Introduction
Diabetes mellitus (DM) is a heterogeneous metabolic disorder indicated by chronic hyperglycemia in postprandial and fasting states due to the impairment of insulin production by pancreas β cells, insulin action at the selected tissues or both.1 On a global scale, diabetes has reached epidemic proportions and is one of the significant contributors to the global economic burden of non-communicable disease. According to the International Diabetes Federation, the overall number of adult with diabetes worldwide in 2017 was approximately 425 million, and the number is estimated to be 629 million in 2045.2 A recent report on the global economic burden of diabetes in adults aged 20–79 stated that the cost of diabetes globally was US$1.31 trillion in 2015, thus making it as one of the most expensive diseases to manage.3 Due to the considerable cost associated with diabetes management, diabetes patients opted for complementary and alternative medicine (CAM) therapies to manage the disease. According to Dayeef, Karyono, Sujuti,4 the prevalence of CAM namely medicinal plants in both developed and developing countries is attributable to its affordability, accessibility, and efficacy.
Annona muricata (Annonaceae) is one of the medicinal plants that has been widely studied for its therapeutic values. This widespread tree is local to tropical America but currently is planted worldwide. It is distributed for the most part in tropical and sub-tropical regions, including South and North America, Australia, India, Nigeria and Malaysia. A. muricata is known by different local names, for example soursop (English), graviola (Portuguese), Sirsak (Indonesia) and guanabana (Latin America).5 Review of the literature indicated that all parts of A. muricata possess therapeutic values against numerous illnesses. The fruit has been reported to possess anti-depressive6 and anti-cancer 7,8 effects. Leaves are effective against fever,9 headache, sleep disorder, rheumatism,10,11 viral infection,12 breast cancer13 and bacterial infection.14 The stem bark may have adaptogenic15 and antioxidant16 potentials. In addition, the plant contains various phytoconstituents including alkaloids,17 annonaceous acetogenins,18 cyclopeptides,19 megastigmanes,20 flavonol triglycosides,21 and phenolics.22 Despite numerous studies of this plant and its properties, to date, no comprehensive evidence-based review has been conducted to establish the potential antidiabetic effect of A. muricata. Hence, this review aimed to summarize current and comprehensive studies of the efficacy of A. muricata as an antidiabetic agent.
Materials and Methods
Search strategy
Six online databases (PubMed, ScienceDirect, Web of Science, Scopus, Google Scholar and SpringerLink) were used to conduct a comprehensive search of articles published until December 2018 with no end date limit. The following keywords were chosen based on MeSH terms; Annona/ soursop in combination with diabetes, glucose, and insulin.
Study selection and inclusion/ exclusion criteria
The title and abstract of each article were assessed and the duplicated articles were removed. The remaining articles were further assessed to determine their compatibility with the inclusion criterion, which were studies of the antidiabetic activity of A. muricata. Review articles, conference proceedings, commentaries, and abstracts in symposiums and congresses were excluded. Additionally, a manual search was carried out from the reference list of included articles for additional data or forward citations. All remaining titles, abstracts, and full articles were independently reviewed by two collaborators (IAA and NAY).
Data extraction
The information extracted from the articles included the year of publication, country, study models (in vivo, in vitro, clinical, and in silico), plant part used, extraction solvent and parameters measured (level of blood glucose, insulin, bodyweight, antioxidants, enzymes, lipid profile and histological study of pancreas and liver).
Results and Discussion
Literature search
The literature search yielded a total of 167 records (from the year 2002 to April 2018). The following are the numbers of articles extracted from the respective databases: Google Scholar (n = 68), ScienceDirect (n = 35), PubMed (n = 25), Web of Science (n = 19), Scopus (n = 10), and SpringerLink (n=10). No relevant study was obtained through a manual search of the references of included papers or from any other data sources. After removing the duplicate articles, 132 articles remained, of which 91 were excluded based on the review of title and abstract. Forty-one articles were further assessed by reading the full text, and 24 of them were excluded. Ultimately, 17 articles met the inclusion criterion (Figure 1). Those articles were classified into five groups based on the study models; clinical (n = 1), in vivo (n = 10), in vitro (n = 4), in vivo and in vitro (n = 1), and in silico (n = 1).
Most of the studies were conducted by researchers from Nigeria (8 studies), followed by Indonesia (4 studies), then Brazil (2 studies), and Cameroon, Peru and India (1 study each). The number of studies per region is related to the geographical distribution of A. muricata as local researchers tend to focus on native plants with potential ethnomedicinal uses.5 The application of traditional and complementary medicine to manage diabetes in these regions also prompted research activity focused on medicinal plants. The highest number of articles were published in 2015 and 2017 (4 studies for each year), followed by 2008 (3 studies) and 2010 and 2013 (2 studies in for each year).
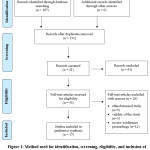 |
Figure 1: Method used for identification, screening, eligibility, and inclusion of the articles Click here to View figure |
Clinical study
Based on the criteria, one clinical study was identified. Arroyo, Jaime, Ronceros Gerardo, Aníbal 23 evaluated the adjuvant effect of A. muricata in patients with type 2 diabetes mellitus. Three groups (n = 15) of patients were given 1, 2, and 3 capsules of ethanol extract of A. muricata leaves (180 mg) respectively, along with 5 mg of glibenclamide for a month. Three other groups were given glibenclamide only. A greater decrease in blood glucose level was observed in those who received A. muricata extract and glibenclamide, with 11% of them (5 patients) experienced side effects such as burning pain in epigastrium (two patients) and nausea (three patients).
In vivo studies
The in vivo studies of A. muricata revealed that various parts of the plant showed potential antidiabetic effects when tested using rodent models (Table 1). Most of these studies tested the antidiabetic effect of leaf extracts (64%), whereas the other 27% focused on the seed extracts and only 9% examined the bark extract. Water and methanol were the most frequently used solvents used to extract bioactive components of A. muricata (36% each), followed by ethanol (18%) and a mixture of methanol/ chloroform (10%). The studies show that polar solvents are preferable probably because they provide higher recovery yield of phenolic compounds than non-polar solvents.24-26 Phenolic compounds from medicinal plants have been associated with various pharmacological activities of the medicinal plants, including anti-aging, antioxidant, anti-inflammatory, and anti-hyperglycemia.27-29 The most common animal model used to study the antidiabetic activity of this plant was the streptozotocin (STZ)-induced diabetic rat models (64%). Other models used were alloxan-induced diabetic (9%), clozapine-induced diabetic (9%) and glucose preloaded models (9%). Ivorra, Paya, Villar30 reported that the STZ-induced diabetic rat model is widely used because it is considered to be a good model for the preliminary screening of bioactive agents against diabetes.
Leaf extracts of A. muricata have been shown to have positive effects on the metabolic parameters of diabetes. For example, the leaf extracts in various doses were found to significantly decrease blood glucose levels, increase bodyweight, improve the serum lipid profile by decreasing total cholesterol (TC), triglycerides (TGs), low density lipoprotein-cholesterol (LDL-C), and very low density lipoprotein-cholesterol (VLDL-C), and increase the level of high density lipoprotein-cholesterol (HDL-C) and the percentage of anti-atherogenic index (AAI). They also altered the activity of the antioxidant enzymes such as glutathione, superoxide dismutase (SOD), and catalase.31-33 Adeyemi, Komolafe, Adewole, Obuotor, Abiodun, Adenowo34 and Adewole, Caxton-Martins35 suggested that leaf extract of A. muricata might contain bioactive compounds that exert anti-hyperglycemic effect by stimulating and enhancing the secretion and action of insulin. This premise was supported by Adewole, Ojewole36 who reported that leaf extracts of A. muricata could prevent degeneration of β-cells and to regenerate and proliferate the pancreatic β-cells destructed by STZ, thus replacing damaged cells and increasing the area of insulin immunoreactive β-cells and insulin production.
The seeds of A. muricata have also been reported to possess promising antidiabetic effects. Pinto, Cerqueira-Lima, dos Santos Suzarth, de Souza, Tosta, da Silva, de Oliveira Pires, de Almeida Queiroz, Teixeira, Dourado37 found that diabetic mice treated with the extract of seed oil had lower glucose levels, higher insulin concentrations and a well-preserved pancreatic islet area, compared to untreated diabetic rats. Additionally, the histology of the liver tissues suggested the presence of a hepatoprotective effect, as the A. muricata-treated group had the normal architecture of hepatocytes with partial recovery of glycogen storage compared to the STZ-induced group. In addition to that, Hasanah, Sundhani, Nurulita38 found that 400 mg/kg of ethanol seed extract to be effective in reducing blood glucose of glucose preloaded rats. In another study, a significant effect on the blood glucose level was observed at higher doses of the methanolic seed extract (600 and 800mg/kg) in clozapine-induced diabetic rats.39 The observed hypoglycemic effect might be due to the secondary metabolites presented in A. muricata seed extracts like alkaloids, flavonoids, triterpenoids and tannins.40,41 As for the bark extract, Ahalya, Shankar, Kiranmayi42 demonstrated that 300 mg/kg of the ethanolic bark extract significantly reduced blood glucose level of alloxan-induced diabetic rats. The same extract also resulted in a significant improvement in the rat lipid profile by lowering TG, LDL, and TC and increasing HDL.
Table 1: Effect of A. muricata L. on metabolic parameters determined through in vivo studies
| Plant part-extract used | Dose-route of administration | Test Model | Duration of study | Findings | Reference |
| Leaves-methanol extract | 100mg/kg – i.p. | STZ-induced diabetic rats | 10 weeks | ↑ Bodyweight, | (33) |
| ↓ glucose level | |||||
| Leaves-methanol extract | 25, 50, 100, 200, 400mg/kg – i.p. | STZ-induced diabetic rats | 10 weeks | ↓ TC, TG, LDL, VLDL | (31) |
| ↑ HDL and AAI | |||||
| Leaves-aqueous extract | 100, 200mg/kg – p.o. | STZ-induced diabetic rats | 5 hours | Both doses: | (32) |
| ↓ glucose level | |||||
| 4 weeks | Both doses: ↓ glucose level, LDL, creatinine, AST, ALT, MDA, nitrites, restored TG, TC, SOD, CAT content | ||||
| Seeds-methanol extract | 600, 800 mg/kg – p.o. | Clozapine- induce diabetic rats | 10 days | ↓ Glucose level | (39) |
| Seeds- ethanol extract | 100, 200, 400 mg/kg – p.o. | Glucose preloaded rat | 120 minutes | ↓ Glucose level | (38) |
| Bark-ethanol extract | 150, 300 mg/kg – p.o. | Alloxan-induced diabetic rats | 6 hours | ↓ Glucose level | (42) |
| 14 days | ↑ Bodyweight, | ||||
| ↓ glucose level | |||||
| ↓ TC, TG, LDL, VLDL | |||||
| ↑ HDL | |||||
| Leaves-methanol extract | 100 mg/kg – i.p. | STZ-induced diabetic rats | 10 weeks | ↑ Bodyweight, | (34) |
| ↓ glucose level, regenerate pancreatic β-cells | |||||
| Leaves- aqueous extract | 100 mg/kg – p.o. | STZ-induced diabetic rats | 60 days | No change in bodyweight | (36) |
| ↓ glucose, ROS, MDA, TC, TG, LDL | |||||
| ↑ insulin level and antioxidant enzymes, enhance pancreas protective effect | |||||
| ↑ insulin level and antioxidant enzymes, enhance pancreas protective effect | |||||
| Leaves- aqueous extract | 100 mg/kg orally – p.o. | STZ-induced diabetic rat | 8 hours | Normalize bodyweight, ↓ glucose level, ↑ insulin, ↑ antioxidant enzymes, protect pancreatic β cells | (35) |
| Leaves- aqueous extract | 100 mg/kg orally – p.o. | STZ-induced diabetic rats | 60 days | No change in body and liver weight, ↓ glucose level, TC, TG, LDL, normalize hexokinase and glucokinase activities, ↑ antioxidant enzymes | (43) |
| Seed oil | 1.0 mL/kg – p.o. | STZ induced T1D BALB/c mice | 48 days | ↓ Glucose level, ↑ insulin levels, preserve pancreatic islets area, hepatoprotective effect | (37) |
i.p. (intraperitoneal), p.o. (per oral), STZ (streptozotocin), TC (total cholesterol), TG (triglyceride), LDL (low density lipoprotein), VLDL (very low density lipoprotein), HDL (high density lipoprotein), AAI (advanced atherogenic index), AST (aspartate aminotransferase), ALT (alanine aminotransferase), ROS (reactive oxygen species), SOD (superoxide dismutase), MDA (malondialdehyde), CAT (catalase).
In vitro studies
All of the in vitro studies of A. muricata demonstrated a significant effect of different parts of the plant on inhibition of -amylase and -glucosidase activities and on strengthening antioxidant capacities (Table 2). -amylase is an enzyme that catalyzes the hydrolysis of polysaccharide (e.g., starch) into oligosaccharides (e.g., sucrose, maltose and lactose). These oligosaccharides are further degraded into absorbable monosaccharides (e.g., glucose, fructose, and galactose) by -glucosidase in the intestine.43 Therefore, inhibition of these enzymes will eventually lead to suppression of postprandial hyperglycemia as the amount of glucose being absorbed into the blood circulation is delayed.44 Oxidative stress plays a major role in the development of diabetes complications both in the microvascular and macrovascular systems. Oxidative stress in diabetics occurs primarily due to excessive oxygen free radical production from auto-oxidation of glucose, glycated protein, and glycation of antioxidative enzymes.45 Oxygen free radicals are responsible for oxidative damage to macromolecules such as proteins, lipids, and nucleic acids in diabetic patients.46 Most of these side effects can be prevented by the presence of antioxidants.47
A study conducted by Berawi, Shidarti, Nurdin, Lipoeto, Wahid 48 on the leaf of A. muricata demonstrated a potential inhibitory activity of the methanol extract against -amylase and -glucosidase while the water extract showed high total phenolic content. In another study, Justino, Miranda, Franco, Martins, da Silva, Espindola 49 reported that, the ethyl acetate and n-butanol fractions of the ethanolic extract of A. muricata leaves had potent antioxidant capacity, suppressed the activities of α-amylase, α-glucosidase, and pancreatic lipase, inhibited formation of advanced glycation end-products and reduced lipid peroxidation in the liver as compared to the other fractions. Lipid peroxidation is known to be induced in type 2 diabetes mellitus due to hyperglycemia-induced free radicals production.50 Thus, Justino et al.’s findings suggested a promising use of the A. muricata leaf, particularly its polyphenol-enriched fractions, as a therapy for treating diabetes. Same finding was reported by Hardoko, Wijoyo, Halim 51 on the aqueous extract of leaf in terms of suppressing α-glucosidase activity. The pericarp, pulp and seeds of A. muricata have also been reported to possess promising enzyme inhibitory effects. Aqueous extract of these parts inhibited the activity of α glucosidase and α amylase in a concentration dependent manner.52
Table 2: Effect of A. muricata L. on metabolic parameters determined through in vitro studies
| Plant part | Dose/ concentration | Test Model | Extract/ fraction | Effect | Reference |
| Leaves | 0.3, 0.6, 0.9, 1.2, 1.5 mg/ml | α-amylase assay | Methanol extract | Inhibited α-amylase activity | (49) |
| α-glucosidase assay | Methanol extract | Inhibited α-glucosidase activity | |||
| Total phenol content | Aqueous extract | High total phenol content | |||
| Leaves | 20, 40, 60, 80 µg/ml | α-amylase assay | Ethanol extract and n-butanol fraction | Inhibited α-amylase activity | (50) |
| 500, 1000, 1500 µg/ml500, 1500, 2000 µg/ml | α-glucosidase assayPancreatic lipase inhibition | Inhibited α-glucosidase activity | |||
| Inhibited pancreatic lipase activity | |||||
| 0.15, 1.5 and 15 µg/ml | Lipid peroxidation | Reduced liver lipid peroxidation | |||
| Pericarp, pulp and seed | 0.2, 0.4, 0.6, 0.8 mg/ml | α-amylase assay | Aqueous extract | Concentration-dependent inhibition | (53) |
| α-glucosidase assay | Concentration-dependent inhibition | ||||
| Leaves | 6.25, 12.5, 22 and 50 ppm | α-glucosidase assay | Aqueous extract | Concentration-dependent inhibition | (52) |
mg/ml (milligram per milliliter), µg mL-1 (microgram per milliliter), IC50 (concentration required to reduce the rate of reaction by 50%), DPPH (2,2-diphenyl-1-picrylhydrazyl), ORAC (oxygen radical absorbance capacity), FRAP (ferric reducing antioxidant power).
In silico study
One computational study has been carried out to study the effect of A. muricata on metabolic parameters related to diabetes condition.53 In that study, the effect of selected A. muricata active compounds (rutin, muricatocin A, anonaine, isolaureline, xylopine, and kaempferol 3-O-rutinoside) on inhibition of the Forkhead Box O1 (FOXO1) protein was evaluated. The FOXO1 protein plays a crucial role in the proliferation process of pancreatic β-cells and inhibition of the FOXO1 protein activity in the nucleus will enhance insulin sensitivity, as previously reported.54,55 The study revealed that active compounds in A. muricata leaves had similar or less free binding energy than the control. Muricatocin A and rutin were found to have a same binding effect against 66% of amino acid residues, whereas the other four active compounds had the equal binding ability of 33% of amino acid residues as compared to the control with hydrogen bonds.
Possible mechanism underlying the antidiabetic action of A. muricata L.
Review of the literature indicated that the observed blood-glucose-lowering effect of A. muricata might be due to the following several possible mechanisms of action:
Pancreatic β-cell protection/ regeneration
Pancreatic β-cells are responsible for the restoration and secretion of insulin, a hormone that is important for the metabolism of carbohydrate, fat, and protein. Reduced β-cell mass and impaired insulin secretion are responsible for the progression of hyperglycemic conditions to diabetes; therefore, preservation of β-cells is an important therapeutic goal to prevent diabetes development and progression. Several studies have reported the pancreas protective effects of A. muricata which include regeneration of pancreatic β-cells after STZ destruction,34 preservation of pancreatic islet area,37 proliferation or renewal of β-cells,32 and inhibition of FOXO1 protein.53 These pancreas protective effects resulted in significant improvement in insulin secretion and action33,34,39 that eventually lead to the normalization of the blood glucose level in diabetic rat models.
Hepatoprotective Effect
The liver plays a vital role in the glucose homeostasis by controlling numerous pathways of glucose metabolism, namely gluconeogenesis, glycogenesis and glycolysis. Thus, the hepatoprotective effect of A. muricata could bring a significant contribution to its observed antidiabetic effect. Histology of the liver tissues showed a typical architecture of hepatocytes in the A. muricata treated group, with partial recovery of glycogen storage, compared to the STZ-induced group.32 Justino, Miranda, Franco, Martins, da Silva, Espindola49 also demonstrated the effect of this plant in reducing liver lipid peroxidation.
Antioxidant Properties
It has been shown that in the diabetic states, antioxidant defenses are altered due to the presence of chronic hyperglycemia, which results in reduced antioxidant levels, increased free radical regeneration, and increased oxidative stress.56 Oxidative stress plays a significant role in the development of diabetes complications both in the microvascular and macrovascular systems. Oxidative stress in people with diabetes occurs primarily due to excessive oxygen free radical production from auto-oxidation of glucose, glycated protein, and glycation of antioxidative enzymes.45 Oxygen-free radicals are responsible for oxidative damage to macromolecules such as proteins, lipids, and nucleic acids in diabetic patients.46 Most of these side effects can be prevented by the presence of antioxidants.47 Hence, plants with antioxidant ingredients have the potential to fight against oxidative stress and contribute to the amelioration of hyperglycemia condition. In vivo studies using diabetic rat model have shown that administration of A. muricata extracts can lower blood glucose level and simultaneously restored antioxidant enzymes namely superoxide dismutase (SOD), catalase and nitrite oxide and also reduced lipid peroxidation by suppressing malondialdehyde and transaminase activities.32,35,36,57 In accordance with in vivo findings, in vitro studies using DPPH, FRAP and ORAC assays demonstrated the ability of A. muricata’s extracts to scavenge free radicals.49 Furthermore, phytochemical analysis of A. muricata revealed the presence of total phenolic compounds such as tannins and flavonoids that are known for their antioxidant properties.48,58 These findings suggested the protective role of this plant’s extracts against oxidative stress and associated their activities with the observed anti-hyperglycemic effect.
Intestinal α-glucosidase and α-amylase inhibitory effect
All the in vitro studies of A. muricata showed inhibitory activities against the enzymes α-amylase and α-glucosidase. -amylase is an enzyme that catalyzes the hydrolysis of polysaccharide (e.g., starch) into oligosaccharides (e.g., sucrose, maltose and lactose). These oligosaccharides are further degraded into absorbable monosaccharides (e.g., glucose, fructose, and galactose) by -glucosidase in the intestine.43 Therefore, inhibition of these enzymes will eventually lead to suppression of postprandial hyperglycemia as the amount of glucose being absorbed into the blood circulation is delayed.44 These findings might explain the low blood glucose level after A. muricata ingestion.
Conclusion
A. muricata L. is a coveted tropical tree that has been widely used in traditional medicine to treat different medical conditions. A literature review indicates A. muricata to be a potential therapeutic agent for the management of diabetes mellitus and its complications. It promotes better glycemic control, normalizes lipid parameters, stimulates insulin secretion and action and improves the morphology of pancreas and liver. Though past studies seem to provide sound pharmacological confirmation of the folkloric usage of this plant, they suffered from several limitations. We conclude with the critical caveat that; many questions still need to be addressed regarding A. muricata as a safe antidiabetic agent. Firstly, further investigations are required to identify and characterize the active principle ingredients responsible for its antidiabetic effect, establish its safety profile, and determine the therapeutic index of all active ingredients. The conclusion must be made as to whether the antidiabetic effect is due to a single compound or the synergistic effect of multiple compounds. Secondly, studies on the mechanism of action at the cellular and molecular levels should be conducted because available data does not fully explain the observed effect. By knowing the target pathway, specific effect of the extract or its active principle could be monitored, thus, enabling dosage adjustment. Metabolic studies are also needed to determine the effect of the hepatic first-pass and digestive enzymes on the bioactivity of the extract or its active principle. Thirdly, there is a lack in a clinical validation as most of the preclinical data retrieved has not been clinically validated. Despite a body of preclinical evidence suggesting its potential use for managing diabetes, it is necessary to conduct well-designed clinical trials that are randomized and systematic to validate the efficacy and safety of this plant further. Also, trials should test and compare whether the extract/ active compound of A. muricata is useful as a single therapy or, at best, as an adjuvant therapy with concurrent oral hypoglycemic agents. Considering that A. muricata is widely consumed worldwide, further exploration of its potential is worthwhile.
Author Disclosure Statement
The authors declared there is no potential conflict of interest concerning the research, authorship, and publication of these articles.
Acknowledgments
This work was supported by the short-term research grant (304/CIPPT/6315187) from Universiti Sains Malaysia.
References
- American Diabetes Association. 2. Classification and diagnosis of diabetes. Diabetes care. 2017;40(Supplement 1):S11-S24.
- IDF Diabetes Atlas, 8th edition 2017. International Diabetes Federation website http://www.diabetesatlas.org. Accessed March 20, 2018.
- Zhang P., Gregg E. Global economic burden of diabetes and its implications. Lancet Diabetes Endocrinol. 2017;5(6):404-405.
- Dayeef A.Y.M., Karyono S., Sujuti H. The influence of Annona muricata leaves extract in damaging kidney cell and inducing caspase-9 activity. J Pharm Biol Sci. 2013;8(5):48-52.
- Coria-Tellez A.V., Montalvo-Gónzalez E., Yahia E.M., Obledo-Vázquez E.N. Annona muricata: A comprehensive review on its traditional medicinal uses, phytochemicals, pharmacological activities, mechanisms of action and toxicity. Arab J Chem. 2016;11(5):662-691.
- Hasrat J., De Bruyne T., De Backer J., Vauquelin G., Vlietinck A. Isoquinoline derivatives isolated from the fruit of Annona muricata as 5‐HTergic 5‐HT1A receptor agonists in rats: unexploited antidepressive (lead) products. J Pharm Pharmacol. 1997;49(11):1145-1149.
- Sun S., Liu J., Zhou N., Zhu W., Dou Q.P., Zhou K. Isolation of three new annonaceous acetogenins from Graviola fruit (Annona muricata) and their anti-proliferation on human prostate cancer cell PC-3. Bioorg Med Chem Lett. 2016;26(17):4382-4385.
- Sun S., Liu J., Sun X., Zhu W., Yang F., Felczak L., Dou Q. P., Zhou K.. Novel Annonaceous acetogenins from Graviola (Annona muricata) fruits with strong anti-proliferative activity. Tetrahedron Lett. 2017;58(19):1895-1899.
- Ong H., Norzalina J. Malay herbal medicine in gemencheh, Negri Sembilan, Malaysia. 1999;70(1):10-14.
- Mishra S., Ahmad S., Kumar N., Sharma B. Annona muricata (the cancer killer): a review. Glob J Pharma Res. 2013;2(1):1613-1618.
- De Sousa O.V., Vieira G.D., De Pinho J.J.R., Yamamoto C.H., Alves M.S. Antinociceptive and anti-inflammatory activities of the ethanol extract of Annona muricata leaves in animal models. Int J Mol Sci. 2010;11(5):2067-2078.
- Gajalakshmi S., Vijayalakshmi S., Devi Rajeswari V.. Phytochemical and pharmacological properties of Annona muricata: a review. Int J Pharm Pharm Sci. 2012;4(2):3-6.
- Endrini S., Suherman S., Widowati W.. Annona muricata leaves have strongest cytotoxic activity against breast cancer cells. Univ Med. 2015;33(3):179-184.
- Chithra K., Shaji C., Thomas B. Evaluation of major phytochemical constituents of two edible fruit yielding species of Annonaceae: Annona muricata and Annona reticulata L. J Med Plants Stud. 2016;4(4):198-202.
- Padma P., Chansauria J., Khosa R., Ray A. Effect of Annona muricata and Polyalthia cerasoides on brain neurotransimitters and enzyme monoamine oxidase following cold immobilization stress. J Nat Remedies. 2001;1(2):144-146.
- Omale J., Olakunle S. In vitro and in vivo evaluation of antioxidant activity of Annona muricata stem bark extracts in Rattus norvegicus. Planta Med. 2015;81(16).
- Yang C., Gundala S.R., Mukkavilli R., Vangala S., Reid M.D., Aneja R. Synergistic interactions among flavonoids and acetogenins in Graviola (Annona muricata) leaves confer protection against prostate cancer. 2015;36(6):656-665.
- Rupprecht J.K., Hui Y.H., McLaughlin J.L. Annonaceous acetogenins: a review. J Nat Prod. 1990;53(2):237-278.
- Kossouoh C., Moudachirou M., Adjakidje V., Chalchat J.C., Figuérédo G. Essential oil chemical composition of Annona muricata leaves from Benin. J Essent Oil Res. 2007;19(4):307-309.
- Matsushige A., Matsunami K., Kotake Y., Otsuka H., Ohta S. Three new megastigmanes from the leaves of Annona muricata. J Nat Med. 2012;66(2):284-291.
- Nawwar M., Ayoub N., Hussein S., Hashim A., El-Sharawy R., Wende K., Harms M., Lindequist U. Flavonol triglycoside and investigation of the antioxidant and cell stimulating activities of Annona muricata Arch Pharm Res. 2012;35(5):761-767.
- Jimenez V.M., Gruschwitz M., Schweiggert R.M., Carle R., Esquivel P. Identification of phenolic compounds in soursop (Annona muricata) pulp by high-performance liquid chromatography with diode array and electrospray ionization mass spectrometric detection. Food Res Int. 2014;65:42-46.
- Arroyo J., Jaime M., Ronceros Gerardo R.P., Aníbal V. Hypoglycemic effect adjuvant extract ethanolic leaf Annona muricata (guanábana), in patients with diabetes type 2 in treatment of glibenclamide. An Fac Med. 2009;70:163-167.
- Agu K.C., Okolie N.P., Eze I., Anionye J.C., Falodun A. Phytochemical analysis, toxicity profile, and hemomodulatory properties of Annona muricata (Soursop). Egypt J Haematol. 2017;42(1):36.
- Gavamukulya Y., Abou-Elella F., Wamunyokoli F., AEl-Shemy H. Phytochemical screening, anti-oxidant activity and in vitro anticancer potential of ethanolic and water leaves extracts of Annona muricata (Graviola). Asian Pac J Trop Med. 2014;7:S355-S363.
- Dailey A., Vuong Q.V. Effect of extraction solvents on recovery of bioactive compounds and antioxidant properties from macadamia (Macadamia tetraphylla) skin waste. Cogent Food Agric. 2015;1(1):1115646.
- Kawakami K., Aketa S., Nakanami M., Iizuka S., Hirayama M. Major water-soluble polyphenols, proanthocyanidins, in leaves of persimmon (Diospyros kaki) and their α-amylase inhibitory activity. Biosci Biotechnol Biochem. 2010;74(7):1380-1385.
- Hossain M.A., Shah M.D. A study on the total phenols content and antioxidant activity of essential oil and different solvent extracts of endemic plant Merremia borneensis. Arab J Chem. 2015;8(1):66-71.
- Lesjak M., Beara I., Simin N., Pintać D., Majkić T., Bekvalac K., Orčić D., Mimica-Dukić >\N. Antioxidant and anti-inflammatory activities of quercetin and its derivatives. J Funct Foods. 2018;40:68-75.
- Ivorra M., Paya M., Villar A. A review of natural products and plants as potential antidiabetic drugs. J Ethnopharmacol. 1989;27(3):243-275.
- Adeyemi D., Komolafe O., Adewole S., Obuotor E. Anti hyperlipidemic activities of Annona muricata (Linn). Internet J Altern Med. 2009;7(1):1-7.
- Florence N.T., Benoit M.Z., Jonas K, Alexandra T., Désiré D. D. P., Pierre K., Théophile D. Antidiabetic and antioxidant effects of Annona muricata (Annonaceae), aqueous extract on streptozotocin-induced diabetic rats. J Ethnopharmacol. 2014;151(2):784-790.
- Adeyemi D.O., Komolafe O.A., Adewole O.S., Obuotor E.M., Adenowo T.K. Anti hyperglycemic activities of Annona muricata (Linn). Afr J Tradit Complementary Altern Med. 2009;6(1):62-69.
- Adeyemi D., Komolafe O., Adewole O., Obuotor E., Abiodun A., Adenowo T. Histomorphological and morphometric studies of the pancreatic islet cells of diabetic rats treated with extracts of Annona muricata. Folia Morphol. 2010;69(2):92-100.
- Adewole S.O., Caxton-Martins E.A. Morphological changes and hypoglycemic effects of Annona muricata(annonaceae) leaf aqueous extract on pancreatic β-cells of streptozotocin-treated diabetic rats. Afr J Biomed Res. 2006;9(3):173-187.
- Adewole S.O., Ojewole J. Immunohistochemical and Biochemical effects of Annona muricata (Annonaceae) leaf aqueous extract on pancreatic beta cells of streptozotocin-treated diabetic rats. Pharmacologyonline. 2006;2:335-355.
- Pinto L.C., Cerqueira-Lima A.T., dos Santos Suzarth S., de Souza R., Tosta B.R., da Silva B., de Oliveira Pires A., de Almeida Queiroz G., Teixeira T.O., Dourado K.M.C. Anonna muricata L. (soursop) seed oil improves type 1 diabetes parameters in vivo and in vitro. PharmaNutrition. 2018;6(1):1-8.
- Hasanah R.U.R., Sundhani E., Nurulita N.A. Effect of ethanolic extract of Annona muricata seeds powder to decrease blood glucose level of wistar male rats with glucose preload. UNEJ e-Proceeding. 2017:112-115.
- Agbai E., Mounmbegna P., Njoku C., Nwanegwo C., Awemu G., Iwuji S. Effect of Annona muricata seed extract on blood glucose, total and differential white cell count after repeated exposure to clozapine. Res Neurosci. 2015;4(1):10-15.
- Ukwubile C.A. Phytochemical screening and anti-ovarian cancer properties of Annona muricata Linn (Annonaceae) seed ethanol extract. Int J Pharm Front Res. 2012;1(3):9-17.
- Komansilan A., Abadi A.L., Yanuwiadi B., Kaligis D.A. Isolation and identification of biolarvicide from soursop (Annona muricata Linn) seeds to mosquito (Aedes aegypti) larvae. Int J Eng Technol. 2012;12(03):28-32.
- Ahalya B., Shankar K.R., Kiranmayi G. Exploration of anti-hyperglycemic and hypolipidemic activities of ethanolic extract of Annona muricata bark in alloxan induced diabetic rats. Int J Pharm Sci Rev Res. 2014;25(2):21-27.
- Adewole S., Ojewole J. Protective effects of Annona muricata (Annonaceae) leaf aqueous extract on serum lipid profiles and oxidative stress in hepatocytes of streptozotocin-treated diabetic rats. Afr J Tradit Complementary Altern Med. 2009;6(1):30-41.
- Hanhineva K., Törrönen R., Bondia-Pons I., Pekkinen J., Kolehmainen M., Mykkänen H., Poutanen K. Impact of dietary polyphenols on carbohydrate metabolism. Int J Mol Sci. 2010;11(4):1365-1402.
- Kim J.S., Kwon C.S., Son K.H. Inhibition of alpha-glucosidase and amylase by luteolin, a flavonoid. Biosci Biotechnol Biochem. 2000;64(11):2458-2461.
- Ramakrishna V., Jailkhani R. Evaluation Of Oxidative Stress In Insulin Dependent Diabetes Mellitus (IDDM) Patients. Diagn Pathol. 2007;2(1):22-28.
- Martinez-Sanchez G., Popov I., Perez-Davison G., Al-Dalaen S., Horwat-Delaporte R., Giuliani A., Leon-Fernandez O.S. Contribution to characterization of oxidative stress in diabetic patients with macroangiopatic complications. Acta Farm Bonaerense. 2005;24(2):197-203.
- Matough F.A., Budin S.B., Hamid Z.A., Alwahaibi N., Mohamed J. The role of oxidative stress and antioxidants in diabetic complications. Sultan Qaboos Univ Med J. 2012;12(1):5-18.
- Berawi K.N., Shidarti L., Nurdin S.U., Lipoeto N.I., Wahid I. Comparison Effectiveness of Antidiabetic Activity Extract Herbal Mixture of Soursop Leaves (Annona Muricata), Bay Leaves (Syzygium Polyanthum) and Pegagan Leaves (Centella Asiatica). Biomed Pharmacol J. 2017;10(3):1481-1488.
- Justino A.B., Miranda N.C., Franco R.R., Martins M.M., da Silva N.M., Espindola F.S. Annona muricata leaf as a source of antioxidant compounds with in vitro antidiabetic and inhibitory potential against α-amylase, α-glucosidase, lipase, non-enzymatic glycation and lipid peroxidation. Biomed Pharmacother. 2018;100:83-92.
- Davì G., Falco A., Patrono C. Lipid peroxidation in diabetes mellitus. Antioxid Redox Signal. 2005;7(1-2):256-268.
- Hardoko Y.H., Wijoyo S., Halim Y. In vitro antidiabetic activity of “green tea” soursop leaves brew through α-glucosidase inhibition. Int J PharmTech Res. 2015;8(1):30-37.
- Adefegha S.A., Oyeleye S.I., Oboh G. Distribution of phenolic contents, antidiabetic potentials, antihypertensive properties, and antioxidative effects of soursop (Annona muricata ) fruit parts in vitro. Biochem Res Int. 2015;2015:1-8.
- Damayanti D.S., Utomo D.H., Kusuma C. Revealing the potency of Annona muricata leaves extract as FOXO1 inhibitor for diabetes mellitus treatment through computational study. In silico Pharmacol. 2017;5(1):3-10.
- Martinez S.C., Tanabe K., Cras-Méneur C., Abumrad N.A., Bernal-Mizrachi E., Permutt M.A. Inhibition of Foxo1 protects pancreatic islet β-cells against fatty acid and endoplasmic reticulum stress–induced apoptosis. 2008;57(4):846-859.
- Nagashima T., Shigematsu N., Maruki R., Urano Y., Tanaka H., Shimaya A., Shimokawa T., Shibasaki M. Discovery of novel forkhead box O1 inhibitors for treating type 2 diabetes: improvement of fasting glycemia in diabetic db/db mice. Mol Pharmacol. 2010;78(5):961-970.
- Kamalakkannan N., Prince P.S.M. Antihyperglycaemic and antioxidant effect of rutin, a polyphenolic flavonoid, in streptozotocin‐induced diabetic wistar rats. Basic Clin Pharmacol Toxicol. 2006;98(1):97-103.
- Ojewole J.A. Antinociceptive, anti-inflammatory and antidiabetic effects of Bryophyllum pinnatum (Crassulaceae) leaf aqueous extract. J Ethnopharmacol. 2005;99(1):13-19.

This work is licensed under a Creative Commons Attribution 4.0 International License.






