actions. 2022;12(3):367. https://doi.org/310.3390/biom12030367.
CrossRef - Mondal P., Natesh J., Penta D., Meeran S.M. Progress and promises of epigenetic drugs and epigenetic diets in cancer prevention and therapy: A clinical update. Paper presented at: Seminars in Cancer Biology2022.
CrossRef - Clifford M.N., van der Hooft J.J., Crozier A. Human studies on the absorption, distribution, metabolism, and excretion of tea polyphenols. The American journal of clinical nutrition. 2013;98(6):1619S-1630S. https://doi.org/1610.3945/ajcn.1113.058958.
CrossRef - Higdon J.V., Frei B. Tea catechins and polyphenols: health effects, metabolism, and antioxidant functions. Crit Rev Food Sci Nutr. 2003;43(1):89-143. https://doi.org/110.1080/10408690390826464.
CrossRef - Kumar M., Kumar D., Kumar S., Kumar A., Mandal U.K. A recent review on bio-availability enhancement of poorly water-soluble drugs by using bioenhancer and nanoparticulate drug delivery system. Current Pharmaceutical Design. 2022;28(39):3212-3224. https://doi.org/3210.2174/1381612829666221021152354.
CrossRef
Abbreviations
AMPK – AMP-Activated Protein Kinase
Apaf-1 – Apoptotic Protease Activating Factor-1
BACs – Bioactive compounds
DISC – Death-Inducing Signaling Complex
DNA – Deoxyribonucleic acid
EC – Epicatechin
ECG – EpicatechinGallate
ECM – Extracellular Matrix
EGC – Epigallocatechin
EGCG – EpigallocatechinGallate
MMPs – Matrix Metalloproteinases
PC – Prostate Cancer
ROS – Reactive Oxygen Species
SOD – Superoxide Dismutase
VEGF – Vascular Endothelial Growth Factor

This work is licensed under a Creative Commons Attribution 4.0 International License.





